Abstract
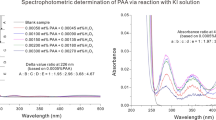
A micro-determination of peroxide value is proposed. The iodine liberated from potassium iodide-silica gel reagent with peroxides in the oil was complexed with starch in 0.01 N hydrochloric acid. The blue color was then measured colorimetrically at 560 nm.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Lea, C.H., Proc. Soc. London Ser. B. 108:175 (1931).
Eskin, N.A.M., and C. Frankel, JAOCS 53:746 (1976).
Takagi, T., Y. Mituno, and M. Masumura, Lipids 13:147 (1978).
Asakawa, T., and S. Matsushita, Eiyo To Shokuryo (Tokyo) 28: 403 (1975).
Asakawa, T., and S. Matsushita, Eiyo To Shikuryo (Tokyo) 29:408 (1976).
Asakawa, T., and S. Matsushita, Bull. Res. Inst. Food Sci., Kyoto Univ. 40:12 (1977).
The standard method of Japan Oil Chemists’ Society, 2.4.12–71.
Author information
Authors and Affiliations
About this article
Cite this article
Asakawa, T., Matsushita, S. Colorimetric determination of peroxide value with potassium iodide-silica gel reagent. J Am Oil Chem Soc 55, 619–620 (1978). https://doi.org/10.1007/BF02678958
Issue Date:
DOI: https://doi.org/10.1007/BF02678958