Abstract
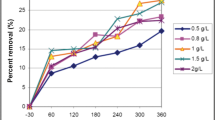
The testing samples in this experiment were obtained from an acrylic fibre manufacturing companys industrial wastewater. The water was the waste of the acrylic polymerization process. The company is located in Ulsan, Korea. The concentration of acrylonitrile (AN) in the wastewater was about 25–35 mg/L. Concentrations of 3–10 mg/ L of methyl acrylate (M-35) were also found. The samples were treated by the TiO2/UV system and were analyzed to determine the values of CODcr, ammonia, nitrite nitrogen, and nitrate nitrogen by using an Auto Analyzer (Bran+ Luebbe, Germany) and a TOC (Tekmar Dohrmann, USA). Various reaction parameters, such as TiO2 content, light intensity and wavelength, and the number of UV lamps were varied and their effects or decomposition efficiency were analyzed. The adsorption onto TiO2 surfaces by organic materials in the wastewater was negligible. The reaction-rate constant was also calculated. The reaction rate constant for the G36T6L lamp at both 185 nm and 256 nm was 0.0661 hr-1 which is 1.3 times higher than that of the TUV36T5 lamp at 256 nm. While the reaction rate was increased by increasing the surface area of the photocatalyst, the excess photocatalyst blocked the light sources, causing a photoenclosure effect. The stability of the treated wastewater was greatly increased because the elimination of the concentration of nitrite was followed by an increase in the concentration of nitrate. Generally, the ratio of BOD5/CODcr is used as the criterion for determining biodegradability. A ratio of 0.3 is needed for biological degradation. The ratio of the treated wastewater increased to 0.5 after 12 hours of reaction. The ratio increased to 0.8 after 20 hours.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
Abdullah, M., Low, G.K. and Matthews, R.W., “Effects of Common Inorganic Anions on Rates of Photocatalytic Oxidation of Organic Carbon over Illuminated Titanium Dioxide,”J. Phys. Chem.,94, 6820 (1990).
Augugliaro, J. Blanco, Gálvez, J. Cáceres, Vázquez, E. García, López, V. Loddo, López, Muñoz M. J., Rodríguez, S. Malato, MarcÌ, G., Palmisano, L., Schiavello, M. and Ruiz, J. Soria, “Photocatalytic Oxidation of Cyanide in Aqueous TiO2 Suspensions Irradiated by Sunlight in Mild and Strong Oxidant Conditions,”Catalysis Today,54, Issues 2–3, 245 (1999).
Bozzi, A., Guasaquillo, I. and Kiwi, J., “Accelerated Removal of Cyanides from Industrial Effluents by Supported TiO2 Photo-catalysts,”Applied Catalysis B: Environmental,51, Issue 3, 203 (2004).
Chen, H.Y., Zahraa, O. and Bouchy, M., “Inhibition of Adsorption and Photocatalytic Degradation of an Organic Contaminant in Aqueous Suspension of TiO2 by Inorganic Ions,”J. Photochem. & Photobio. A: Chem.,108, 37 (1997).
Chu, W., Jafvert, C. T., Diehl, C.A., Marley, K. and Larson, R. A., “Phototransformations of Polychlorobiphenyls in Brij 58 Micellar Solutions,”Environ. Sci. Technol.,32(13), 1989 (1998).
Chun, H. and Wang, Y., “Decolorization and Biodegradability of Photocatalytic Treated Azo Dyes and Wool Textile Wastewater,”Chemosphere,39(12), 2107 (1999).
Daneshvar, N., Rabbani, M., Modirshahla, N. and Behnajady, M.A., “Kinetic Modeling of Photocatalytic Degradation of Acid Red 27 in UV/TiO2 Process,”Journal of Photochemistry and Photobiology A: Chemistry,168, Issues 1–2, 39 (2004).
Hong, Z., Suohong, X., Junbo, Z. and Xinhe, B., “Kinetic Study on the Photo-catalytic Degradation of Pyridine in TiO2 Suspension Systems,”Catalysis Today,93, 857 (2004).
Lee, J. C., Kim, M. S., Kim, C.K., Chung, C.H., Cho, S. M., Han, G.Y., Yoon, K. J. and Kim, B.W. “Removal of Paraquat in Aqueous Suspension of TiO2 in an Immersed UV Photoreactor,”Korean J. Chem. Eng.,20(5), 862 (2003).
Na, Y. S., Kim, D.H., Lee, C.H., Lee, S.W., Park, Y. S., Oh, Y. K., Park, S.H. and Song, S.K., “Photocatalytic Decolorization of Rhodamine B (RhB) by Fluidized Bed Reactor with Hollow Ceramic Ball Photocatalyst,”Korean J. Chem. Eng.,21(2), 430 (2004).
Nigam, P., Banat, I. M., Singh, D. and Marchant, R., “Microbial Process for the Decolorization of Textile Effluent Containing Azo/Diazo and Reactive Dyes,”Process Biochemistry,31(5), 435 (1996).
Wang, K.H., Hsieh, Y.H., Wu, C.H. and Chang, C.Y., “The pH and Anion Effects on the Heterogeneous Photocatalytic Degradation of o-Methyl Benzoic Acid in TiO2 Aqueous Suspension,”Chemosphere,40, 389 (2000).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Na, Y.S., Lee, C.H., Lee, T.K. et al. Photocatalytic decomposition of nonbiodegradable substances in wastewater from an acrylic fibre manufacturing process. Korean J. Chem. Eng. 22, 246–249 (2005). https://doi.org/10.1007/BF02701492
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02701492