Abstract
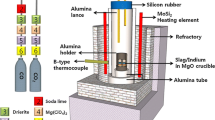
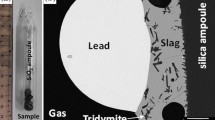
The distribution of tin, selenium and tellurium between alumina-containing fayalitic slags and metallic copper was measured at 1200 and 1300°C under controlled CO-CO2 atmosphere with oxygen partial pressure (pO2) in the rangePO 2 = 10-6 to 10-11 atm (1 atm = 1.013 x 102 kPa). The solubility of Sn in slag was observed to increase linearly with increasing P1/2O2. It was deduced that Sn is present in the slag in the form of SnO or Sn2 and the activity coefficient of SnO in the slag was calculated to be 1.9 at 1200°C and 0.8 at 1300°C. The solubility of Se in the slag decreases with increasing oxygen partial pressure up topO 2l = 4 x 10−8 atm, but above this oxygen partial pressure it becomes practically constant and the ratio (pet Se in slag/pet Se in copper) = 0.018 (at 1200°C) and 0.036 (at 1300°C). The solubility of Te shows a similar variation with oxygen partial pressure and the ratio (pet Te in slag/pet Te in copper) = 0.026 (at 1200°C) and 0.032 (at 1300°C) abovepol 2 = 106 atm. A concept of molecular dissolution of chalcogen elements in slag was developed on the basis of thermodynamic properties of slag, and the observed solubility of Se and Te was explained in terms of the chemical stability of the molecular cluster FeSe and FeTe iη the slag.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
M Nagamori, P. J. Mackey, and P. Tarassoff:Met. Trans. B, 1975, vol. 6B, pp. 295–301.
M. Nagamori:Met. Trans., 1974, vol. 5, pp. 539–48.
M. Nagamori:Met. Trans., 1974, vol. 5, pp. 531–38.
R. Altman and H. H. Kellogg:Trans. Inst. Mining Met., 1972, vol. 81C, pp. 163–75.
J. R. Taylor and J. H. E. Jeffes:Trans. Inst. Mining Met., 1975, vol. 84C, pp. 18–24.
W. J. Rankin and A. K. Biswas:Proc. Australas. Inst. MiningMet., 1974, no. 249, pp. 5–9.
O. Kubaschewski, E. LI, Evans, and C. B. Alcock:Metallurgical Thermochem-istry, Pergamon Press, New York, N.Y., 1967.
K. S. Pitzer and L. Brewer:Thermodynamics by G. N. Lewis and M. Randall, revised ed., McGraw-Hill, New York, N.Y., 1961.
K. K. Kelley: U.S. Bureau of Mines, Bull. 584, 1960.
R. G. Behrens, R. S. Lemons, and G. M. Rosenblatt:J. Chem. Thermodyn., 1974, vol. 6, pp. 457–66.
D. D. Wagman, W. H. Evans, V. B. Parker, I. Harlow, S. M. Bailey, and R. H. Schumm:Nat. Bur. Stand. (U.S.), Tech. Note 270-3, 1968.
D. W. Muenow, J. W. Hastie, R. Hauge, R. Bautista, and J. L. Margrave:Trans. Faraday Soc, 1969, vol. 65, pp. 3210–20.
J. M. Toguri and N. H. Santander:Can. Met. Quart., 1969, vol. 8, pp. 167–71.
R. W. Ruddle, B. Taylor, and A. P. Bates:Trans. Inst. Mining Met., 1966, vol. 75C, pp. 1–12.
M. Hansen:Constitution of Binary Alloys, McGraw-Hill, New York, N.Y., 1958.
J. P. Hager, S. M. Howar, and J. H. Jones:Met. Trans., 1970, vol. 1, pp. 415–30.
M. Hino, T. Azakami, and M. Kameda:Nippon Kogyo Kaishi (Japan), 1973, vol. 89, pp. 749–54.
E. J. Michal and R. Schuhmann:Trans. AIME, 1952, vol. 194, pp. 723–28.
N. Korakas:Trans. Inst. MiningMet., 1962, vol. 72, pp. 35–53.
E. H. Baker:J. Chem. Soc. A, 1968, pp. 1089–92.
E. H. Baker:J Chem. Soc. A, 1967, pp. 1558–60.
N. J. Themelis, G. C. McKerrow, P. Tarrassoff, and G. D. Hallett:J Metals, 1972, vol. 24, no. 4, pp. 25–32.
A. G. Matyas:Metal-Slag-Gas Reactions and Processes, Z. A. Foroulis and W. W. Smelzer, eds., pp. 999–1011, Electrochemical Society, Princeton, N.J., 1975.
S. S. Wang, N. H. Santander, and J. M. Toguri:Met. Trans., 1974, vol. 5, pp. 261–65.
M. Nagamori, T. Hatakeyama, and M. Kameda:Trans. Jap. Inst. Metals, 1970, vol. 11, pp. 190–94.
B. Predel and J. Piehl:Z. Metallkd., 1975, vol. 66, pp. 33–38.
Author information
Authors and Affiliations
Additional information
M. Nagamori is with Centre de Recherche Industrielle du Québec, Ste-Foy, Quebec and P. J. Mackey is Smelter Technical Superintendent, Noranda Mines Limited, Noranda, Quebec, Canada. Both authors were Formerly with Noranda Research Centre, Pointe Claire, Quebec.
Rights and permissions
About this article
Cite this article
Nagamori, M., Mackey, P.J. Distribution equilibria of Sn, Se and Te between FeO-Fe2O3-SiO2-AI2O3-CuO0.5 slag and metallic copper. Metall Trans B 8, 39–46 (1977). https://doi.org/10.1007/BF02656349
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02656349