Abstract
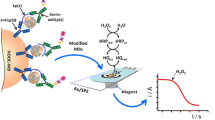
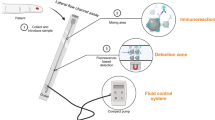
A membrane immuno-chromatographic system that selectively separates plasma lipoproteins and generates a signal in proportion to the concentration of cholesterol (HDL-C) within high-density lipoprotein (HDL) was investigated as a point-of-care device for the prognosis of coronary heart disease. The system consists of three functional membrane strip pads connected in a sequence for: (from the bottom) immuno-separation based on biotinstreptavidin reaction, catalytic conversion of cholesterol to hydrogen peroxide, and production of a signal. For immunochromatography, a monoclonal antibody, specific to apolipoprotein B100 that is present on the surfaces of low-density lipoproteins (LDL) and very low-density lipoproteins (VLDL), with a high binding constant (5xl010 L/mol) was raised and chemically conjugated to streptavidin. The conjugate was first reacted with lipoprotein particles, and this mixture was absorbed by the capillary action into the biotin pad of the system. After being transferred by medium, immunocapture of LDL and VLDL particles onto the biotin pad took place, andin situ generation of a signal in proportion to HDL-C consecutively occurred. The capture was selective as well as effective (minimum 90% of LDL and VLDL in clinical concentration ranges), and the detection limit of HDL-C was far lower than 20 mg/dL. To construct a userfriendly device, we are currently investigating the automation of such processes of reactions and separation by adapting a liquid flow-controlling technology that programs the times for the immune reaction and separation. My group further pursues an interdisciplinary study to develop a micro system employing semiconductor-based technologies that will eventually enable the handling of sub-micro liter volume of body fluid as a specimen.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Allen, M. P., DeLizza, A., Ramel, U., Jeong, H. and Singh, P.,“A Noninstrumented Quantitative Test System and its Application for Determining Cholesterol Concentration in Whole Blood,”Clin. Chem.,36,1591 (1990).
Artiss, J. D., Feldbruegge, D. H., Kroll, M. H., McQueen, M. J., Pry, T., Zak, B. and Ziegenhorn, J., Measurement of Cholesterol Concentration, Methods for Clinical Laboratory Measurement of Lipid and Lipoprotein Risk Factors, N. Rifai, and G. R. Warnick (eds.), AACC Press, Washington, DC (1991).
Bachorik, P. S. and Albers, J. J.,“Precipitation Methods for Quantification of Lipoproteins,”Meth. Enzymol.,129, 78 (1986).
Bachorik, P. S. and Ross, J. W.,“National Cholesterol Education Program Recommendations for Measurement of Low-Density Lipoprotein Cholesterol: Executive Summary,”Clin. Chem.,41, 1414 (1995).
Cantarero, L. A., Butler, J. E. and Osborne, J. W.,“The Adsorptive Characteristics of Proteins for Polystyrene and their Significance in Solid-Phase Immunoassays,”Anal. Biochem.,105, 375 (1980).
Fisher, W. R. and Schumaker, V. N.,“Isolation and Characterization of Apolipoprotein B-100,”Meth. Enzymol.,128, 247 (1986).
Gordon, D. J. and Rifkind, B. M.,“High-Density Lipoprotein ⥭he Clinical Implications of Recent Studies,”New Eng. J. Med.,321, 1311 (1989).
Gordon, T., Castelli, W. P., Hjortland, M. C., Kannel, W. B. and Dawber, T. R.,“High Density Lipoprotein as a Protective Factor Against Coronary Heart Disease,”Amer. J. Med.,62, 707 (1977).
Gotto, J., Antonio, M., Pownall, H. J. and Havel, R. J.,“Introduction to the Plasma Lipoproteins,”Meth. Enzymol.,128, 3 (1986).
Hermanson, G. T., Mallia, A. K. and Smith, P. K.,“Immobilized Affinity Ligand Techniques,” Academic Press, Inc., New York (1992).
Hsu, S.-M. and Soban, E.,“Color Modification of Diaminobenzidine (DAB) Precipitation by Metallic Ions and its Application for Double Immunohistochemistry,”J. Histochem. Cytochem.,30, 1079 (1982).
Johnson, W. J., Mahlberg, F. H., Rothblat, G. H. and Phillips, M. C., “Cholesterol Transport Between Cells and High-Density Lipoproteins,”Biochim. Biophy.Acta,1085, 273 (1991).
Kohler, G. and Milstein, C.,“Continuous Culture of Fused Cells Secreting Antibody of Predefined Specificity,”Nature,256, 495 (1975).
Law, W. T., Doshi, S., McGeehan, J., McGeehan, S., Gibboni, D., Nikolioukine, Y., Keane, R., Zheng, J., Rao, J. and Ertingshausen, G.,“Whole-Blood Test for Total Cholesterol by a Self-Metering, Self-Timing Disposable Device with Built-In Quality Control,”Clin. Chem.,43, 384 (1997).
Mackness, M. I. and Durrington, P. N.,“Lipoprotein Separation and Analysis for Clinical Studies, Lipoprotein Analysis” C. A. Converse, and E. R. Skinner (eds.), A Practical Approach, IRL Press, Oxford (1992).
Matson, R. S. and Little, M. C., “Strategy for the Immobilization of Monoclonal Antibodies on Solid-Phase Supports”J. Chromatogr.,458, 67 (1988).
Nauck, M., Winkler, K., Marz, W. and Wieland, H., “Quantitative Determination of High-, Low-, and Very-Low-Density Lipoproteins and Lipoprotein(a) by Agarose Gel Electrophoresis and Enzymatic Cholesterol Staining”Clin. Chem.,41,1761 (1995).
Nilsson, S., Lager, C., Laurell, T. and Birnbaum, S., “Thin-Layer Immunoaffinity Chromatography with Bar Code Quantitation of C-Reactive Protein”Anal. Chem.,67, 3051 (1995).
Noble, D., “Home Test for Cholesterol”Anal. Chem.,65, 1037A (1933).
Paek, S.-H., Bachas, L. G. and Schramm, W., “Defined Analyte-Enzyme Conjugates as Signal Generators in Immunoassays”Anal. Biochem.,210,145 (1993).
Paek, S. H., Jang, M. R., Mok, R. S., Kim, S. C. and Kim, H. B., “Immunochromatographic Membrane Strip Assay System for a Single-Class Plasma Lipoprotein Cholesterol, Exemplified by High-Density Lipoprotein Cholesterol Measurement”Biotechnol Bioeng.,62, 145 (1999).
Rifai, N., Warnick, G. R., McNamara, J. R., Belcher, J. D., Grinstead, G. F., Frantz, J. and Ivan, D., “Measurement of Low-Density-Lipoprotein Cholesterol in Serum: a Status Report”Clin. Chem.,38,150 (1992).
Rudel, L. L., Lee, J. A., Morris, M. D. and Felts, J. M., “Characterization of Plasma Lipoproteins Separated and Purified by Agarose-Column Chromatography”Biochem. J.,139, 89 (1974).
Schaefer, E. J., “Overview of the Diagnosis and Treatment of Lipid Disorders, Methods for Clinical Laboratory Measurement of Lipid and Lipoprotein Risk Factors” N. Rifai, and G R. Warnick (eds.), AACC Press, Washington, DC (1991).
Schramm, W. and Paek, S.-H.,“Antibody-Antigen Complex Formation with Immobilized Immunoglobulins”Anal. Biochem.,205, 47 (1992).
Skinner, E. R., “The Separation and Analysis of High-density Lipoprotein (HDL) and Low-density Lipoprotein (LDL) Subfractions, Lipoprotein Analysis” C. A. Converse, and E. R. Skinner (eds.), A Practical Approach, IRL Press, Oxford (1992).
Sterling, B., Kiang, T., Subramanian, K., Saltman, M., Smart, W., Tsay, M., Sugarman, J., Patel, D., Monger, D., Martin, D., Gibbons, I. and Voss, F., “Simultaneous Patient-Side Measurement of Hemoglobin, Glucose, and Cholesterol in Finger-Stick Blood”Clin. Chem.,38, 1658 (1992).
Tall, A. R., “Plasma High Density Lipoproteins”J. Clin. Invest.,86, 379 (1990).
Warnick, G. R. and Wood, P. D., “National Cholesterol Education Program Recommendations for Measurement of High-Density Lipoprotein Cholesterol: Executive Summary”Clin. Chem.,41,1427 (1995).
Weber, P. C., Ohlendorf, D. H., Wendoloski, J. J. and Salemme, F. R., “Structural Origins of High-Affinity Biotin Binding to Streptavidin”Science,243, 85 (1989).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Paek, SH., Cho, JH., Kang, MS. et al. On-Site diagnostic device based on immuno-separation of proteins. Korean J. Chem. Eng. 18, 263–269 (2001). https://doi.org/10.1007/BF02698469
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02698469