Abstract
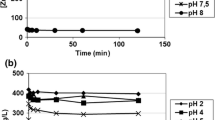
Potentially, biosorption is an economic process for metal sequestering from water. Carboxylated alginic acid showed high uptake capacities for heavy metals of 5-6 meq/g dry mass. For application to actual plating waste-water, the carboxylated alginic acid was immobilized using PVA. In order to remove chelating or organic materials in plating wastewater, oxidation using sodium hypochlorite was performed as a pretreatment. When carboxylated alginic acid bead was applied in a packed-bed contactor, the breakthrough point of copper ion in the acid-alkaline wastewater appeared around 350 bed volumes; the breakthrough point of nickel ion in the chelating wastewater emerged around 200 bed volumes. The adsorption capacity for heavy metal of the carboxylated alginic acid bead was higher than that of a commercial ion exchanger (IR-120 plus) in plating wastewater.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
Bolto, B. A. and Pawlowski, L., “Wastewater Treatment by Ion-Exchange,” lth ed., Great Britain, New York, U.S.A., 209 (1988).
Chen, J. P., Chen, W. R. and Hsu, R. C., “Biosorption of Copper from Aqueous Solutions by Plant Root Tissues,”J. Ferment. Bioeng.,81, 458 (1996).
Dawson, R. M. C., Elliot, D. C., Elliot, W. H. and Jones, K. M., “Data for Biochemical Research,” 3rd ed., Oxford Science Pub., 250 (1986).
Franson, H. A. N., “Standard Method,” Port City Press, Maryland, U.S.A. (1989).
Jeon, C., Park, J. Y. and Yoo, Y. J., “Immobilization of Alginic Acid Using PVA for Lead Adsorption;” 216th ACS National Meeting, Boston, August 21–28 (1998).
Joseph, D. E., “Industrial Wastewater Treatment (A Guide Book);” 1st ed., CRC Press, Inc., New York, 51 (1995).
Kim, H. T. and Lee, K., “Application of Insoluble Cellulose Xanthate for the Removal of Heavy Metals from Aqueous Solution,”Korean J. Chem. Eng.,16, 298 (1999).
Kim, J. S., Chah, S. and Yi, J., “Preparation of Modified Silica for Heavy Metal Removal,”Korean J. Chem. Eng.,17, 118 (2000).
Kim, S. J., Hwang, K. R. and Cho, S. Y., “Simultaneous Removal of Cyanide and Copper Ions in a Semi-Fluidized Exchanger Bed,”Korean J. Chem. Eng.,16, 664 (1999).
Kratochvil, D., Pimentel, P. and Voleky, B., “Removal of Trivalent and Hexavalent Chromium by Seaweed Biosorbent,”Environ. Sci. Technol.,32, 2693 (1998).
Lee, D. H., Kim, S. J. and Moon, H., “Preparation of a Clinnoptilolite-Type Korean Natural Zeolite,”Korean J.Chem. Eng,16, 525 (1999).
Lee, H. S. and Suh, J. H., “Continuous Biosorption of Heavy Metal Ions by Ca-Loaded Laminaria Japonica in Fixed Bed Column,”Korean J.Chem. Eng.,17, 477 (2000).
Muthukumran, K., Balasubramanian, N. and Ramakrushna, T. V., “Removal and Recovery of Chromium from Plating Waste Using Chemically Activated Carbon,”Water Sci. Technol.,31(9), 109 (1995).
Park, J. Y., Jeon, C. and Yoo, Y J., “Characteristics of Metal Biosorption of Oxidized Undaria Pinnatifida,”J.Microbiol. Biotechnol.,9(5), 650 (1999).
Patterson, J. W., “Industrial Wastewater Treatment Technology,” 2nd ed., Butterworth, 156 (1985).
Pradhan, S., Sarita, S., Lal, C. R. and Dorothy, L. P., “Evaluation of Metal Biosorption Efficiency of Laboratory GrownMicrocystis Under Various Environmental Conditions,”J.Microbiol. Biotechnol.,8, 53 (1998).
Volesky, B., “Biosorption of Heavy Metals,”1st ed., CRC Press, Inc., New York, 320 (1990).
Watson, M. R., “Pollution Control in Metal Finishing,” 1st ed., NDC, 253 (1987).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Jeon, C., Park, J.Y. & Yoo, Y.J. Removal of heavy metals in plating wastewater using carboxylated alginic acid. Korean J. Chem. Eng. 18, 955–960 (2001). https://doi.org/10.1007/BF02705625
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF02705625