Abstract
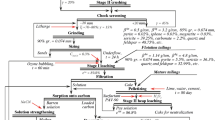
As ore grades drop at today’s large mines, and environmental regulations for waste discharges increase, economics drives new recovery innovations. This paper describes some technological advances in the recovery processes for copper, gold, silver, lead and zinc. It does not discuss pyrometallurgy. Korea, like many nations with industrial economies, consumes large amounts of these metals, and is dependent on overseas suppliers for nearly its entire supply. This paper also discusses how technology for recovery of metal now in Korea, for example from existing wastes, is important. Improved reagents for both leaching and froth flotation of copper minerals, plus bio-oxidation and pressure autoclaving for gold have improved metal recovery. Copper recovery by SX-EW is expanding rapidly, and the method is also commercial for zinc. Biooxidation of encapsulating waste materials and, in some instances, of the desired metal has reached commercial scale. Pressure leaching on a large scale solves specific problems for several metals. Improvements in gold and silver cyanidation include variants of activated carbon adsorption from solution. Zinc and lead still largely depend on flotation for the treatment of ores. However, innovation in hydrometallurgical extraction has been spurred by environmental concerns. Computer modeling and process control worldwide have likely led to the largest improvements in recovery. The limited availability of land, clean water and sites for waste disposal in many countries affects the economics of re-treatment of existing wastes. Some of the new technologies could be combined, for example in modular plants that can be moved between reclamation sites to economic advantage on the Korean Peninsula.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Alpers, C. N and Blowes, D. W., “The Environmental Geochemistry of Sulfide Oxidation” Washington, DC, American Chemical Society (1990).
Avecia, Inc., “Acorga Technical Library;” CD ROM, Wilmington (2001).
Brierley, C., “Commercial Status of Bioleaching,” IN Randol Gold & Silver Forum ’99, Denver:, Randol International Inc., 137 (1999).
Brittan, M. and Arthur, B., “Using Diagnostic Process Analysis to Improve Cash Flow At Newmonts Carlin Mill # 4”Mining Engineering,52, 37 (2000).
Cathles, L. M, “Personal Perspectives from Attempts to Model the Industrial Scale Leaching of Copper-Bearing Mine Waste,” In The Environmental Geochemistry of Sulfide Oxidation (ed. C.N. Alpers and D. W Blowes, pp. 123–31), Washington, DC, American Chemical Society (1994).
Chae, Y.-B., “Recovery of Available Resources from Gold Mining Waste,”Jour. Of Korean Recycling,8, 37 (1999).
Chon, H.-T, Ahn, J. S. and Jung, M. C., “Heavy Metal Contamination of Some Abandoned Base Metal and Au-Ag Mines in Korea: A Review,” Proceeding of the Korean Society of Groundwater and Environment’99, 51 (1999).
Cope, L. W., “Chloride Process Successfully Recovers Silver at the Itos Plant in Bolivia,”Eng. and Min. Jour.,201, 26 (2000).
De la Vergne, J. R., “Hard Rock Miners Handbook (CD),” Tempe: Mc-Intosh Redpath Engineering Inc., 2nd edition (2001).
DeVoto, R. H. and McNulty, T. P., “Banning Cyanide Use at McDonald (U.S.A.);Mining Engineering,52, 19 (2000).
Free, M. L., Oolman, T., Nagpal, S. and Dahlstrom, D. A., “Bioleaching of Sulfide Ores — Distinguishing Between Indirect and Direct Mechanisms;“ In Ross W. Smith and Manoranjan Misra, eds., Mineral Bioprocessing, TMS, Warrendale, PA, 485 (1991).
Garcia, C., Ballester, A., Gonzalez, F., Blazquez, M. L. and Acosta, M., “Chemical and Microbiological Transformations in a Pyritic Tailing Pond,”Minerals Engineering,9, 1127 (1996).
Hiskey, J. B. and Sanchez, V. M., “Alkaline Pressure Oxidation of Gold-Bearing Arsenopyrite Concentrates,,”Mineral Processing and Extractive Metallurgy Review,15, 65 (1995).
James, L. P., “Metalliferous Recent Sediments? Mineralogical and Historical Approaches to the Study of Utah Mill Tailings,,” In H. Doelling, ed. Energy and Mineral Resources of Utah, Utah Geological Association Publication,18, 59 (1990).
Jones, W. G., Clauber, C. and Linge, H. G., “Fundamental Aspects of Gold Cyanide Adsorption on Activated Carbon,,” In R. Bhappu and R. Harden, Eds., Gold Forum on Technology and Practices, Littleton, Colorado, Society for Mining, Metallurgy and Exploration, Inc., 278 (1989).
Kawatra, S. K. and Natarajan, K. A., “Mineral Biotechnology: Microbial Aspects of Mineral Beneficiation, Metal Extraction and Environmental Control,,” Denver: Society for Mining, Metallurgy and Exploration (2001).
Kral, S., “Tailings and Mine Waste 01,”Mining Engineering,53, 25 (2001).
Langmuir, D., “Aqueous Environmental Chemistry,,” New Jersey, Prentice-Hall Simon and Shuster, 598 (1997).
Ness, V., “Alternatives to Conventional Mixer Settlers,,” In Randol Cooper Hydrometallurgy Roundtables ’99, Denver, Randol International Inc., 29 (1999).
Park, M. E., Sung K. Y. and Koh, Y. K., “Formation of Acid Mine Drainage and Pollution of Geological Environment Accompanying Sulfidation Zone of Nonmetallic Deposits: Reaction Path Modeling of the Formation of AMD at Tongnae Pyrophyllite Mine,”Econ. Environ. Geol.,33, 405 (2000).
Prasad, A. and Pandey, B. D., “Alternative Processes for Treatment of Chalcopyrite — a Review,”Minerals Engineering,11, 763 (1998).
Shuey, S. A., “Bioleaching: the Next Era in Refractory Mineral Processing.”Eng and Min. Jour.,200, 16BB (1999).
Spence, J. R. and Soderstrom, M. D., “Practical Aspects of Copper Solvent Extraction from Acidic Leach Liquors,” SME Annual Meeting, Denver, 19(1999).
Wells, J., “New Developments in Extractive Metallurgical and Mineral Processing, What these may mean for the Definition of “Ore” and “Ore Reserves,” P. and D., Assoc. of Canada Short Course Notes, Toronto, 239 (1999).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
James, L.P., Cooksey, W.H., Park, ME. et al. Economic and environmental applications for recent innovations of nonferrous metallurgy in some industrial nations. Korean J. Chem. Eng. 18, 948–954 (2001). https://doi.org/10.1007/BF02705624
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF02705624