Abstract
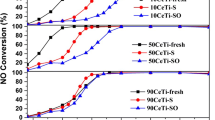
The catalytic activity of sulfated titania (ST) calcined at a variety of temperatures has been investigated for selective catalytic reduction (SCR) of NO by NH3. The NO removal activity of ST catalyst mainly depends on its sulfur content, indicating critical role of sulfur species on the surface of TiO2. The role of sulfur is mainly the formation of acid sites on the catalyst surface. The presence of both BrØnsted and Lewis acid sites on the surface of sulfated titania has been identified by IR study with the adsorption of NH3 and pyridine on ST. The reduction of the intensity of IR bands representing BrØsted acid sites is more pronounced than that revealing Lewis acid sites as the calcination temperature increases. It has been further clarified by IR study of ST500 catalyst evacuated at a variety of temperatures. The NO removal activity also decreases with the increase of the catalyst calcination temperature. It simply reveals that BrØnsted acid sites induced by sulfate on the catalyst surface are primarily responsible for the enhancement of catalytic activity of ST catalyst containing sulfur for NO reduction by NH3.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Refrences
Alemany, L. J., Lietti, L., Ferlazzo, N., Forzatti, P., Busca, G., Giamello, E. and Bregani, F., “Reactivity and Physicochemical Characteriza-tion of V2O5-WO3/TiO2De-NOxCatalyst”,J. Catal,155, 117 (1996).
Amiridis, M. D., Wachs, I. E, Deo, G., Jehng, J. and Kim, D. S., “Reac-tivity of V2O5 Catalyst for the Selective Catalytic Reduction of NO by NH3: Influence of Vanadia Loading, H2O, and SO2,”,J. Catal,161, 247 (1996).
Busca, G., Saussey, H., Saur, O., Lavalley, J. C. and Lorenzelli, V., “FT-IR Characterization of the Surface Acidity of Different Titanium Di-oxide Anatase Preparations”,Appl. Catal.,14, 245 (1985).
Chen, L. P. and Yang, R. T., “Selective Catalytic Reduction of NO with NH3 on SO 2-4 /TiO2 Superacid Catalyst”,J. Catal,139, 277 (1993).
Choi, E. Y, Nam, I. S. and Kim, Y G., “TPD Study of Mordenite-Type Zeolites for Selective Catalytic Reduction of NO by NH3”,J. Catal,161, 597 (1996).
Choo, S. T., Lee, Y G., Nam, I. S., Ham, S. W. and Lee, J. B., “Charac-teristics of V2O5 Supported on Sulfated TiO2 for Selective Catalytic Reduction of NO by NH3”,Appl. Catal. A: General,200, 177 (2000a).
Choo, S. T., Koh, J. Y, Nam, I. S., Ham, S. W. and Lee, J. B., “Charac-teristics of V2O5 Catalyst Supported on TiO2 Prepared from Metati-tanic Acid for Selective Catalytic Reduction of NO by NH3”,HWA-HAKKONGHAK,38, 597 (2000b).
Ebitani, K., Tanaka, T. and Hattori, H., “X-ray Absorption Spectroscopic Study of Platinum Supported on Sulfate Ion-treated Zirconium Ox-ide”,Appl. Catal. A: General,102(2), 102 (1993).
Ferwerda, R, Mass, J. H. and Duijneveldt, F B., “Pyridine Adsorption onto Metal Oxides: An ab initio Study of Model Systems”,J. Mol. Catal. A: General,104, 319 (1996).
Ham, S. W., Nam, I. S. and Kim, Y G., “Activity and Durability of Iron-exchanged Mordenite-type Zeolite Catalyst for the Reduction of NO by NH3”Korean J. Chem. Eng.,17, 318 (2000).
Ham, S. W. and Nam, I. S., “Selective Catalytic Reduction of Nitrogen Oxides by Ammonia”, Royal Society of Chemistry,Catalysis,16, 236 (2002).
Hino, M. and Arata, K., “Synthesis of Solid Superacid Catalyst with Acid Strength of H0<-16.05”,J. Chem. Soc, Chem. Commun., 851 41, (1980).
Jung, S. M. and Grange, P., “Characterization and Reactivity of Pure TiO2-SO 2-4 SCR Catalyst: Influence of SO 2-4 Content”,Catal. Today,59, 305 (2000a).
Jung, S. M. and Grange, P., “The Investigation of Mechanism of SCR Reaction on a TiO2-SO 2-4 Catalyst by DRIFTS”,Appl. Catal. B: Envi-ronmental,27, L11 (2000b).
Kustov, L. M., Kazansky, V. B., Figueras, F. and Tichit, D., “Investiga-tion of the Acidic Properties of ZrO2 Modified by SO 2-4 Anions”,J. Catal,150, 143 (1994).
Navarrete, J., Lopez, T. and Gomez, R, “Surface Acidity of Sulfated TiO2-SiO2 Sol-Gels”,Langmuir,12, 4385 (1996).
Nortier, P., Fourre, P., Mohamed Saad, A. B., Saur, O. and Lavalley, J. C., “Effects of Crystallinity and Morphology on the Surface Properties of Alumina”,Appl. Catal,61, 141 (1990).
Orsenigo, C., Lietti, L., Tronconi, E., Forzatti, P. and Bregani, F., “Dy-namic Investigation of the Role of Surface Sulfates in NO Reduc-tion and SO2 Oxidation over V2O5-WO3/TiO2 Catalyst”,Ind. Eng. Chem. Res.,37, 2350 (1998).
Pasel, J., Speer, V, Albrecht, G, Richter, F. and Papp, H., “Metal Doped Sulfated ZrO2 as Catalyst for the Selective Catalytic Reduction (SCR) of NO with Propane”,Appl. Catal. B: Environmental,25, 105 (2000).
Primet, M., Pichat, P. and Mathieu, M., “Infrared Study of the Surface of Titanium Dioxides. I. Hydroxyl Groups”,J. Phy. Chem.,75(9), 1216(1971).
Sayari, A. and Dicko, A, “The State of Platinum in Pt on Sulfated Zir-conia Superacid Catalysts”,J. Catal,145, 561 (1994).
Song, X. and Sayari, A., “Sulfated Zirconia-Based Strong Solid-Acid Catalyst: Recent Progress”,Catal. Rev. Sci. Eng.,38(3), 329 (1996).
Tanabe, K., “Solid Acids and Bases Their Catalytic Properties^' Kodan-sha, Tokyo (1970).
Ward, J. W., “Infrared Studies of Zeolite Surfaces and Surface Reac-tions”, Zeolite Chemistry and Catalysis, Rabo, J. A., eds., American Chemical Society, Washington D.C. (1976).
Yamaguchi, T, “Recent Progress in Solid Superacid;Appl. Catal.,61(1), 1 (1990).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Choo, S.T., Nam, IS., Ham, SW. et al. Effect of calcination temperature on the characteristics of SOsk4/2-/TiO2 catalysts for the reduction of NO by NH3 . Korean J. Chem. Eng. 20, 273–278 (2003). https://doi.org/10.1007/BF02697240
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02697240