Abstract
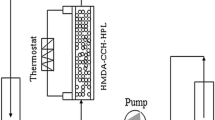
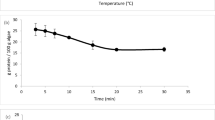
This study investigated the production behavior of amino acids from polypeptone under various operation parameters and reactor modes (batch and continuous). In the batch experiments, the effect of heating time on amino acid production was studied using different type of reactor materials (Hastelloy (HAS) and SUS). The results showed that the production behavior of amino acids such as Gly, Ala, and Leu was quite similar regardless of reactor materials. To further investigate the relationship of reaction time on the amino acids production, experiments were conducted in batch and continuous reaction at the same conditions (T=523 K and P=30 MPa). The results showed that it is possible to get the same result at the same reaction time using a batch and continuous reaction mode if the reaction time is started after heating time in a batch reaction mode. From the study of reaction pressure, it was observed that the production of amino acids is not favored at reaction pressure between 4 and 30 MPa at 523 K.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Aki, S. and Abraham, M. A., “Catalytic supercritical water oxidation of pyridine: comparison of catalysts,”Ind. Eng. Chem. Res.,38, 358 (1999a).
Aki, S. and Abraham, M. A., “Catalytic supercritical water oxidation of pyridine: kinetics and mass transfer,”Chem. Eng. Sci.,54, 3533 (1999b).
Anitescu, G. Z., Zhang and Tavlarides, L. L., “A kinetic study of methanol oxidation in supercritical water,”Ind. Eng. Chem. Res.,38, 2231 (1999).
Daimon, H., Kang, K., Sato, N. and Fujie, K., “Development of marine waste recycling technologies using sub- and supercritical water,”J. Chem. Eng. Japan,34, 1091 (2001).
Kang, K., Quitain, A. T., Urano, S., Daimon, H. and Fujie, K., “Rapid sample injection in semi-batch hydrothermal treatment of solid wastes,”Ind. Eng. Chem. Res.,40, 3717 (2001).
Kang, K. Y. and Chun, B. S., “Behavior of hydrothermal decomposition of silk fibroin to amino acids in near-critical water,”Korean J. Chem. Eng.,21, 654 (2004).
Kang, K. Y. and Chun, B. S., “Behavior of amino acid production from hydrothermal treatment of fish-derived wastes,”Korean J. Chem. Eng.,21, 1147 (2004).
Krammer, P. and Vogel, H., “Hydrolysis of esters in subcritical and supercritical water,”J. Supercrit. Fluids,16, 189 (2000).
Lachance, R., Paschkewitz, J., DiNaro, J. and Tester, J. W., “Thiodiglycol hydrolysis and oxidation in sub- and supercritical water,”J. Supercrit. Fluids,16, 133 (1999).
Martino, J. C. and Savage, P. E., “Oxidation and thermolysis of methoxy-, nitro-, and hydroxy-substituted phenols in supercritical water,”Ind. Eng. Chem. Res.,38, 1784 (1999).
Meyer, J. C., Marrone, P. A. and Tester, J. W., “Acetic acid oxidation and hydrolysis in supercritical water,”AIChE J.,41, 2108 (1995).
Pitzer, K. S., “Self-ionization of water at high temperature and the thermodynamic properties of the ions,”J. Phys. Chem.,86, 4704 (1982).
Sasaki, M., Kabyemela, B., Malaluan, R., Hirose, S., Takeda, N., Adschiri, T. and Arai, K., “Cellulose hydrolysis in subcritical and supercritical water,”J. Supercrit. Fluids,13, 261 (1998).
Savage, P. E., Gopalan, S., Mizan, T. I., Martino, C. J. and Brock, E. E., “Reactions at supercritical conditions: applications and fundamentals,”AIChE J.,41, 1723 (1995).
Tanger IV, J. C. and Pitzer, K. S., “Calculation of the ionization constant of H2O to 2,273 K and 500 MPa,”AIChE J.,35, 1631 (1989).
Yoshida, H., Terashima, M. and Takahashi, Y., “Production of organic acids and amino acids production from fish meat by subcritical water hydrolysis,”KagakuKougyou,50, 57 (1999).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kang, KY., Lim, JH. & Kim, YH. Effects of various reaction conditions on the hydrothermal treatment of polypeptone. Korean J. Chem. Eng. 23, 81–84 (2006). https://doi.org/10.1007/BF02705696
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02705696