Abstract
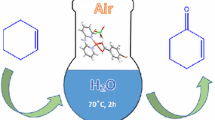
In aqueous alkaline solutions copper phenanthroline complexes catalyze the oxidation of primary alcohols and cyclohexanol to carboxylic acids at 333–373 K and\(P_{O_2 } \)=0.2–0.4 MPa. Unlike in radical-chain and metal-complex oxidation, secondary and acyclic alcohols are not oxidized in these conditions.
Abstract
В водных щелочных растворах фенантролиновые комплексы меди катализируют окисление первичных спиртов и циклогексанола в карбоновые кислоты при температурах 333–313 К и давлении кислорода 0,2–0,4 МПа. Вторичные ациклические спирты не окисляются в этих условиях в отличие от радикальноцепного и металлокомплексного окисления.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
W. Brackman, C. I. Gaasbeek: Recl. Trav. Chim., Pays-Bas,85, 242 (1966).
O. N. Emanuel, A. M. Sakharov, I. P. Skibida: Izv. Akad. Nauk SSSR, Ser. Khim.,12, 2692 (1975).
I. V. Kozhevnikov, V. E. Taraban'ko, K. I. Matveev: Kinet. Katal.,21, 940 (1980).
E. T. Denisov, N. I. Mitskevich, V. E. Agabekov: Mechanisms of the Oxidation of Oxygen-Containing Compounds in Liquid Phase, p. 26. Nauka i Tekhnika, Minsk 1975.
A. E. Shilov, A. A. Shteinman: Kinet. Katal.,14, 149 (1973).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Tretyakov, V.P., Zimtseva, G.P., Rudakov, E.S. et al. Unusual selectivity of alcohol oxidation by oxygen in aqueous alkaline solutions of copper phenanthroline complexes. React Kinet Catal Lett 19, 263–266 (1982). https://doi.org/10.1007/BF02074042
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02074042