Abstract
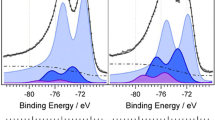
To reduce the effect of methanol permeated from the anode, the structure of the cathode was modified from a single layer with Pt black catalyst to two-layer with PtRh black and Pt black catalysts, respectively. The current density of the direct methanol fuel cell (DMFC) using the two-layer cathode was improved to 228 mA/cm-2 compared to that (180 mA/cm-2) of the DMFC using the single layer cathode at 0.3 V and 303 K. From the cyclic voltammograms (CVs), it is indicated that the amount of adsorbates on the metal catalyst in the two-layer cathode is less than that of adsorbates in the single layer cathode after methanol test. In addition, the adsorbates were removed very rapidly by electrochemical oxidation from the two-layer cathode. It is suggested fromex situ X-ray absorption near edge structure analysis that the d-electron vacancy of Pt atom in the two-layer cathode is not changed by the methanol test. Thus, Pt is not covered with the adsorbates, which agrees well with the results of CV.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
AricÒ, A., CretÌ, P., Kim, H., Mantegna, R., Giordano, N. and Antonucci, V., “Analysis of the Electrochemical Characteristics of a Direct Methanol Fuel Cell Based on a Pt-Ru/C Anode Catalyst,”J. Electrochem. Soc.,143, 3950 (1996).
Bedrane, S., Descorme, C. and Duprez, D., “Towards the Comprehension of Oxygen Storage Processes on Model Three-way Catalysts,”Catal. Today,73, 233 (2002).
Chang, H., Kim, J. R., Cho, J.H., Kim, H.K. and Choi, K.H., “Materials and Processes for Small Fuel Cells,”Solid State Ionics,148, 601 (2002).
Elliott, J.M., Birkin, P. R., Barlett, P.N. and Attard, G. S., “Platinum Microelectrodes with Unique High Surface Areas,”Langmuir,15, 7411 (1999).
Friedrich, K.A., Geyzers, K. P., Dickinson, A. J. and Stimming, U., “Fundamental Aspects in Electrocatalysis: from the Reactivity of Singlecrystals to Fuel Cell Electrocatalysts,”J. Electroanal. Chem.,524, 261 (2002).
Kim, H.K., Cho, J.H. and Chang, H.,Hybrid Polymer Electrolyte to Reduce the Fuel Cross-over in DMFC, Proceeding of 201th ECS symposium, Abstract No 180, Philadelphia, USA (2002).
Koch, D.F.A., Rand, D.A. J. and Woods, R., “Binary Electrocatalysts for Organic Oxidations,”J. Electroanal. Chem.,70, 73 (1976).
Lee, S.-A., Park, K.W., Kwon, B.-K. and Sung, Y.-E., “PtRu Nanoparticle Electrocatalysts Prepared by Various Synthetic Methods for Use in Direct Methanol Fuel Cells,”J. Ind. Eng. Chem.,9, 63 (2003).
Lee, S. J., Mukerjee, S., McBreen, J., Rho, Y.W., Kho, Y. T. and Lee, T.H., “Effects of Nafion Impregnation on Performances of PEMFC Electrodes,”Electrochim. Acta,43, 3693 (1998).
Lee, S. J., Mukerjee, S., Ticianelli, E. A. and MeBreen, J., “Electrocatalysis of CO Tolerance in Hydrogen Oxidation Reaction in PEM Fuel Cells,”Electrochim. Acta,44, 3283 (1999).
Lee, C. S. and Yi, S.C., “Numerical Methodology for Proton Membrane Fuel Cell Simulation using Computational Fluid Dynamics Technique,”Korean J. Chem. Eng.,21, 1153 (2004).
Ma, Z. Q., Cheng, P. and Zhao, T. S., “A Palladium-alloy Deposited Nafion Membrane for Direct Methanol Fuel Cells,”J. Membrane Science,215, 327 (2003).
Markovic, N.M., Gasteiger, H.A., Ross, Jr., P.N., Jiang, X., Villegas, I. and Weaver, M. J., “Electro-oxidation Mechanisms of Methanol and Formic Acid on Pt-Ru Alloy Surfaces,”Electrochim. Acta,40, 91 (1995).
Miyake, M., Wainright, J. S. and Savinell, R. F., “Evaluation of a Sol-Gel Derived Nafion/Silica Hybrid Membrane for Polymer Electrolyte Membrane Fuel Cell Applications: II. Methanol Uptake and Methanol Permeability,”J. Electrochem. Soc.,148, A905 (2001).
Morimoto, Y. and Yeager, E. B., “Comparison of Methanol Oxidations on Pt, Pt®u and Pt¦Sn Electrodes,”J. Electroanal. Chem.,444, 95 (1998).
Mukerjee, S., Lee, S. J., Ticiannelli, E.A., McBreen, J., Grgur, B.N., Markovic, N. M., Ross, P.N., Giallombardo, J. R. and De Castro, E. S., “Investigation of Enhanced CO Tolerance in Proton Exchange Membrane Fuel Cells by Carbon Supported PtMo Alloy Catalyst,”Electrochem. Solid State Lett.,2, 12 (1999).
Novakova, J., “Reduction of NO by Hydrogen Versus Reduction by CO over Pt, Pd and Rh Clusters in NaX Zeolite,”Appl. Catal. B: Environ.,30, 445 (2001).
O’Grady, W. E., Hagans, P. L., Pandya, K. I. and Mariche, D. L., “Structure of Pt/Ru Catalysts Using X-ray Absorption Near Edge Structure Studies,”Langmuir,17, 3047 (2001).
Park, S.G., “Pervaporation Characteristics of Polyetherimide/ γ-Alumina Composite Membrane for a Quaternary Equilibrium Mixture of Acetic Acid-Ethanol-Ethyl Acetate-Water,”Korean J. Chem. Eng.,21, 882 (2004).
Ross, P.N., Kinoshita, K., Scarpellino, A. J. and Stonehart, P., “Electrocatalysis on Binary Alloys: I. Oxidation of Molecular Hydrogen on Supported Pt-Rh Alloys,”Electroanal. Chem. Interfa. Electrochem.,59, 177 (1975).
Russell, A. E., Maniquet, S., Mathew, R. J., Yao, J., Roberts, M. A. and Thompsett, D., “Insitu X-ray Absorption Spectroscopy and X-ray Diffraction of Fuel Cell Electrocatalysts,”J. Power Sources,96, 226 (2001)
Santra, A.K. and Goodman, D.W., “Catalytic Oxidation of CO by Platinum Group Metals: from Ultrahigh Vacuum to Elevated Pressures,”Electrochim. Acta,47, 3595 (2002).
de Souza, J. P. I., Queiroz, S. L., Bergamaski, K., Gonzalez, E. R. and Nart, F. C., “Electro-Oxidation of Ethanol on Pt, Rh, and PtRh Electrodes. A Study Using DEMS and in-Situ FTIR Techniques,”J. Phys. Chem. B,106, 9825 (2002).
Teo, B.K.,EXAFS: Basic Principles and Data Analysis, Springer-Verlag, New York, USA (1986).
Thomas, S. C., Ren, X., Gottesfeld, S. and Zelenay, P., “Direct Methanol Fuel Cells: Progress in Cell Performance and Cathode Research,”Electrochemical Acta,47, 3741 (2002).
Umeda, M., Kokubo, M., Mohamedi, M. and Uchida, I., “Porous-microelectrode Study on Pt/C Catalysts for Methanol Electrooxidation,”Electrochim. Acta,48, 1367 (2003).
Viswanathan, R., Hou, G., Liu, R., Bare, S. R., Modica, F., Mickelson, G., Segre, C.U., Leyarovska, N. and, Smotkin, E. S., “In-situ XANES of Carbon-Supported Pt-Ru Anode Electrocatalyst for Reformate-Air Polymer Electrolyte Fuel Cells,”J. Phys. Chem. B,106, 3458 (2002).
Wei, Z., Wang, S., Yi, B., Liu, J., Chen, L., Zhou, W., Li, W. and Xin, Q., “Influence of Electrode Structure on the Performance of a Direct Methanol Fuel Cell,”J. Power Source,106, 364 (2002).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Pak, C., Lee, S.J., Lee, SA. et al. The effect of two-layer cathode on the performance of the direct methanol fuel cell. Korean J. Chem. Eng. 22, 214–218 (2005). https://doi.org/10.1007/BF02701487
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02701487