Abstract
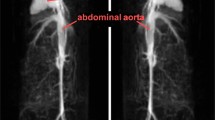
Rationale and objectives: To develop and partially characterize a new class of potential blood pool magnetic resonance (MR) contrast agents.Methods: Various copolymeric chelates of gadolinium-diethylenetriamine pentaacetic acid (Gd-DTPA) were prepared with differing molecular weights of polyethylene glycol (PEG) or polypropylene glycol (PPG) as linkers between the monomeric chelate units. Gadolinium content of the polymeric chelates was determined by atomic absorption spectra. Relaxivity of the polymeric chelates was measured at 1.5 Tesla and compared with Gadolinium-DTPA. MR angiography (MRA) was performed in rabbits comparing Gd-DTPA with Gd-copolymers.Results: The gadolinium content of the copolymeric chelates ranged from 2.95 to 22.2% on weight basis. The molecular weight of the PEG linkers in the copolymers ranged from about 150 to about 3400. Ther1 (1/T1, mM−1 s−1) for Gd-DTPA=4.1. Ther1 values for the different Gd-containing polymers ranged from 3.8 to 5.8, with the lowestr1 for the polymer prepared with the lowest-molecular-weight complex. The higher-molecular-weight complexes resulted in moderately higher relaxivity. MRA with Gd-copolymers, in rabbits, showed markedly greater vascular enhancement relative to an equivalent dose of Gd-DTPA. Vascular enhancement was much more sustained with the copolymeric agent and confined to vascular space; i.e. no appreciable background tissue enhancement—a reflection of distribution into extravascular fluid space—was observed.Conclusions: Relative to Gd-DTPA monomers, PEG-containing Gd-DTPA polymeric complexes provided moderate increases in relaxivity but markedly greater efficacy during in vivo MRA. In vitro relaxivity studies of Gd-copolymers showed only an approximately 50% increase inr1 relaxivity compared with Gd-DTPA. The PEG-containing complex's lack of rigidity may have diminished the effect of spin diffusion on relaxation, thereby accounting for this modest increase. The greater efficacy of Gd-copolymers during in vivo MRA may reflect compartmentalization within the vascular space and possibly enhanced relaxation of the macromolecular copolymers in the blood. Gd-copolymers are promising agents that merit additional study.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
Blustajn J, Cuenod C-A, Clement O, Siauve N, Vuillemin-Bodaghi V, Frija G. Measurement of liver blood volume using a macromolecular MRI contrast agent at equilibrium. Mag Res Imaging 1997;15(4):415–21.
Shames D, Kuwatsuru R, Vexler V, Mühler A, Brasch R. Measurement of capillary permeability to macromolecules by dynamic magnetic resonance imaging: a quantitative noninvasive technique. Magn Reson Med 1993;29:616–22.
Demsar F, Roberts TPL, Schwickert HC, Shames DM, van Dijke CF, Mann JS, Saeed M, Brasch RC. An MRI spatial mapping technique for microvascular permeability and tissue blood volume based on macromolecular contrast agent distribution. Magn Reson Med 1997;37:236–42.
Brasch RC. Rationale and applications for macromolecular Gd-based contrast agents. Magn Reson Med 1991;22:282–7.
Weinmann HJ, Laniado M, Mutzel W. Pharmacokinetics of Gd-DTPA/dimeglumine after intravenous injection into healthy volunteers. Physiol Chem Phys Med NMR 1988;16:167–78.
Wedeking P, Tweedle MF. Comparison of the biodistribution of153Gd-labeled Gd(DTPA)−2 and Gd(DOTA) and Gd(acetate) in mice. Int J Rad Appl Inst 1988;15:395–401.
Weinmann HJ, Brasch RC, Press WR, Wesby GE. Characteristics of gadolinium-DTPA complex: a potential NMR contrast agents. Am J Roentgenol 1984;142:619–24.
Brasch RC, Weinmann HJ, Wesbey GE. Contrast enhanced NMR imaging: animal studies using gadolinium-DTPA complex. Am J Roentgenol 1984;142:625–30.
Unger EC, Schilling JD, Bernhard VM, Awad AN, McIntrye KE, Yoshino MT, Pond GD, Darkazanli A, Hunter GC. Magnetic resonance angiography of the foot and ankle. J Magn Reson Imaging 1995;5:1–5.
Wagnon BK, Jackels SC. Synthesis, characterization, and aqueous proton relaxation enhancement of a manganese (II) heptaaza macrocyclic complex having pendant arms. Inorg Chem 1987;28(10):1923–7.
Burton DR, Forsen S, Karlstrom G, Dwek RA. Proton relaxation enhancement (PRE) in biochemistry: a critical survey. Prog NMR Spectrosc 1979;13(1):1–45.
In: U.S. Pharmacopeia/National Formulary (USP XXII/NF XVII). United States Pharmacopeial Convention, Inc., Rockville, MD 1990.
Shen D, Fritz TA, Wu G, Kulik B, Palestrant D, Unger EC. Block copolymer magnetic resonance contrast agents. Invest Radiol 1994;29(Suppl 2):S217–9.
Shen D, Unger EC, Wu G, Fritz TA, Kulik B, New TE. Copolymeric MR Contrast Agents. New York: Society of Magnetic Resonance Imaging, 1992 Abstract.
Unger EC, Wu G. Copolymers and Their Use as Contrast Agents in Magnetic Resonance Imaging and in Other Applications. U.S. Patent 5,385,719. Filed September 22, 1992. Issued January 31, 1995.
Unger EC, Wu G. Copolymers and Their Use as Contrast Agents in Magnetic Resonance Imaging and in Other Applications. U.S. Patent 5,458,127. Filed September 13, 1994. Issued October 17, 1995.
Unger EC, Wu G. Copolymers and Their Use as Contrast Agents in Magnetic Resonance Imaging and in Other Applications. U.S. Patent 5.517,993. Filed June 7, 1995. Issued May 21, 1996.
Snow RA, Ladd DL, Toner JL. Polyalkylene Oxide Polymer Conjugates with Chelating Agents for Therapeutic and Diagnostic Imaging Compositions and Methods. PCT publication WO 94/08624.
Elizondo G, Fretz CJ, Stark DD, Rocklage SM, Quay SC, Worah D, Tsang Y-M, Chen MC-M, Ferrucci JT. Preclinical evaluation of MnDPDP: new paramagnetic hepatobiliary contrast agent for MR imaging. Radiology 1991;178:73–8.
Mena I, Manganese. In: Disorders of Mineral Metabolism, vol. 1. Orlando, FL: Academic Press, 1981:233–70.
Keen CL, Lonnerdal B. Manganese toxicity in man and experimental animals. In: Schramm VL, Wedler FC, editors. Manganese in Metabolism and Enzyme Function, Orlando, FL: Academic Press, 1986:35–47.
Misselwitz B, Mühler A, Weinmann H-J. A toxicologic risk for using manganese complexes? a literature survey of existing data through several medical specialties. Invest Radiol 1995;30(10):611–20.
Sur SK, Bryant RG. Nuclear- and electron-spin relaxation rates in symmetrical iron, manganese, and gadolinium ions. J Phys Chem 1995;99(17):6301–8.
Kellar KE, Henrichs PM, Hollister R, Koenig SH, Eck J, Wei D. High relaxivity linear Gd(DTPA)-polymer conjugates: the role of hydrophobic interactions. Mag Res Med 1997;38(5):712–6.
Lester CC, Bryant RG. Magnetically coupled paramagnetic relaxation agents. Magn Res Med 1992;24:236–32.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Unger, E.C., Shen, D., Wu, G. et al. Gadolinium-containing copolymeric chelates—a new potential MR contrast agent. MAGMA 8, 154–162 (1999). https://doi.org/10.1007/BF02594593
Issue Date:
DOI: https://doi.org/10.1007/BF02594593