Abstract
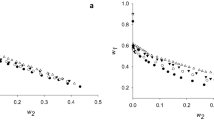
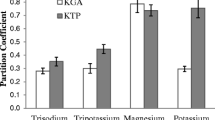
This article presents a study of polyethylene glycol 1000 (PEG1000)/potassium phosphate aqueous two-phase systems (ATPSs) forBacillus subtilis NS99 alkaline protease extraction. The objectives were to evaluate effects of system pH (7.5, 8.5,9.5, and 10.5), and NaCl concentration (0,4,7, and 10% (w/w)) on ATPS binodal curves, effects of system pH, NaCl concentration, and tie-line length (TLL) on alkaline protease partition coefficient (K) and yield (Y%) at room temperature (30±2 ‡C). Casein hydrolysis was used for determination of alkaline protease activity. It was revealed that system pH had the slightest effect on locations of binodal curves (except at pH 10.5). In contrast, addition of NaCl appeared to have a significant effect on phase characteristics since binodal curves of systems with NaCl (4-10% (w/w)) shifted significantly towards the origin in comparison to the ones without NaCl. Increased NaCl concentration from 4 to 10% (w/w), however, showed trivial influence on locations of the binodal curves. Changes of system compositions due to variation in system pH, TLL, and NaCl concentrations obviously resulted in varied obtainable K and Y% of alkaline proteases. Longer TLL and higher pH generally resulted in higher K. In contrast, the lower NaCl concentration, the higher K. Since the same phase volume ration (1:1) was used throughout the experiments, Y% depended solely on K. The most suitable PEG1000/potassium phosphate ATPS was determined at pH 9.5, and comprised PEG1000, potassium phosphate, and NaCl 18.0,13.0, and 0% (w/w), respectively. This system resulted in considerably high K, and Y% of 20.0, and 95.1%, respectively. Information from this study will be important for further development of an ATPS extraction unit for alkaline protease recovery.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
Albertsson, P. A.,Partition of cell particles and macromolecules, Wiley, New York (1986).
Capezio, L., Romanini, D., Pico, G. A. and Nerli, B., “Partition of whey milk proteins in aqueous two-phase systems of polyethylene glycolphosphate as a starting point to isolate proteins expressed in transgenic milk,”J. Chromatogr. B,819, 25 (2005).
Chouyyok, W., Wongmongkol, N., Siwarungson, N. and Prichanont, S., “Extraction of alkaline protease using aqueous two-phase system from cell freeBacillus subtilis TISTR 25 fermentation broth,“Process Biochem.,40, 3514 (2005).
Conway, B. E.,Ionic hydration in chemistry and biophysics, Elsevier, Amsterdam (1981).
Cunha, M. T., Aires-Barros, M. R. and Cabral, J. M. S.,Extraction for rapid protein isolation, in: Hatti-Kaul, R. and Mattiasson, B. (Eds.),Isolation and purification of proteins, Marcel Dekker, Inc., U.S.A., 321 (2003).
Grebeskova, R. N., Ryshlava, J. M., Fedorova, L. G., Kochetova, S. P., Babloyan, O. O. and Vinogredona, G. L., “Using alkaline protease to intensify the processing of leather raw material,”Biotechnology,4, 788 (1988).
Haraguchi, L. H., Mohamed, R. S., Loh, W. and Pessoa Filho, P. A., “Phase equilibrium and insulin partitioning in aqueous two-phase systems containing block copolymers and potassium phosphate,”Fluid Phase Equilibr.,215, 1 (2004).
Hong, D. P., Kuboi, R. and Komasawa, I., “Extraction of proteins and polymers using reverse micelle and percolation process,”Korean J. Chem. Eng.,14, 334 (1997).
Hotha, S. and Banik, R. M., “Production of alkaline protease byBacillus thuringiensis H14 in aqueous two-phase systems,”J. Chem. Tech. Biotechnol.,69, 5 (1997).
Isable, D. V. M. and Otero, C., “Biphasic aqueous media containing polyethylene glycol for the enzymatic synthesis of oligosaccharides from lactose,”Entyme Microbiol. Technol.,33, 118(2003).
Johansson, H. O., Lundh, G., Karlstrom, G. and Tjerneld, F., “Effects of hydrophobicity and counter ions on the partitioning of amino acids in thermoseparating Ucon-waler two-phase systems,”Bioseparation,5, 269 (1995).
Kepka, C., Collet, E., Persson, J., Stahl, A., Lagerstedt, T., Tjerneld, F. and Veide, A., “Pilot-scale extraction of an intracellular recombinant cutinase fromE. coli cell homogenate using a thermoseparating aqueous two-phase system,”J. Biotechnol.,103, 165 (2003).
Klomklao, S., Benjakul, S., Visessanguan, W., Simpson, B. K. and Kishimura, H., “Partitioning and recovery of proteinase from tuna spleen by aqueous two-phase systems,”Process Biochem.,40, 3061 (2005). Krestov, G. A.,Thermodynamics of ionic processes in solutions (Rus), Khimia, Liningrad (1984).
Lima, A. S., Alegre, R. M. and Meirelles, A. J. A., “Partitioning of pectinolytic enzymes in polyethylene glycol/potassium phosphate aqueous two-phase systems,”Carbohyd. Polym.,50, 63 (2002).
Marcos, J. C., Fonseca, L. P., Ramalho, M. X. and Cabral, J. M. S., “Application of surface response analysis to optimization of penicillin acylase purification in aqueous two-phase systems,”Enz. Microb. Technol.,31, 1006(2002).
Ng, T. K. and Wenealy, W. R.,Industrial applications of thermostable enzymes, Wiley, New York (1986).
Oliveira, L. A., Sarubbo, L. A., Porto, A. L. F., Campos-Takaki, G. M. and Tambourgi, E. B., “Partition of trypsin in aqueous two-phase systems of poly(ethylene glycol) and cashew-nut tree gum,”Process Biochem.,38, 693 (2002).
Owen, P. W., InMicrobial enzymes and biotechnology, ed. W. M. Fogarty, Applied Science, London, p. 270 (1983).
Park, D. H., Lee, H. J. and Lee, E. K., “Crystallization of alkaline pretease as a means of purification process,”Korean J. Chem. Eng.,14, 64 (1997).
Phadatara, S. V., Deshpande, V. V. and Srinivasan, M. C., “High activity alkaline protease fromConidioboulus coronatus: enzyme production and compatibility with commercial detergents,”Enz. Microbiol. Technol.,15, 72 (1993).
Reh, G., Nerli, B. and Pico, G., “Isolation of alpha-1-antitrypsin from human plasma by partitioning in aqueous biphasic systems of polyethyleneglycol-phosphate,”J. Chromatogr. B,780, 389 (2002).
Sebastiao, M. J., Cabral, J. M. S. and Aires-Barros, M. R., “Fusarium solanipisi recombinant cutinase partitioning in PEG/potassium phosphate aqueous two-phase systems,”Biotechnol. Tech.,7, 631 (1993).
Sinha, R., Singh, S. P., Ahmed, S. and Garg, S. K., “Partitioning of aBacillus alkaline protease in aqueous two-phase systems,”Biores. Technol.,55, 163(1996).
Wilson, S. A., Young, O. A., Coolbear, T. and Daniel, R. M., “The use of proteases from extreme thermophiles for meat tenderization,”Meat Science,32, 93 (1992).
Xu, Y., Vitolo, M., Albuquerque, C. N. and Pessoa, A. Jr., “Affinity partitioning of glucose-6-phosphate dehydrogenase and hexokinase in aqueous two-phase systems with free triazine dye ligands,”J. Chromatogr. B,780, 53 (2002).
Zaslavsky, B. Y., Miheeva, L. M., Rodnikova, M. N., Spivak, G. V., Harkin, V. S. and Mahmudov, A. U.,J. Chem. Soc., Faraday Trans. I,85, 2857(1989).
Zaslavsky, B. Y.,Aqueous two-phase partitioning: Physical chemistry and bioanalytical applications, Marcel Dekker, Inc., U.S.A. (1995).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wongmongkol, N., Prichanont, S. Partition of alkaline protease in aqueous two-phase systems of polyethylene glycol 1000 and potassium phosphate. Korean J. Chem. Eng. 23, 71–76 (2006). https://doi.org/10.1007/BF02705694
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02705694