Abstract
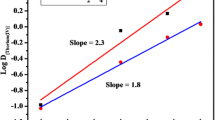
The extraction of thorium(IV) from perchlorate solutions with di-n-butyldithiophosphoric acid (HBudtp) in various organic solvents occurs through an ion exchange mechanism. The extracted species in the organic phase is an eight-coordinate complex Th(Budtp)4. The higher values of the distribution ratio obtained in HBudtp-benzene-water system than in HBudtp-n-butanol-water system are explained by higher solubility of the complex species in nonpolar solvents. The position of the extraction curves in the pH-range lower than 0.7 reduces the complexation of thorium(IV) with Budtp− in the aqueous phase and also the hydrolysis process.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
M. Curtui, I. Haiduc,Radiochem. Radioanal. Lett., 50 (1981) 55.
M. Curtui, I. Haiduc, Iovanca Haiduc,J. Radioanal. Nucl. Chem., Lett., 165 (1992) 95.
K. Sasse, Methoden der Organischen Chemie (Houben-Weil) Band XII, Teil 2, G. Thieme Verlag, Stuttgart, 1964.
M. Pinta, Recherche et dosage des elements traces, Dunod, Paris, 196.
G. Marcu, M. Curtui, I. Haiduc,J. Inorg. Nucl. Chem., 39 (1977) 1415.
A.D. Jones, G.R. Chopin,Actinide Rev., 1 (1969) 311.
A.A. Pinkerton, A.E. Storey, J.M. Zellweger,J. Chem. Soc., Dalton Trans., (1981) 1475.
M. Curtui, G. Marcu, I. Haiduc,Studia Unov. Babes-Bolyai, Chemia, 21 (1976) 74.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Curtui, M., Haiduc, I. Solvent extraction of thorium(IV) with dibutyldithiophosphoric acid in various organic solvents. Journal of Radioanalytical and Nuclear Chemistry Letters 186, 273–280 (1994). https://doi.org/10.1007/BF02162758
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02162758