Summary
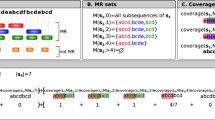
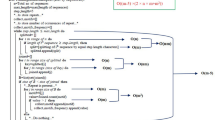
51 polypeptides of known 3-dimensional structures have been submitted to a search for internal similarities. It is shown that the frequency of proteins displaying significant amounts of internal similarities is higher than predicted by chance. A non-negligeable part of those similarities probably occurs in connection with the existence of ordered secondary structures. Indeed, similarity occurs at a much more important rate when analyses are restricted to protein subsequences corresponding to α helices or β pleated sheets. Furthermore, the correlation existing between the rates at which linear and inverted repeats occur inside protein subregions of ordered secondary structures suggests that a significant part of short similarities are analogies rather than homologies. An hypothesis is put forward suggesting that the regular alternations of hydrophobicity which characterize most of α helices and β strands could provoke the occurrence of significant amounts of similarities inside protein sequences.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Amzel LL, Chen BL, Phizackerley RP, Poljak RJ, Saul G (1974) High resolution X-Ray diffraction studies of the Fab' Fragment of IgG NEW. Progr Immunol 1:85–92
Bauer K (1971) Homology of a partial sequence of calf thymus histone IV with several non-histone proteins. Int J Pept Protein Res 3:165–172
Dayhoff MO (1978) Atlas of protein sequences. National Biomedical Research Foundation Suppl 3, pp 359–362
Delhaise P, Wuilmart C, Urbain J (1980) Relationships between alpha and beta secondary structures and amino acid pseudosymmetrical arrangments. Eur J Biochem 105:553–564
Eck RV, Dayhoff MO (1966) Evolution of the structure of Ferredoxin based on living relics of primitive amino acid sequences. Science 152:363–366
Fitch WM (1966) An improved method of testing for evolutionary homology. J. Mol Biol 16:9–16
Fitch WM (1970) Further improvements in the method of testing for evolutionary homology among proteins. J Mol Biol 49:1–14
Fitch WM (1973) Aspects on molecular evolution. Ann Rev Genet 7:343–380
Greller LD, Erhan S (1974) Short length amino acid sequence homology among ancestrally unrelated proteins. Int J Pept Protein Res 6:165–174
Levitt M, Greer J (1977) Automatic identification of secondary structure in globular proteins. J Mol Biol 114:181–239
McLachlan AD (1972) Repeating sequences and gene duplication in proteins. J Mol Biol 64:417–437
Richardson J (1977) Beta-sheet topology and the relatedness of proteins. Nature 268:495–500
Schiffer M, Edmundson AB (1967) Use of helical wheels to represent the structures of proteins and to identify segments with helical potential. Biophys J 7:121–135
Sternberg M, Thornton JM (1977) On the conformation of proteins: hydrophobic ordering of strands in beta-pleated sheets. J Mol Biol 115:1–17
Urbain J (1969) Evolution of immunoglobulins and ferredoxins and the occurrence of pseudosymmetrical sequences. Biochem Genet 3:249–269
Woese CR (1971) Evolution of macromolecular complexity. J Theor Biol 33:29–34
Wuilmart C, Wijns L, Urbain J (1975) Linear and inverted repetitions in protein sequences. J. Mol Evol 5:259–278
Wuilmart C, Delhaise P, Urbain J (1982) The sharing of amino acid short spans by ancestrally unrelated proteins may be the result of ubiquitous alpha and beta secondary structures. Bio system 15:221–232
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Wuilmart, C., Delhaise, P. Linear repetitions of amino acids and convergent evolution inside protein subregions of ordered secondary structures. J Mol Evol 19, 355–361 (1983). https://doi.org/10.1007/BF02101639
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF02101639