Summary
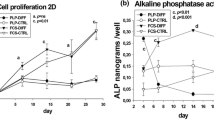
Periostea were dissected from 17-day-old chicken embryo calvariae, placed on millipore filters, and cultured on fluid media containing serum or on serum and plasma containing “plasma clots,” in three ways: 1) with the osteogenic layer facing the filter, 2) with the osteogenic layer away from the filter, 3) folded such that the osteogenic layer was in apposition with itself within the fold. The cultures were studied histologically as well as biochemically. Periostea that were cultured folded showed differentiation of osteoblastlike cells after 2 days, and production of osteoid at day 4. Tissues cultured with the osteogenic layer away from the filter demonstrated similar osteoblastic differentiation and osteoid production. Both types of cultures exhibited an increase in histochemically detectable alkaline phosphatase activity over the 4 day culture period that was associated with osteoblasts and the osteogenic area. Periostea cultured with the osteogenic layer facing the filter produced no osteoid. In these cultures, histochemically detectable alkaline phosphatase activity decreased and virtually disappeared over the 4 day culture period. The possibility that the creation of a suitable micro-environment is required for osteodifferentiation in this culture system is discussed.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Fell HB (1932) The osteogenic capacity in vitro of periosteum and endosteum isolated from the limb skeleton of fowl embryos and young chicks. Anatomy LXVI, pp 11–180
Gaillard PJ (1934) Development changes in the composition of the body fluids in relation to growth and differentiation of tissue cultures. Protoplasma XXIII, pp 145–174
Fitton-Jackson S, Smith RH (1957) Studies on the biosynthesis of collagen. J Biophys Biochem Cyto 3:897–911
Goldhaber P (1966) Remodelling of bone in tissue culture. J Dent Res 45:490–499
Binderman I, Duksin O, Harrell A, Katzir E, Sachs L (1964) Formation of bone tissue in culture from isolated bone cells. J Cell Biol 61:427–439
Marvaso V, Bernard GW (1977) Initial intramembraneous osteogenesis in vitro. Am J Anat 149:453–468
Raisz LG, Dietrich JW, Canalis EM (1976) Factors influencing bone formation in organ culture. Isr J Med Sci 12:108–114
Brighton CT, Fox JL, Seltzer D (1976) In vitro growth of bone and cartilage from rat periosteum under various oxygen tensions (Abstract). 22nd Annual ORS, January 28–30, p 51
Osdoby P, Caplan AI (1979) Osteogenesis in cultures of limb mesenchymal cells. Dev Biol 73:84–102
Nijweide P, Van der Plas A (1975) Studies on the uptake of calcium and strontium by isolated periosteal cells. Proc Kon Ned Akad Wet C78:418–434
Tenenbaum HC, Heersche JNM (1982) Differentiation of osteoblasts and production of mineralized bone in vitro. Calcif Tissue Int 34:76–79
Paul J (1975) Cell and tissue culture, 5th ed. Churchill-Livingston, London
Burstone MS (1960) Enzymes in dentinogenesis and osteogenesis: calcification in biological systems. AAAS, Symposium Publication, Washington, pp 225–227
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Slater CR (1976) Control of myogenesis in vitro by chick embryo extract. Dev Biol 50:264–284
Linkhart TA, Glegg CH, Hauschka SD (1981) Myogenic differentiation in permanent clonal mouse myoblast cell lines: regulation by macromolecular growth factor in the culture medium. Dev Biol 66:19–30
Burger EH, Gribnau JD, Thesingh CW (1981) Initiation of bone formation and resorption in vitro: effects of oxygen tension and medium pH. Cell Biol Int Rep 5(8):764
Tenenbaum HC, Smith I, Heersche JNM (1983) Differentiation of osteoid producing cells in vitro: evidence for the requirement of a microenvironment. ASBMR 5th Annual Scientific Meeting (Abstract A15)
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Tenenbaum, H.C., Heersche, J.N.M. Differentiation of osteoid-producing cellsin vitro: Possible evidence for the requirement of a microenvironment. Calcif Tissue Int 38, 262–267 (1986). https://doi.org/10.1007/BF02556604
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF02556604