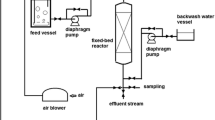
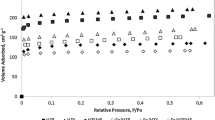
Abstract
NO removal activity and the durability of iron-exchanged mordenite type zeolite catalyst (FeHM) have been examined in a continuous fixed bed flow reactor. The catalytic activity for NO reduction by NH3 in the presence of oxygen was much higher than that in the absence of oxygen, and it was fully reversible with respect to the presence of oxygen in the feed gas stream. The oxidation ability of SCR catalysts including FeHM was critical for both reactions of NH3 and SO2 oxidation, thus for the NO removal activity and its sulfur tolerance. The maximum conversion of NO for FeHM catalyst with respect to the reaction temperature shifted to the higher temperature due to its mild oxidation ability. The deactivation behaviors such as the changes of the physicochemical properties of the catalyst and the loss of NO removal activity induced by SO2 could not be distinguished, regardless of the metals exchanged in zeolite. However, the amount of deactivating agents deposited on the catalyst surface depended on the species of metals exchanged on the mordenite type zeolite, which was mainly attributed to the oxidation ability of metals for SO2 conversion to SO3.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Amiridis, M. D., Puglisi, F., Dumesic, J. A., Millman, W. S. and TopsØe, N.-Y., “Kinetic and Infrared Spectroscopic Studies of Fe-Y Zeolites for the Selective Catalytic Reduction of Nitric Oxide by Ammonia,”J. Catal.,142, 572 (1993).
Bauerle, G. L., Wu, S. C. and Nobe, K., “Parametric and Durability Studies of NOx Reduction with NH3 on Fe-Cr Oxide Catalysts,”Ind. Eng. Chem. Prod. Res. Dev.,17(2), 123 (1978).
Chen, J. P. and Yang, R. T., “Selective Catalytic Reduction of NOx with NH3 on SO42-/TiO2 Superacid Catalyst,”J. Catal.,139, 277 (1993).
Choi, E. Y., Nam, I. S., Kim, Y. G., Chung, J. S., Lee, J. S. and Nomura, M., “An X-ray Absorption Study of Copper Ion Exchanged H-mordenite for Selective Catalytic Reduction of NO by NH3,”J. Mol. Catal.,69, 247 (1991).
Choi, E. Y., Nam, I. S. and Kim, Y. G., “TPD Study of Mordenitetype Zeolites for Selective Catalytic Reduction of NO by NH3,”J. Catal.,161(2), 597(1996).
Chung, C. M., Choi, H., Choo, S. T., Nam, I. S. and Kim, Y. G., “A Kinetic Study for Selective Catalytic Reduction of NO by NH3 over Cu-exchanged Mordenite Type Zeolite Catalyst,”HWAHAK KONGHAK,34, 790 (1996).
Ham, S.W., “Activity and Durability of Copper Ion-exchanged Mordenite for NO Reduction by NH3,” Ph. D. Thesis, Dept. of Chem. Eng., Pohang Univ. of Sci. & Tech. (1995b).
Ham, S.W., Choi, H., Nam, I. S. and Kim, Y. G., “Deactivation of Copper-Ion-Exchanged Hydrogen-Mordenite-Type Zeolite Catalyst by SO2 for NO Reduction by NH3,”Catal. Today,11, 611 (1992).
Ham, S.W., Choi, H., Nam, I. S. and Kim, Y. G., “Effect of Copper Contents on Sulfur Poisoning of Copper Ion-Exchanged Mordenite for NO Reduction by NH3,”Ind. & Eng. Chem. Res.,34, 1616 (1995a).
Ham, S.W., Choi, H., Nam, I. S. and Kim, Y. G., “Effect of Oxygen on Selective Catalytic Reduction of NO by NH3 over Copper Ion Exchanged Mordenite-Type Zeolite Catalyst,”Catal. Lett.,42, 35 (1996).
Iizuka, T. and Lunsford, J. H., “Active Intermediates in the Reduction of Nitric Oxide by Ammonia over a CoY Zeolite,”J. Am. Chem. Soc.,100, 6106 (1978).
Ito, E., Hultermans, R. J., Lugt, P. M., Burgers, M. H. W., Rigutto, M. S., van Bekkum, H. and van den Bleek, C.M., “Selective Catalytic Reduction of NOx with Ammonia over Cerium-Exchanged Mordenite,”Appl. Catal. B:Environmental,4, 95 (1994).
Kiovsky, J. R., Kotadia, P. B. and Lim, C. T., “Evaluation of a New Zeolitic Catalyst for NO Reduction with NH3,”Ind. Eng. Chem. Prod. Res. Dev.,19, 218 (1980).
Markvart, M. and Pour, V., “The Influence of Oxygen on the Catalytic Reduction of Nitric Oxide by Ammonia,”J. Catal.,7, 279 (1967).
Medros, F. G., Eldridge, J.W. and Kittrell, J. R., “Dual-catalytic System to Broaden the Window of Operability in the Reduction of NOx with Ammonia,”Ind. Eng. Chem. Res.,28, 1171 (1989).
Nam, I. S., Eldridge, J.W. and Kittrell, J. R., “Model of Temperature Dependence of a Vanadia-Alumina Catalyst for NO Reduction by NH3: Fresh Catalyst,”Ind. Eng. Chem. Prod. Res. Dev.,25, 186 (1986a).
Nam, I. S., Eldridge, J.W. and Kittrell, J. R., “Deactivation of a Vanadia-Alumina Catalyst for NO Reduction by NH3,”Ind. Eng. Chem. Prod. Res. Dev.,25, 192 (1986b).
Nam, I. S., Hwang, W. C., Ham, S.W. and Kim, Y. G., “Activity and Durability of Natural Zeolite Containing Cupric Ions for NO Reduction by NH3,”Catalytic Science and Technology,1, 165 (1990).
Petunchi, J. O. and Hall, W. K., “Redox Catalysis over Iron Zeolites: Kinetics and Mechanism,”J. Catal.,78, 327 (1982).
Pins, W. L. and Nuninga, Z. L., “Design and Experience with Catalytic Reactors for SCR-DENOX,”Catalysis Today,16, 187 (1993).
Ramis, G., Busca, G., Bregani, F. and Forzatti, P., “Fourier Transform-Infrared Study of the Adsorption and Coadsorption of Nitric Oxide, Nitrogen Dioxide and Ammonia on Vanadia-Titania and Mechanism of Selective Catalytic Reduction,”Appl. Catal.,64, 243 (1992).
Schmidt, R., Amiridis, M. D., Dumesic, J. A., Zelewski, L. M. and Millman, W. S., “In Situ Mössbauer Spectroscopy Studies of Fe-Y Zeolites for the Selective Catalytic Reduction of Nitric Oxide by Ammonia,”J. Phys. Chem.,96, 8142 (1992).
Seiyama, T., Arakawa, T., Matsuda, T., Takita, Y. and Yamazoe, N., “Catalytic Activity of Transition Metal Ion Exchanged Y Zeolites in the Reduction of Nitric Oxide with Ammonia,”J. Catal.,48, 1 (1977).
Takagi, M., Kawai, T., Soma, M., Onishi, T. and Tamaru, K., “Mechanism of Catalytic Reaction between NO and NH3 on V2O5 in the Presence of Oxygen,”J. Phys. Chem.,80(4), 430 (1976).
Williamson, W. B. and Lunsford, J. H., “Nitric Oxide Reduction with Ammonia over Cu(II)Y Zeolites,”J. Phys. Chem.,80(24), 2664 (1976).
Wong, W. C. and Nobe, K., “Reduction of NO with NH3 on Al2O3-and TiO2-Supported Metal Oxide Catalysts,”Ind. Eng. Chem. Prod. Res. Dev.,25, 179(1986).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ham, SW., Nam, IS. & Kim, Y.G. Activity and durability of iron-exchanged mordenite-type zeolite catalyst for the reduction of NO by NH3 . Korean J. Chem. Eng. 17, 318–324 (2000). https://doi.org/10.1007/BF02699047
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02699047