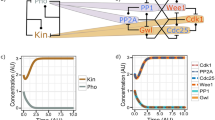
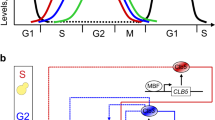
Summary
DNA replication and mitosis are dependent on the activity of cyclin-dependent protein kinase(CDK) enzymes, which are heterodimers of a catalytic subunit with a cyclin subunit. Cyclinbinding to specific individual proteins is thought to provide potential substrates to Cdk. Protein binding by cyclins is assessed in terms of its mechanism and biological significance, using evidence from diverse organisms including, substrate specificity in animal Cdk enzymes containing D-,A-, and B-type cyclins and extensive cyclin gene manipulations in yeasts. Assembly of protein complexes with cyclin/Cdk is noted and the capacity of the cyclin-dependent kinase subunit Cks, in such complex, to extend the range of Cdk substrates is documented and discussed in terms of cell cycle regulation. Cell cycle progression involves changing abundance of individual cyclins, due to changing rates of their transcription or proteolysis, with consequent changes in the substrates of CDK through the cell cycle. Some overlap of the functions of individual cyclins in vivo has been identified byn cyclin deletions and is suggested to follow a pattern in which cyclins well enough to preserve viability as groups of cyclins are removed by proteolysis. Cyclin accumulation is particularly important in terminating the G1 phase , when it raises CDK activity and starts events leading to DNA replication. It is suggested that plants share this mechanism. The distribution of cyclins and Cdk in maize root tip cells during mitosis and cytokinesis indicates the presence of Cdk1 (Cdc2a) and cyclin CycBlzm;2 at the mature and disassembling preprophase band and the presence of CycBlzm;2 at condensing and condensed chromosomes. Both observations correlate with the earlier-reported capacity of injected metaphase cyclin/CDK to accelerate preprophase band disassembly and chromosomes condensation and with observations correlate with the earlier-reported capacity of injected metaphase cyclin/CDK to accelerate preprophase band disassembly and chromosome condensation and with observation of the location of Cdk and cyclins in other laboratories. Additionally CycBlzm;2 is seen at the nuclear envelope during its breakdown, which correlates with an acceleration of the process by injected metaphase cyclin B/CDK. A phenomenon possibly unique to the plant kingdom is the persistence of mitotic cyclins after anaphase. Participation of cyclins in cytokinesis is indicated by the concentration of the mitotic cyclin CycA1;zm;1 at the phragmoplast. It is suggested that cyclins have a genral function of spatially focusing Cdk activity and that in the plant cell the concentrations cyclins are important mediators of CDK activity at the cytoskeleton, chromosomes, spindle, nuclear envelope, and phragmoplast.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Adams PD, Sellers WR, Sharma SK, Wu AD, Nalin CM, Kaelin WG Jr (1996) Identification of a cyclin-cdk2 recognition motif present in substrates and p21-like cyclin-dependent kinase inhibitors. Mol Cell Biol 16:6623–6633
— Li X, Sellers WR, Baker KB, Leng X, Harper JW, Taya Y, Kaelin WG Jr (1999) Retinoblastoma protein contains a C-terminal motif that targets it for phosphorylation by cyclin-cdk complexes. Mol Cell Biol 19:1068–1080
Aerne BL, Johnson AL,Toyn JH, Johnston LH (1998) Swi5 controls a novel wave of cyclin synthesis in late mitosis. Mol Biol Cell 9: 945–956
Amon A, Tyers M, Futcher B, Nasmyth K (1993) Mechanisms that help the yeast cell cycle clock tick: G2 cyclins transriptionally activate G2 cyclins and repress Gl cyclins. Cell 74: 993–1007
— Irniger S, Nasmyth K (1994) Closing the cell cycle circle in yeast: G2 cyclin proteolysis initiated at mitosis persists until the activa-tion of Gl cyclins in the next cycle. Cell 77:1037–1050
Asada T, Kuriyama R, Shibaoka H (1996) TKRP125, a kinesin-related protein involved in the centrosome-independent organization of the cytokinetic apparatus in tobacco BY-2 cells. J Cell Sei 110:179–190
Bähler J, Steever AB, Wheatley S, Wang Yl, Pringle JR, Gould KL, McCollum D (1998) Role of polo kinase and Midlp in determin-ing the site of cell division in fission yeast. J Cell Biol 143: 1603–1616
Bazan JF (1996) Helical fold prediction for the cyclin box. Proteins Struct Funct Genet 24:1–17
Beach D, Durkacz B, Nurse P (1982) Functionally homologous cell cycle control genes in budding and fission yeast. Nature 300: 706–709
Bögre L, Calderini O, Binarova P, Mattauch M, Till S, Kiegerl S, Jonak C, Pollaschek C, Barker P, Huskisson NS, Hirt H, Heberle-Bors E (1999) A MAP kinase is activated late in plant mitosis and becomes localized to the plane of cell division. Plant Cell 11: 101–113
Brizuela L, Draetta G, Beach D (1987) pl3sucl acts in the fission yeast cell cycle as a component of the p34cdc2 protein kinase. EMBO J 6: 3507–3514
Brown NR, Noble MEM, Lawrie A, Morris M, Tunnah P, Divita G, Johnson LN, Endicott JA (1999) Effects of phosphorylation of threonine 160 on cyclin-dependent kinase-2 structure and activ-ity. J Biol Chem 274: 8746–8756
Brown RC, Lemmon BE (1997) The quadripolar microtubule system in lower land plants. J Plant Res 110: 93–106
Clarke DJ, Segal M, Mondesert G, Reed SI (1999) The Pdsl anaphase inhibitor and Mecl kinase define distinct checkpoints coupling S phase with mitosis in budding yeast. Curr Biol 9: 365–368
Cleary AL, Smith LG (1998) The Tangledl gene is required for spatial control of cytoskeletal arrays associated with cell division during maize leaf development. Plant Cell 10:1875–1888
Colasanti J, Tyers M, Sundaresan V (1991) Isolation and characterisation of cDNA clones encoding a functional p34scdc2 homologue fromTea mays. Proc Natl Acad Sci USA 88: 3377–3381
— Cho S-O, Wick S, Sundaresan V (1993) Localisation of the func-tional p34cdc2 homologue of maize in root tip and stomatal complex cells: association with predicted division sites. Plant Cell 5: 1101–1111
Correa-Bordes J, Nurse P (1995) p25ruml orders S-phase and mitosis by acting as an inhibitor of the p34cdc2 mitotic kinase. Cell 83: 1001–1009
Cross FR, Blake CM (1993) The yeast Cln3 protein is an unstable activator of Cdc28. Mol Cell Biol 13: 3266–3271
Dahl M, Meskiene I, Bögre L, Ha DTC, Swoboda I, Hubman R, Hirt H, Heberle-Bors E (1995) The D-type alfalfa gene cycMs4 com-plements Glcyclin-deficient yeast and is induced in the Gl phase of the cell cycle. Plant Cell 7:1847–1857
Day IS, Reddy AS, Golovkin M (1996) Isolation of a new mitotic-like cyclin from Arabidopsis: complementation of a yeast cyclin mutant with a plant cyclin. Plant Mol Biol 30: 565–575
Deshaies RJ (1995) The self destructive personality of a cell cycle in transition. Curr Opin Cell Biol 7: 781–789
De Veylder L, Engler JD, Burssens S, Manevski A, Lescure B, Van Montagu M, Engler G, Inzé D (1999) A new D-type cyclin ofArabidopsis thaliana expressed during lateral root primordia formation. Planta 208: 453–462
Doonan J, Fobert P (1997) Conserved and novel regulators of the plant cell cycle. Curr Opin Cell Biol 9: 824–830
Dowdy SF, Hinds PW, Louie K, Reed SI, Arnold A, Weinberg RA (1993) Physical interaction of the retinoblastoma protein with human D cyclins. Cell 73: 499–511
Draetta G, Beach D (1988) Activation of cdc2 protein kinase during mitosis in human cells: cell cycle-dependent phosphorylation and subunit rearrangement. Cell 54: 17–26
Dudits D, Magyar Z, Deak M, Meszaros T, Miscolczi P, Feher A, Brown S, Kondorosi E, Athanasiadis A, Pongor S, Bako L, Koncz C, Gyorgyey J (1998) Cyclin dependent and calcium dependent kinase families: response of cell division cycle to hormone and stress signals. In: Francis D, Dudits D, Inzé D (eds) Plant cell division. Portland, Colchester, pp 21–45
Dynlacht BD, Flores O, Lees JA, Harlow E (1994) Differential regulation of E2F transactivation by cyclin/cdk2 complexes. Genes Dev 8:1772–1786
Enoch T, Matthias P, Nurse P, Nigg EA (1991) p34cdc2 acts as a lamin kinase in fission yeast. J Cell Biol 112: 797–807
Epstein CB, Cross FR (1995) CLB5 a novel B cyclin from buding yeast with a role in S phase. Genes Dev 6:1695–1706
Evans T, Rosenthal ET, Youngblom J, Distel D, Hunt T (1983) Cyclin: a protein specified by maternal mRNA in sea urchin eggs that is destroyed at each cleavage division. Cell 33: 386–389
Ferreira PCG, Hemerly AS, Vilarroel R, Van Montagu M, Inzé D (1991) The Arabidopsis functional homolog of the p34cdc2 protein kinase. Plant Cell 3: 531–540
— Engler JD, van Montagu M, Engler G, Inze D (1994) Develop-mental expression of the Arabidopsis cyclin genecycl At. Plant Cell 6:1763–1774
Fisher D, Nurse P (1996) A single fission yeast mitotic cyclin B p34cdc2 kinase promotes both S-phase and mitosis in the absence of Gl cyclins. EMBO J 15: 850–860
Fitch I, Dahmann C, Surana U, Amon A, Nasmyth K, Goetsch L, Byers B, Futcher B (1992) Characterization of four B-type cyclin genes of the budding yeastSaccharomyces cerevisiae. Mol Biol Cell 1992 3: 805–818
Fobert PR, Coen ES, Murphy GJP, Doonan JH (1994) Patterns of cell division revealed by transcriptional regulation of genes during the cell cycle in plants. EMBO J 13: 616–624
Forsburg SL, Nurse P (1994) Analysis ofSchizosaccharomyces pombe cyclin pucl: evidence for a role in cell cycle exit. J Cell Sci 107: 601–613
Funabiki H, Murray AW (2000) TheXenopus chromokinesin Xkid is essential for metaphase chromosome alignment and must be degraded to allow anaphase chromosome movement. Cell 102: 411–424
Gallant P, Nigg E A (1994) Identification of a novel vertebrate cyclin: cyclin B3 shares properties with both A-and B-type cyclins. EMBO J 13: 595–605
Gaudin V, Lunness PA, Fobert PR, Towers M, Riou-Khamlichi C, Murray JA, Coen E, Doonan JH (2000) The expression of D-cyclin genes defines distinct developmental zones in snapdragon apical meristems and is locally regulated by the Cycloidea gene. Plant Physiol 122: 1137–1148
Geng Y, Whoriskey W, Park MY, Bronson RT, Medema RH, Li T, Weinberg RA, Sicinski P (1999) Rescue of cyclin Dl deficiency by knockin cyclin E. Cell 97: 767–777
Genschik P, Criqui MC, Parmentier Y, Derevier A, Fleck J (1998) Cell cycle-dependent proteolysis in plants: identification of the destruction box pathway and metaphase arrest produced by the protease inhibitor MG132. Plant Cell 10:2063–2075
Glotzer M, Murray AW, Kirschner MW (1991) Cyclin is degraded by the ubiquitin pathway. Nature 349:132–138
Gorst JR, Sek FJ, John PCL (1991) Level of p34cdc2-like protein in dividing, differentiating and dedifferentiating cells of carrot. Planta 185: 304–310
Gunning BES (1982) The cytoskeletal apparatus: its development and spatial regulation. In: Lloyd CW (ed) The cytoskeleton in plant growth and development. Academic Press, New York, pp 230–288
Hadwiger JA, Wittenberg C, Richardson HE, de Barros Lopes M, Reed SI (1989) A family of cyclin homologues that control the Gl phase in yeast. Proc Natl Acad Sci USA 86: 6255–6259
Hagting A, Jackman M, Simpson K, Pines J (1999) Translocation of cyclin Bl to the nucleus at prophase requires a phosphorylation-dependent nuclear import signal. Curr Biol 9: 680–689
Harris SD, Kraus PR (1998) Regulation of septum formation inAspergillus nidulans by a DNA damage checkpoint pathway. Genetics 148: 1055–1067
Hasezawa S, Nagata T (1992) Okadaic acid as a probe to analyse the cell cycle progression in plant cells. Bot Acta 105: 63–69
Hata S, Kouchi H, Suzuka I, Ishii T (1991) Isolation and charac-terisation of cDNA clones for plant cyclins EMBO J 10: 2681–2688
Hayles J, Fisher D, Woolard A, Nurse P (1994) Temporal order of S phase and mitosis in fission yeast is determined by the state of the p34cdc2-mitotic B cyclin complex. Cell 78: 813–822
Heese M, Mayer U, Jurgens G (1998) Cytokinesis in flowering plants: cellular processes and developmental integration. Curr Opin Plant Biol 1:486–491
Hemerly A, Bergounioux C, Van Montagu M, Inze D, Ferreira P (1992) Genes regulating the plant cell cycle: isolation of a mitotic-like cyclin fromArabidopsis thaliana. Proc Natl Acad Sci USA 89: 3295–3299
— Engler Jde A, Bergounioux C, Van Montagu M, Engler G, Inzé D, Ferriera P (1995) Dominant negative mutants of Cdc2 kinase uncouple cell division from iterative plant development. EMBO J 14:3925–3936
Hepler PK, Sek FJ, John PCL (1994) Nuclear concentration and mitotic dispersion of the essential cell cycle protein, pl3sucl, examined in living cells. Proc Natl Acad Sci USA 91: 2176–2180
Hirt H, Pay A, Gyorgyey J, Bako L, Newmeth K, Borge L, Schweyen R, Heberle-Bors E, Dudits D (1991) Complementation of a yeast cell cycle mutant by an alfalfa cDNA encoding a protein kinase homologous to p34cdc2v. Proc Natl Acad Sci USA 88:1636–1640
Hoffmann I, Draetta G, Karsenti E (1994) Activation of the phos-phatase activity of human cdc25A by a cdk2-cyclin E dependent phosphorylation at the Gl/S transition. EMBO J 13: 4302–4310
Hoyt MA (1997) Eliminating all obstacles: regulated proteolysis in the eukaryotic cell cycle. Cell 91:149–151
Huang KN, Odinsky SA, Cross FR (1997) Structure-function analy-sis of theSaccharomyces cerevisiae Gl cyclin Cln2. Mol Cell Biol 17:4654–4666
Huntley RP, Murray JAH (1999) The plant cell cycle. Curr Opin Plant Biol 2: 440–446
— Healy S, Freeman D, Lavender P, de Jager S, Greenwood J, Makker J, Walker E, Jackman M, Xie Q, Bannister AJ, Kouzarides T, Gutierrez C, Doonan JH, Murray JAH (1998) The maize retinoblastoma protein homologue ZmRb-1 is regulated during leaf development and displays conserved interactions with Gl/S regulators and plant cyclin D (CycD) proteins. Plant Mol Biol 37: 155–169
Hush JM, Wu L, John PCL, Hepler LH, Hepler PK (1996) Plant mitosis promoting factor disassembles the microtubule pre-prophase band and accelerates prophase progression inTrades-cantia. Cell Biol Int 20: 275–287
Ito M, Iwase M, Kodama H, Lavisse P, Komamine A, Nishihama R, Machida Y, Watanabe A (1998) A novel cis-acting element in pro-moters of plant B-type cyclin genes activates M phase-specific transcription. Plant Cell 10: 331–341
Jackman M, Firth M, Pines J (1995) Human cyclins B1 and B2 are localised to strikingly different structures: Bl to microtubules, B2 primarily to Golgi apparatus. EMBO J 14: 1646–1654
Jacobs T (1995) Cell cycle control. Annu Rev Plant Physiol Plant Mol Biol 46: 317–339
Jeffrey PD, Russo AA, Polyak K, Gibbs E, Hurwitz J, Massague J, Pavletich NP (1995) Mechanism of CDK activation revealed by the structure of a cyclin A-CDK2 complex. Nature 376: 313–320
John PCL (1996) The plant cell cycle: conserved and unique features in mitotic control. In: Meijer L, Guidet S, Vogel L (eds) Progress in cell cycle research, vol 2. Plenum, New York, pp 59–72
— Sek FJ, Lee MG (1989) A homologue of the cell cycle control protein p34cdc2v participates in the cell division cycle ofChlamy-domonas and a similar protein is detectable in higher plants and remote taxa. Plant Cell 1:1185–1193
—— Carmichael JP, McCurdy DW (1990) p34cdc2v homologue level, cell division, phytohormone responsiveness and cell differentia-tion in wheat leaves. J Cell Sci 97: 627–630
— Hayles J (1991) Association of the plant p34cdc2 v-like protein with pl3sucl: implications for control of cell division cycles in plants. Protoplasma 161: 70–74
Katsuta J, Shibaoka H (1992) Inhibition by kinase inhibitors of the development and disappearance of the preprophase band of microtubules in tobacco BY-2 cells. J Cell Sci 103: 397–405
Kim HY, Cho Y (1997) Structural similarity between the pocket region of retinoblastoma tumour suppressor and the cyclin-box. Nat Struct Biol 4: 390–3955
King RW, Peters JM, Tugendreich S, Rolfe M, Bieter P, Kirschner MW (1995) A 20s complex containing CDC27 and CDC16 catalyzes the mitosis-specific conjugation of ubiquitin to cyclin B. Cell 81: 279–288
Kobayashi H, Stewart E, Poon R, Adamczewski JP, Gannon J, Hunt T (1992) Identification of the domains in cyclin A required for binding to, and activation of, p34cdc2v and p32cdk2 protein kinase sub-units. Mol Biol Cell 3:1279–1294
Koch C, Moll T, Neuberg M, Ahorn H, Nasmyth K (1993) A role for the transcription factors Mbpl and Swi4 in progression from Gl to S phase. Science 261:1551–1557
Kouchi H. Sekine M, Háta S (1995) Distinct classes of mitotic cyclins are differentially expressed in the soybean shoot apex during the cell cycle. Plant Cell 7:1143–1155
Landrieu I, Odaert B, Wieruszeski JM, Drobecq H, Rousselot-Pailley P, Inze D, Lippens G (2000) pl3sucl and the WW domain of PIN1 bind to the same phosphothreonine-proline epitope. J Biol Chem 276: 1434–1438
Lew DJ, Reed SI (1993) Morphogenesis in the yeast cell cycle: reg-ulation by Cdc28 and cyclins. J Cell Biol 120:1305–1320
— Dulic V, Reed SI (1991) Isolation of three novel human cyclins by rescue of Gl cyclin (Cln) function in yeast. Cell 66: 1197–1206
— Marini NJ, Reed SI (1992) Different Gl cyclins control the timing of cell cycle commitment in mother and daughter cells of the budding yeastS. cerevisiae. Cell 69: 317–327
— Weinert T, Pringle JR (1997) Cell cycle control inSaccharomyces cerevisiae. In: Pringle JR, Broach JR, Jones EW (eds) The molec-ular and cellular biology of the yeastSaccharomyces. Cold Spring Harbor Laboratory Press, Plainview, NY, pp 607–695
Li LJ, Naeve GS, Lee AS (1993) Temporal regulation of cyclin A-plO7 and p33cdk2 complexes binding to a human thymidine kinase promoter element important for Gl-S phase transcriptional regulation. Proc Natl Acad Sci USA 90: 3554–3558
Li J, Meyer AN, Donoghue DJ (1997) Nuclear localization of cyclin Bl mediates its biological activity and is regulated by phosphorylation. Proc Natl Acad Sci USA 94: 502–507
Liu B, Cyr RJ, Palevitz BA (1996) A kinesin-like protein, KatAp, in the cells ofArabidopsis and other plants. Plant Cell 8:119–132
Lokhorst GM, Sluiman HJ, Star W (1988) The ultrastructure of mitosis and cytokinesis in the sarcinoidChlorokybus atmosphyti-cus (Chlorophyta Charophyceae) revealed by rapid freeze fixation and freeze substitution. J Phycol 24: 237–248
Lopez-Girona A, Furnari B, Mondesert O, Russell P (1999) Nuclear localisation of Cdc25 regulated by DNA damage and 14-3-3 protein. Nature 397: 172–175
Lu PJ, Zhou XZ, Shen M, Lu KP (1999) Function of WW domains as phosphoserine-or phosphothreonine-binding modules. Science 283: 1325–1328
Luciani MG, Hutchins JRA, Zheleva D, Hupp TR (2000) The C-terminal regulatory domain of p53 contains a functional docking site for cyclin A. J Mol Biol 300: 503–518
Magyar Z, Bako L, Bögre L, Dedeoglu D, Dudits D (1993) Activecdc2 genes and cell cycle phase-specific cdc2-related kinase com-plexes in hormone-stimulated alfalfa cells. Plant J 4: 151–161
-Mészáros T, Miskolczi P, Deák M, Fehér A, Brown S, Kondorosi E, Athanasiadis A, Pongor S, Bilgin M, Bakó S, Koncz C, Dudits D (1997) Cell cycle phase specificity of putative cyclin-dependent kinase variants in synchronized alfalfa cells. Plant Cell 9:223–235
Mailand N, Falck J, Lukas C, Syljuasen RG, Welcker M, Bartek J, Lukas J (2000) Rapid destruction of human Cdc25A in response to DNA damage. Science 288:1425–1429
Mariconti L, Albani D, Pitto L, Moroni C, Helin K, Cella R (2000) Molecular characterisation of a plant E2F-like transcription factor. Plant Mol Biol Rep 18: 20
Martm-Castellanos C, Blanco MA, de Prada JM, Moreno S (2000) The pucl cyclin regulates the Gl phase of the fission yeast cell cycle in response to cell size. Mol Biol Cell 11: 543–554
Matsushime H, Roussel MF, Ashmun RA, Sherr CJ (1991) Colony-stimulating factor 1 regulates novel cyclins during the Gl phase of the cell cycle. Cell 65: 701–713
Melan MA, Sluder G (1992) Redistribution and differential extrac-tion of soluble proteins in permeabilized cultured cells. J Cell Sci 101:731–734
Meskiene I, Bögre L, Dahl M, Pirck M, Ha DTC, Swoboda I, Heberle-Bors E, Ammerer G, Hirt H (1995) cycMs3, a novel B-type cyclin gene, is induced in the G0-to-Gl transition of the cell cycle. Plant Cell 5: 759–771
Mews M, Sek FJ, Moore R, Volkmann D, Gunning BES, John PCL (1997) Mitotic cyclin distribution during maize cell division: impli-cations for the sequence diversity and function of cyclins in plants. Protoplasma 200: 128–145
———Volkmann D, John PCL (2000) Immunodetection of four mitotic cyclins and the Cdc2a protein kinase in the maize root: their distribution in cell development and dedifferentiation. Protoplasma 212: 236–249
Michealis C, Ciosk R, Nasmyth K (1997) Cohesins: chromosomal proteins that prevent premature separation of sister chromatids. Cell 91: 35–45
Millar JBA, McGowan CH, Lenaers G, Jones R, Russell P (1991) p80cdc25 inducer is the tyrosine phosphatase that activates p34cdc2v kinase in fission yeast. EMBO J 10: 4301–4309
Mineyuki Y,Yamashita M,Nagahama Y (1991) p34cdc2 kinase homologue in the preprophase band. Protoplasma 162:182–186
— Aioi H, Yamashita M, Nagahama Y (1996) A comparative study on stainability of preprophase bands by the PSTA1R antibody. J Plant Res 109: 185–192
Mironov V, De Veylder L, Van Montagu M, Inzé D (1999) Cyclin-dependent kinases and cell division in plants: the nexus. Plant Cell 11:509–522
Moreno S, Hayles J, Nurse P (1989) Regulation of p34cdc2 protein kinase during mitosis. Cell 58: 361–372
— Nurse P (1994) Regulation of progression through the Gl phase of the cell cycle by theruml gene. Nature 367: 236–242
Morgan DO (1995) Principles of CDK regulation. Nature 374: 131–134
Murray AW, Solomon MJ, Kirschner MW (1989) The role of cyclin synthesis and degradation in the control of maturation promoting factor activity. Nature 339: 275–280
Murray JAH, Freeman D, Greenwood J, Huntley R, Makkerh J, Riou-Khamlichi C, Sorrell DA, Cockroft C, Caimichael JP, Soni R, Shah ZH (1998) Plant D cyclins and retinoblastoma protein homologues. In: Francis D, Dudits D, Inzé D (eds) Plant cell divi-sion. Portland, Colchester, pp 99–127
Nash R, Tokiwa G, Anand S, Erickson K, Futcher AB (1988) The WHI1+ gene ofSaccharomyces cerevisiae tethers cell division to cell size and is a cyclin homolog. EMBO J 7: 4335–4346
Nasmyth K (1996) At the heart of the budding yeast cell cycle. Trends Genet 12:405–412
Nebenführ A, Frohlick JA, Staehelin A (2000) Redistribution of Golgi stacks and other organelles during mitosis and cytokinesis in plant cells. Plant Physiol 124:135–151
Nigg EA (1991) The substrates of the cdc2 kinase. Semin Cell Biol 2:261–270
— (1995) Cyclin-dependent protein kinases; key regulators of the eukaryotic cell cycle. Bioessays 17: 471–480
Nurse P (1990) Universal control mechanism regulating onset of M-phase. Nature 344: 503–508
— (1997) Checkpoint pathways come of age. Cell 91: 865–867
— Bissett Y (1981) Gene required in Gl for commitment to cell cycle and in G2 for control of mitosis in fission yeast. Nature 292: 558–560
Ookata K, Hisanaga SI, Okumura E, Kishimoto T (1993) Associa-tion of p34cdc2 v/cyclin B complex with microtubules in starfish. J Cell Sci 105: 873–881
—— Bulinski JC, Murofushi H, Aizawa H, Itoh TJ, Hotani H, Okumura E, Tachibana K, Kishimoto T (1995) Cyclin B interac-tion with microtubule-associated protein 4 (MAP4) targets p34cdc2 kinase to microtubules and is a potential regulator of M-phase microtubule dynamics. J Cell Biol 128: 849–862
Osborn M, Weber K (1982) Immunofluorescence and immuno-cytochemical procedures with affinity purified antibodies: tubulin containing structures. Methods Cell Biol 24: 98–132
Pagano M, Pepperkok R, Verde F, Ansgorge W, Draetta G (1992) Cyclin A is required at two points in the human cell cycle. EMBO J 11: 961–972
— Tarn SW, Theodoras AM, Beer-Romero P, Del Sal G, Chau V, Yew PR, Dreatta GF, Rolfe M (1995) Role of the ubiquitin-proteosome pathway in regulating abundance of the cyclin-dependent kinase inhibitor p27. Science 269: 682–685
Pan ZQ, Amin A, Hurwitz J (1993) Characterization of the in vitro reconstituted cyclin A or Bl-dependent cdk2 and cdc2 kinase activities. J Biol Chem 268: 20443–20451
Parmentier Y, Criqui MC, Derevier A, Shen W-H, Genschik P (2000) Cell cycle-dependent proteolysis and ectopic expression of cyclin Bl in tobacco BY2 cells. Plant Mol Biol Rep Suppl 18: S01-S64
Patra D, Dunphy WG (1998) Xe-p9, a Xenopus Sucl/Cks protein, is essential for the Cdc2-dependent phosphorylation of the anaphase-promoting complex at mitosis. Genes Dev 12: 2549–2559
— Wang. SX, Kumagai A, Dunphy WG (1999) The Xenopus Sucl/Cks protein promotes the phosphorylation of G2/M regulators. J Biol Chem 274: 36839–36842
Peeper DS, Parker LL, Ewen ME, Toebes M, Hall FL, Xu M, Zantema A, van der Eb AJ, Piwnica-Worms H (1993) A-and B-type cyclins differentially modulate substrate specificity of cyclin-cdk complexes. EMBO J 12: 1947–1954
Pines J, Hunter T (1991) Human cyclins A and Bl are differentially located in the cell and undergo cell cycle-dependent nuclear trans-port. J Cell Biol 115:1–17
———(1994) The differential localisation of human cyclins A and B is due to a cytoplasmic retention signal in cyclin B. EMBO J 13: 3772–3781
Qin LX, Perennes C, Richard L, Bouvier-Durand M, Tréhin C, Inze D, Bergounioux C (1996) G2-and early-M-specific expression of the NTCYC1 cyclin gene inNicotiana tabacum cells. Plant Mol Biol 32:1093–1101
Reichheld J-P, Chaubet N, Shen WH, Renaudin J-P, Gigot C (1996) Multiple A-type cyclins express sequentially during the cell cycle inNicotiana tabacum BY2 cells. Proc Natl Acad Sci USA 93: 13819–13824
Renaudin JP, Colasanti J, Rime H, Yuan ZA, Sundaresan V (1994) Cloning of four cyclins from maize indicates that higher plants have three structurally distinct groups of mitotic cyclins. Proc Natl Acad Sci USA 91: 7375–7379
— Savouré A, Philippe H, Van Montagu M, Inzé D, Rouzé P (1998) Characterization and classification of plant cyclin sequences related to A-and B-type cyclins. In: Francis D, Dudits D, Inzé D (eds) Plant cell division. Portland, Colchester, pp 67–98
Resnitzky D, Hengst L, Reed SI (1995) Cyclin A-associated kinase activity is rate limiting for entrance into S phase and is negatively regulated in Gl by p27Kipl. Mol Cell Biol 15: 4347–4352
Richardson H, Lew DJ, Henze M, Sugimoto K, Reed SI (1992) Cyclin-B homologues inSaccharomyces cerevisiae function in S phase and in G2. Genes Dev 6: 2021–2034
— Wittenberg C, Cross FR, Reed SI (1989) An essential Gl function for cyclin-like proteins in yeast. Cell 59:1127–1133
Riou-Khamlichi C, Huntley R, Jacqmard A, Murray JA (1999) Cytokinin activation ofArabidopsis cell division through a D-type cyclin. Science 283:1541–1544
Rogers S, Wells R, Rechsteiner M (1986) Amino acid sequences common to rapidly degraded proteins: the PEST hypothesis. Science 234: 364–368
Romanowski P, Marr J, Madine MA, Rowles A, Blow JJ, Gautier J, Laskey RA (2000) Interaction of Xenopus Cdc2 x cyclin Al with the origin recognition complex. J Biol Chem 275: 4239–4424
Rosenberg AR, Zindy F, Le Deist F, Mouly H, Metezeau P, Brechot C, Lamas E (1995) Overexpression of human cyclin A advances entry into S phase. Oncogene 10:1501–1509
Russo AA, Jeffrey PD, Pavletich NP (1996) Structural basis of cyclin-dependent kinase activation by phosphorylation. Nat Struct Biol 3: 696–700
Saul DJ, Sudbery PE (1985) Molecular cloning of WHI2, a gene involved in the regulation of cell proliferation inSaccharomyces cerevisiae. J Gen Microbiol 131: 1797–1806
Schneider BL, Yang Q-H, Futcher AB (1996) Linkage of replication to Start by the Cdk inhibitor Sicl. Science 272: 560–562
Schulman BA, Lindstrom DL, Harlow E (1998) Substrate recruit-ment to cyclin-dependent kinase 2 by a multipurpose docking site on cyclin A. Proc Natl Acad Sci USA 95:10453–10458
Schumacher JM, Golden A, Donovan PJ (1998) AIR-2: an Aurora/Ipll-related protein kinase associated with chromosomes and midbody microtubules is required for polar body extrusion and cytokinesis inCaenorhabditis elegans embryos. J Cell Biol 143:1635–1646
Schwob E, Nasmyth K (1993)CLB5 andCLB6, a new pair of B cyclins involved in DNA replication inSaccharomyces cerevisiae. Genes Dev 7: 1160–1175
Böhm T, Mendenhall MD, Nasmyth K (1994) The B-type cyclin kinase inhibitor p40SIC1 controls the Gl to S transition inS. cere-visiae. Cell 79: 233–244
Segal M, Clarke DJ, Maddox P, Salmon ED, Bloom K, Reed SI (2000) Coordinated spindle assembly and orientation requires Clb5p-dependent kinase in budding yeast. J Cell Biol 148: 441–452
Segers G, Gadisseur I, Bergounioux C, Engler JdeA, Jacqmard A, Van Montagu M, Inzé D (1996) TheArabidopsis cyclin-dependent kinase genecdc2bAt is preferentially expressed during S and G2 phases of the cell cycle. Plant J 10: 601–612
Setiady YY, Sekine M, Hariguchi N, Yamamoto T, Kouchi H, Shinmyo A (1995) Tobacco mitotic cyclins: cloning, characteriza-tion, gene expression and functional assay. Plant J 8: 949–957
Shaul O, Mironov V, Burssens S, Van Montagu M, Inzé D (1996) Two Arabidopsis cyclin promoters mediate distinctive transcriptional oscillation in synchronized tobacco B Y-2 cells. Proc Natl Acad Sci USA 93: 4868–4872
Sheaff RJ, Groudine M, Gordon M, Roberts JM, Clurman BE (1997) Cyclin E-CDK2 is a regulator of p27Kipl. Genes Dev 11: 1464–1478
Shen M, Stukenberg PT, Kirschner MW, Lu KP (1998) The essential mitotic peptidyl-prolyl isomerase Pinl binds and regulates mitosis-specific phosphoproteins. Genes Dev 12: 706–720
Sherr CJ (1993) Mammalian Gl cyclins. Cell 73:1059–1065
— (1996) Cancer cell cycles. Science 274:1672–1677
Shirayama M, Toth A, Gálová M, Nasmyth K (1999) APCcdc20 promotes exit from mitosis by destroying the anaphase inhibitor Pdsl and cyclin Clb5. Nature 402: 203–207
Sigrist S, Jacobs H, Stratmann R, Lehner C (1995) Exit from mitosis is regulated byDrosophila fizzy and the sequential destruction of cyclins A, B and B3. EMBO J 14: 4827–4838
Smith HM, Raikhel NV (1999) Protein targeting to the nuclear pore: what can we learn from plants? Plant Physiol 119: 1157–1164
Songyang Z, Blechner S, Hoagland N, Hoekstra MF, Piwnica-Worms H, Cantley LC (1994) Use of an oriented peptide library to deter-mine the optimal substrates of protein kinases. Curr Biol 4: 973–982
Soni R, Carmichael JP, Shah ZH, Murray JAH (1995) A family of cyclin D homologs from plants differentially controlled by growth regulators and containing the conserved retinoblastoma protein interaction motif. Plant Cell 7: 85–103
Sorrell DA, Combettes B, Chaubet-Gigot N, Gigot C, Murray JAH (1999) Distinct cyclin D genes show mitotic accumulation or constant levels of transcripts in tobacco bright yellow-2 cells. Plant Physiol 119: 343–351
Stals H, Bauwens S, Traas J, Van Montagu M, Engler G, Inzé D (1997) Plant CDC2 is not only targeted to the preprophase band, but also co-localises with the spindle, phragmoplast and chromo-somes. FEBS Lett 418:229–234
Stern B, Nurse P (1996) A quantitative model for the cdc2 control of S phase and mitosis in fission yeast. Trends Genet 12: 345–350
Stillman B (1996) Cell cycle control of DNA replication. Science 274:1659–1664
Stuart D, Wittenberg C (1998) Clb5 and Clb6 are required for premeiotic DNA replicatiion and activation of the meiotic S/M checkpoint. Genes Dev 12: 2698–2710
Sundaresan V, Colasanti J (1998) Cyclin dependent kinases in higher plants: spatial and temporal control of cell division. In: Francis D, Dudits D, Inzé D (eds) Plant cell division. Portland, Colchester, pp 47–65
Surana U, Robitsch H, Price C, Schuster T, Fitch I, Futcher AB, Nasmyth K (1991) The role of CDC28 and cyclins during mitosis in the budding yeastS. cerevisiae. Cell 65: 145–161
Tyers M (1996) The cyclin dependent kinase inhibitor p40 SIC1 imposes the requirement for Cln Gl cyclin function at Start. Proc Natl Acad Sci USA 93: 7772–7776
— Tokiwa G, Futcher B (1993) Comparison of theSaccharomyces cerevisiae Gl cyclins: Cln3 may be an upstream activator of Clnl, Cln2 and other cyclins. EMBO J 12:1955–1968
Uetz P, Giot L, Cagney G, Mansfield TA, Judson RS, Knight JR, Lockshon D, Narayan V, Srinivasan M, Pochart P, Qureshi-Emili A, Li Y, Godwin B, Conover D, Kalbfleisch T, Vijayadamodar G, Yang M, Johnston M, Fields S, Rothberg JM (2000) A compre-hensive analysis of protein-protein interactions inSaccharomyces cerevisiae. Nature 403: 623–627
Vlach J, Hennecke S, Amati B (1997) Phosphorylation-dependent degradation of the cyclin-dependent kinase inhibitor p27. EMBO J 16:5334–5544
Vos JW, Hepler PK (1998) Calmodulin is uniformly distributed during cell division in living stamen hair cells ofTradescantia vir-giniana. Protoplasma 201: 158–171
H, Fowke LC, Crosby WL (1997) A plant cyclin-dependent kinase inhibitor gene. Nature 386: 451–452
— Qi Q, Schorr P, Cutler A, Crosby WL, Fowke LC (1998) ICK1, a cyclin dependent protein kinase inhibitor fromArabidopsis thaliana interacts with both Cdc2a and CycD3, and its expression is induced by abscisic acid. Plant J 5: 501–510
Weingartner M, Schweighofer A, Heberle-Bors E, Doonan J, Bögre L (2000) Visualisation of microtubule movements in living mitotic cells. Plant Mol Biol Rep Suppl 18: 2
Wick SM (1991) Spatial aspects of cytokinesis in plant cells. Curr Opin Cell Biol 3: 253–260
Winkler KE, Swenson KI, Kornbluth S, Means AR (2000) Requirement of the prolyl isomerase Pin1 for the replication checkpoint. Science 287:1644–1647
Wittenberg C, Reed SI (1988) Control of the yeast cell cycle is associated with assembly/disassembly of the Cdc28 protein kinase complex. Cell 54:1061–1072
Wu L, Hepler PK, John PCL (1997) Themetl mutation inChlamy-domonas reinhardtii causes arrest at mitotic metaphase with persisting p34cdc2-like H1 histone kinase activity that can promote mitosis when injected into higher-plant cells. Protoplasma 199: 135–150
Xiong Y, Zhang H, Beach D (1992) D type cyclins associate with multiple protein kinases and the DNA replication and repair factor PCNA. Cell 71: 505–514
Yan Z, DeGregori J, Shohet R, Leone G, Stillman B, Nevins JR, Williams RS (1998) Cdc6 is regulated by E2F and is essential for DNA replication in mammalian cells. Proc Natl Acad Sci USA 95: 3603–3608
Zhang K, Tsukitani Y, John PCL (1992) Mitotic arrest in tobacco caused by the phosphoprotein phosphatase inhibitor okadaic acid. Plant Cell Physiol 33: 677–688
— Letham DS, John PCL (1996) Cytokinin controls the cell cycle at mitosis by stimulating the tyrosine dephosphorylation and activation of p34cdc2-like H1 histone kinase. Planta 200: 2–12
Author information
Authors and Affiliations
Additional information
Correspondence and reprints: Plant Cell Biology Group, Research School of Biological Sciences, Australian National University, Canberra, ACT 2601, Australia.
Dedicated to Professor Brian E. S. Gunning on the occasion of his 65th birthday
Rights and permissions
About this article
Cite this article
John, P.C.L., Mews, M. & Moore, R. Cyclin/cdk complexes: Their involvement in cell cycle progression and mitotic division. Protoplasma 216, 119–142 (2001). https://doi.org/10.1007/BF02673865
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02673865