Abstract
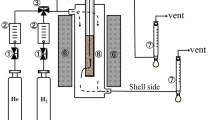
Amorphous silica membranes were deposited by thermal decomposition of tetraethoxysilane at 600–650 ‡C on a porous α-alumina tube with pore size of 110–180 nm or γ-alumina coated α-alumina tube with pore size of 6–8 nm.The forced cross-flow through the porous wall of the support was very effective in plugging macropores. The membranes formed on γ-alumina coated oc-alumina tube showed H2 permeances much higher than the SiO2 membranes formed on the α-alumina tube. This indicated that the γ-alumina film was effective in improving the H2 permeance and H2/N2 selectivity. The permeation tests with CO2, N2, CH4, C3H8 and i-C4H10 showed that a very small number of mesopores remained unplugged by the CVD. Permeation of hydrogen was explained by activated diffusion, and that of the other gases by Knudsen diffusion through the unplugged pores. Thus, the total permeance was composed of permeances due to the activated and Knudsen diffusion mechanisms. The contribution of Knudsen diffusion pores decreased to 0.02 when the γ-alumina film was modified at 650 ‡C until Pfe=50 Pa.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Aoki, K., Yokoyama, S., Kusakabe, K. and Morooka, S.,“Preparation of Supported Palladium Membrane and Separation of Hydrogen”Korean J. Chem. Eng.,13, 530 (1996).
Asaeda, M., Oki, Y. and Manabe, T.,“Preparation of Porous Silica Membranes for Separation of Inorganic Gaseous Mixtures at High Temperatures,” Report on Energy Conversion and Utilization with High Efficiency. Science and Technology for Energy Conversion. Ministry of Education, Science, Sports and Culture, Japan, 253 (1993).
Bakker, W. J. W., Kapteijin, F., Poppe, J. and Moulijn, J. A.,“Permeation Characteristics of a Metal-Supported Silicalite-1 Zeolite Membrane,”J. Membrane Sci.,117, 57 (1996).
Breck, D. W.,“Zeolite Molecular Sieves,” John Wiley, New York, 636 (1974).
Gavalas, G. R., Megiris, C. E. and Nam, S. W.,“Deposition of H2-Permselective SiO2 Films,”Chem. Eng. Sci.,44,1829 (1989).
Ha, H. Y., Nam, S. W., Hong, S.-A. and Lee, W. K.,“Chemical Vapor Deposition of Hydrogen-Permselective Silica Films on Porous Glass Support from Tetraethylorthosilicate,”J. Membrane Sci.,85, 279 (1993).
Hwang, G.-J., Qnuki, K. and Shimizu, S.,“Separation of Hydrogen from a H2-H2O-HI Gaseous Mixture Using a Silica Membrane,”AIChE J.,46, 92 (2000).
Ioannides, T. and Gavalas, G. R.,“Catalytic Isobutane Dehydrogenation in a Dense Silica Membrane Reactor,”J. Membrane Sci.,77, 207 (1993).
Jiang, S., Yan, Y. and Gavalas, G. R.,“Temporary Carbon Barriers in the Preparation of H2-Permselective Silica Membranes,”J. Membrane Sci.,103, 211 (1995).
Jung, K. Y., So, J. H., Park, S. B. and Yang, S. M.,“Hydrogen Separation from the H2/N2 Mixture by Using a Single and Mult Stage Inorganic Membrane,”Korean J. Chem. Eng.,16, 193 (1999).
Kim, S. and Gavalas, G.,“Preparation of H2 Permselective Silica Membranes by Alternating Reactant Vapor Deposition,”Ind. Eng. Chem. Res.,34, 168 (1995).
Kim, S.-S., Choi, H.-K., Park, H.-C., Kim, T.-O. and Sea, B.,“CO2 Separation Using a Surface Modified Silica Membrane,”J. Korean Environmental Sci. Soc.,9, 311 (2000).
Kusakabe, K., Sakamoto, S., Saie, T. and Morooka, S.,“Pore Structure of Silica Membranes Formed by a Sol-Gel Technique using Tetraethoxysilane and Alkyltriethoxysilanes,”Sep. and Pruif. Tech.,16, 139 (1999).
Lange, R. S. A., Keizer, K. and Burggraaf, A. J.,“Analysis and Theory of Gas Transport in Microporous Sol-Gel Derived Ceramic Membranes,”/Membrane Sci.,104, 81 (1995a).
Lange, R. S. A., Keizer, K. and Burggraaf, A. J.,“Aging and Stability Microporous Sol-Gel-Modified Ceramic Membranes,”Ind. Eng. Chem. Res.,34, 3838 (1995b).
Morooka, S., Yan, S., Kusakabe, K. and Akiyama, Y.,“Formation of Hydrogen-Permselective SiO2 Membrane in Macroporous of α-Alumina Support Tube,”J. Membrane Sci.,101, 89 (1995).
Nakao, S., Suzuki, T., Sugawara, T., Tsuru, T. and Kimura, S.,“Preparation of Microporous Membranes by TEOS/O3 CVD in the Opposing Reactant Geometry,”Microporous & Mesoporous Materials,37, 145 (2000).
Noble, R. D. and Stern, S. A.,“Membrane Science and Technology Series 2, Membrane Separations Technology Principles and Applications,” Elsevier Sci. Ltd., Netherlands (1995).
Prabhu, A. K. and Otama, S. T.,“Highly Hydrogen Selective Ceramic Membranes: Application to the Transformation of Greenhouse Gases,”J. Membrane Sci.,176, 233 (2000).
Raman, N. K. and Brinker, C. J.,“Organic Template Approach to Molecular Sieving Silica Membranes,”J. Membrane Sci.,105, 273 (1995).
Sea, B.-K., Kim, S.-S. and Kim, T.-O.,“Pore Size Control of Silica-Coated Alumina Membrane for Gas Separation,”J. Korean Environmental Sci. Soc.,8, 263 (1999).
Shelekhin, A. B., Dixon, A. G. and Ma., Y H.,“Theory of Gas Diffusion and Permeation in Inorganic Molecular-Sieve Membranes,”AIChEJ.,41, 58 (1995).
So, J. H., Yoon, K. Y., Yang, S. M. and Park, S. B.,“Preparations of Metal Impregnated Porous Inorganic Membranes for Hydrogen Separation by Mulit-Step Pore Modifications,”Korean J. Chem. Eng.,16, 180 (1999).
Tsai, C.-Y., Tarn, S.-Y., Lu, Y. and Brinker, C. J.,“Dual-layer Asymmetric Microporous Silica Membranes,”J. Membrane Sci.,169, 255 (2000).
Tsapatsis, M. and Gavalas, G.,“Structure and Aging Characteristics of H2-Permselective SiO2-Vycor Membranes,”J. Membrane Sci.,87, 281 (1994).
Way, J. D. and Roberts, D. L.,“Hollow Fiber Inorganic Membranes for Gas Separations,”Sep. Sci & Tech.,27, 29 (1992).
Wu, J. C. S., Sabol, H., Smith, G. W., Flowers, D. L. and Liu, P. K. T, “Characterization of Hydrogen-Permselective Microporous Ceramic Membranes,”J. Membrane Sci.,96, 275 (1994).
Yoldas, B. E.,“Alumina Sol Preparation from Alkoxides,”Ceramic Bull.,54, 289 (1975).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kin, SS., Sea, BK. Gas permeation characteristics of silica/alumina composite membrane prepared by chemical vapor deposition. Korean J. Chem. Eng. 18, 322–329 (2001). https://doi.org/10.1007/BF02699172
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02699172