Summary
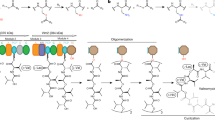
Aspartate transcarbamoylase (ATCase, EC 2.1.3.2) is the first unique enzyme common to de novo pyrimidine biosynthesis and is involved in a variety of structural patterns in different organisms. InEscherichia coli, ATCase is a functionally independent, oligomeric enzyme; in hamster, it is part of a trifunctional protein complex, designated CAD, that includes the preceding and subsequent enzymes of the biosynthetic pathway (carbamoyl phosphate synthetase and dihydroorotase). The complete complementary DNA (cDNA) nucleotide sequence of the ATCase-encoding portion of the hamster CAD gene is reported here. A comparison of the deduced amino acid sequences of the hamster andE. coli catalytic peptides revealed an overall 44% amino acid similarity, substantial conservation of predicted secondary structure, and complete conservation of all the amino acids implicated in the active site of theE. coli enzyme. These observations led to the construction of a functional hybrid ATCase formed by intragenic fusion based on the known tertiary structure of the bacterial enzyme. In this fusion, the amino terminal half (the “polar domain”) of the fusion protein was provided by a hamster ATCase cDNA subclone, and the carboxyl terminal portion (the “equatorial domain”) was derived from a clonedpyrBI operon ofE. coli K-12. The recombinant plasmid bearing the hybrid ATCase was shown to satisfy growth requirements of transformedE. coli pyrB − cells. The functionality of thisE. coli-hamster hybrid enzyme confirms conservation of essential structure-function relationships between evolutionarily distant and structurally divergent ATCases.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Adair LB, Jones ME (1972) Purification and characteristics of aspartate transcarbamylase fromPseudomonas fluorescens. J Biol Chem 247:2308–2315
Bethell MA, Smith KE, White JS, Jones ME (1968) Carbamyl phosphate: an allosteric substrate for aspartate transcarbamylase ofEscherichia coli. Proc Natl Acad Sci USA 60:1442–1449
Blake C (1983) Exons and the evolution of proteins. Trends Biochem Sci 8:11–13
Brabson JS, Maurizi MR, Switzer RL (1985) Aspartate transcarbamylase fromBacillus subtilis. Methods Enzymol 113:627–635
Carrey EA, Hardie DG (1988) Mapping of catalytic domains and phosphorylation sites in the multifunctional pyrimidine-biosynthetic protein CAD. Eur J Biochem 171:583–588
Casadaban MJ, Cohen SN (1979) Lactose genes fused to exogenous promoter in one step using Mu-lac bacteriophage.In vivo probe for transcription control sequences. Proc Natl Acad Sci USA 76:4530–4540
Chang T, Jones ME (1974) Aspartate transcarbamylase fromStreptococcus faecalis. Purification, properties and nature of an allosteric activator site. Biochemistry 13:629–637
Chen EJ, Seeburg PH (1985) Supercoil sequencing—a fast and simple method for sequencing plasmid DNA. DNA 4:165–170
Chou PY, Fasman GD (1974) Conformational parameters for amino acids in helical β-sheet, and random coil regions calculated from proteins. Biochemistry 13:211–245
Chung CH, Goldberg AL (1981) The product of thelon (capR) gene inEscherichia coli is the ATP-dependent protease, protease La. Proc Natl Acad Sci USA 78:4931–4935
Coleman PF, Suttle DP, Stark GR (1977) Purification from hamster cells of the multifunctional protein that initiatesde novo synthesis of pyrimidine nucleotides. J Biol Chem 252:6379–6385
Davidson JN, Niswander LA (1983) Partial cDNA sequence to a hamster gene corrects defect inEscherichia coli pyr B mutant. Proc Natl Acad Sci USA 80:6897–6901
Davidson JN, Patterson D (1979) Alteration in structure of multifunctional protein from Chinese hamster ovary cells defective in pyrimidine biosynthesis. Proc Natl Acad Sci USA 76:1731–1735
Davidson JN, Patterson D (1984) Genes encoding multifunctional proteins—the equivalent of bacterial operons. DNA 3:78
Davidson JN, Carnright DV, Patterson D (1979) Biochemical genetic analysis of pyrimidine biosynthesis in mammalian cell: III. Association of carbamyl phosphate synthtase, aspartate transcarbamoylase, and dihydroorotase in mutants of cultured Chinese hamster cells. Somatic Cell Genet 5:175–191
Davidson JN, Rumsby PC, Tamaren J (1981) Organization of a multifunctional protein in pyrimidine biosynthesis. J Biol Chem 256:5220–5225
Evans DR (1986) CAD, a chimeric protein that initiatesde novo pyrimidine biosynthesis in higher eucaryotes. In: Hardie DG, Coggins JR (eds) Multidomain proteins—structure and evolution. Elsevier, Amsterdam, pp 283–331
Felix H (1982) Permeabilized cells. Anal Biochem 120:211–234
Foltermann KF, Shanley MS, Wild JR (1984) Assembly of the aspartate transcarbamoylase holoenzyme from transcriptionally independent catalytic and regulatory cistrons. J Bacteriol 157:891–898
Foltermann KF, Wild JR, Zink DL, O'Donovan GA (1981) Regulatory variance of aspartate transcarbamoylase among strains ofYersinia enterocolitica andYersinia enterocolitica like organisms. Curr Microbiol 6:43–47
Foltermann KF, Beck DA, Wild JR (1986)In vivo formation of hybrid aspartate transcarbamoylase from native subunits of divergent members of the familyEnterobacteriaceae. J Bacteriol 167:285–290
Freund JN, Jarry BP (1987) Therudimentary gene ofDrosophila melanogaster encodes four enzymic functions. J Mol Biol 193:1–13
Gerhart JC, Schachman HK (1965) Distinct subunits for the regulation and catalytic activity of aspartate transcarbamoylase. Biochemistry 4:1054–1062
Gilbert W, Marchionni M, McKnight G (1986) On the antiquity of introns. Nature 271:501
Grayson DR, Evans DR (1983) The isolation and characterization of the aspartate transcarbamoylase domain of the multifunctional protein, CAD. J Biol Chem 258:4123–4129
Grayson DR, Lee L, Evans DR (1985) Immunochemical analysis of the domain structure of CAD, the multifunctional protein that initiates pyrimidine biosynthesis in mammalian cells. J Biol Chem 260:15840–15849
Hoffman CS, Wright A (1985) Fusions of secreted proteins to alkaline phosphatase: an approach for studying secretion. Proc Natl Acad Sci USA 82:5107–5111
Honzatko RB, Lipscomb WN (1982) Interactions of phosphate ligands ofEscherichia coli aspartate carbamoyltransferase in the crystalline state. J Mol Biol 160:264–286
Honzatko RB, Crawford JL, Monaco HL, Ladner JE, Edwards BFP, Evans DR, Warren SF, Wiley DC, Ladner RC, Lipscomb WN (1982) Crystal and molecular structures of native and ATP-liganded aspartate carbamoyltransferase fromEscherichia coli. J Mol Biol 160:219–263
Hoover TA, Roof WD, Foltermann KF, O'Donovan GA, Bencini DA, Wild JR (1983) Nucleotide sequence of the structural gene (pyrB) that encodes the catalytic polypeptide of aspartate transcarbamoylase ofEscherichia coli. Proc Natl Acad Sci USA 80:2462–2466
Houghton JE (1986) Structural and functional comparisons between OTCase and ATCase, leading to the formation and characterization of an active “domain fusion” betweenargl (OTCase) andpyrB (ATCase). Dissertation, Texas A&M University, College Station TX
Jones ME (1980) Pyrimidine nucleotide biosynthesis in animals: genes, enzymes, and regulation of UMP biosynthesis. Annu Rev Biochem 49:253–279
Kania J, Mueller-Hill B (1980) Artificial protein chimeras. In: Bisswanger H, Schmincke-Ott E (eds) Multifunctional proteins. Wiley, New York, pp 31–47
Kedzie KM (1987) Characterization of thepyrB gene ofSerratia marcescens and hybrid gene formation with thepyrB gene ofEscherichia coli, leading to the production of chimeric ATCases. Dissertation, Texas A&M University, College Station TX
Keppler D, Holstege A (1982) Pyrimidine nucleotide metabolism and its compartmentation. In: Sies H (ed) Metabolic compartmentation. Academic Press, London, pp 149–203
Krause KL, Volz KW, Lipscomb WN (1987) 2.5 A structure of aspartate carbamoyltransferase complexed with the bisubstrate analogue N-(phosphonacetyl)-L-aspartate. J Mol Biol 193:527–553
Lerner CG, Switzer RL (1986) Cloning and structure of theBacillus subtilis aspartate transcarbamoylase gene (pyrB). J Biol Chem 261:11156–11165
Lovatt CJ, Cheng AH (1984) Aspartate transcarbamoylase. Site of end product inhibition of the orotate pathway in intact cells ofCucurbita pepo. Plant Physiol 75:511–515
Lue PF, Kaplan JG (1969) The aspartate transcarbamoylase and carbamoyl phosphate synthetase of yeast: a multifunctional enzyme complex. Biochem Biophys Res Commun 34:426–433
Makoff AJ, Radford A (1978) Genetics and biochemistry of carbamoyl phosphate biosynthesis and its utilization in the pyrimidine biosynthetic pathway. Microbiol Rev 42:307–328
Maley JA, Davidson JN (1988) The aspartate transcarbamoylase domain of a mammalian multifunctional protein expressed as an independent enzyme inEscherichia coli. Mol Gen Genet 213:278–284
Maley JA, Niswander LA, Flower AM, Davidson JN (1984) Dissecting the organization of the hamster CAD gene by expression of cDNA pieces inE. coli mutants. DNA 3:93
Mally MI, Grayson DR, Evans DR (1981) Controlled proteolysis of the multifunctional protein that initiates pyrimidine biosynthesis in mammalian cells: evidence for discrete domains. Proc Natl Acad Sci USA 78:6647–6651
Mas MT, Chen CY, Hitzeman RA, Riggs AD (1986) Active human-yeast chimeric phosphoglycerate kinases engineered by domain interchange. Science 233:788–790
Messing JRC, Seeburg PH (1981) A system for shot-gun DNA sequencing. Nucleic Acids Res 9:309–321
Monaco HL, Crawford JL, Lipscomb WN (1978) Three-dimensional structure of aspartate carbamyltransferase fromEscherichia coli and of its complex with cytidine triphosphate. Proc Natl Acad Sci USA 75:5276–5280
Nishikawa K (1983) Assessment of secondary structure prediction of protein comparison of computerized Chou-Fasman method with others. Biochim Biophys Acta 748:285–299
Ong BL, Jackson JF (1972) Pyrimidine nucleotide biosynthesis inPhaseolus aureus. Biochem J 129:583–593
Prescott LM, Jones ME (1969) Modified methods for the determination of carbamyl phosphate. Analyt Biochem 32:408–419
Rashin AA (1981) Location of domains in globular proteins. Nature 291:85–87
Roof WD, Foltermann KF, Wild JR (1982) ThepyrB operon ofE. coli possesses arho independent attenuator sequence. Mol Gen Genet 187:391–400
Ruiz JC, Wahl GM (1986)Escherichia coli aspartate transcarbamoylase: a novel marker for studies of gene amplification and expression in mammalian cells. Mol Cell Biol 6:3050–3058
Rumsby PC, Campbell PC, Niswander LA, Davidson JN (1984) Organization of a multifunctional protein in pyrimidine biosynthesis. Biochem J 217:435–440
Sancar A, Hack AM, Rupp WD (1979) Simple method for identification of plasmid-coded proteins. J Bacteriol 137:692–693
Sanger FS, Nicklen S, Coulson AR (1977) DNA sequencing with chain-terminating inhibitors. Proc Natl Acad Sci USA 74:5463–5467
Schneider WP, Nichols BP, Yanofsky C (1981) Procedure for production of hybrid genes and proteins and its use in assessing significance of amino acid differences in homologous tryptophan synthetase alpha polypeptides. Proc Natl Acad Sci USA 78:2169–2173
Shigesada K, Stark GR, Maley JA, Niswander LA, Davidson JN (1985) Construction of a cDNA to the hamster CAD gene and its application toward defining the domain for aspartate transcarbamoylase. Mol Cell Biol 5:1735–1742
Silhavy TS, Berman ML, Enquist LW (eds) (1984) Experiments with gene fusions. Cold Spring Harbor Press, Cold Spring Harbor NY, pp 273–281
Simon LD, Randolph B, Irwin N, Binkowski G (1983) Stabilization of proteins by a bacteriophage T4 gene cloned inEscherichia coli. Proc Natl Acad Sci USA 80:2059–2062
Stark GS (1977) Multifunctional proteins: one gene—more than one enzyme. Trends Biochem Sci 2:64–66
Swamy KHS, Goldberg AL (1981)E. coli contains eight soluble proteolytic activities, one being ATP dependent. Nature 292:652–654
Traut TW (1986) Are proteins made of modules? Mol Cell Biochem 70:3–10
Volz KW, Krause KL, Lipscomb WN (1986) The binding of N-(phosphonacetyl)-L-aspartate to aspartate carbamoyltransferase ofEscherichia coli. Biochem Biophys Res Commun 136:822–836
Wild JR, Foltermann KF, O'Donovan GA (1980) Regulatory divergence of aspartate transcarbamoylases within the Enterobacteriaceae. Arch Biochem Biophys 201:506–517
William LG, Bernhardt S, Davis RH (1970) Copurification of pyrimidine specific carbamyl phosphate synthetase and aspartate transcarbamylase ofNeurospora crassa. Biochemistry 9:4329–4335
Zinder ND, Boeke JD (1982) The filamentous phage as vectors for recombinant DNA—a review. Gene 19:1–10
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Major, J.G., Wales, M.E., Houghton, J.E. et al. Molecular evolution of enzyme structure: Construction of a hybrid hamster/Escherichia coli aspartate transcarbamoylase. J Mol Evol 28, 442–450 (1989). https://doi.org/10.1007/BF02603079
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF02603079