Abstract
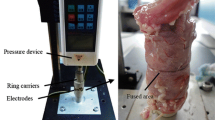
PURPOSE: The study investigated the influence of pulsed electromagnetic fields (PEMFs) on the mechanical strength and collagen content of uncomplicated colonic anastomosis in rats. METHODS: A standardized left colonic resection was performed 3 cm above the peritoneal reflection, and end-to-end anastomosis was constructed with eight interrupted inverting sutures. Beginning immediately after surgery, randomly assigned groups were exposed to one of the following: 1) 100 Hz (frequency), 1 mT (intensity) PEMFs with 16-hour on/8-hour off cycles (n=8); 2) 100 Hz, 2 mT PEMFs with 16-hour on/8-hour off cycles (n=8); 3) 100 Hz, 1 mT PEMFs with 6-hour on/6-hour off cycles (n=6), whereas the control group (n=10) received no PEMFs. Relaparatomy was performed at 72 hours postoperatively, and the bursting pressure of the anastomotic segment was recorded in situ.The hydroxyproline contents of the anastomotic and adjacent perianastomotic segments of equal lengths were determined. RESULTS: Mean bursting pressure values of the groups that received 100 Hz, 1 or 2 mT PEMFs with 16-hour on/8-hour off cycles (90.88±19.13 and 83.88±7.08 mmHg, respectively) were significantly higher than those of the control group (61.66±10.6 mmHg) and the group with 6-hour on/6-hour off cycles (64.83±7.36 mmHg;P <0.05 for all comparisons). Hydroxyproline contents of the anastomotic and perianastomotic segments were consistently higher in the 16-hour on/8-hour off PEMF groups, compared with those of the corresponding segments of the control group. CONCLUSIONS: PEMFs applied externally to unrestrained rats within a “window of PEMF parameters” provided a significant gain in the mechanical strength of the colonic anastomosis, at least 72 hours postoperatively. Associated relative increases in the hydroxyproline contents of the (peri)anastomotic colonic segments suggest that an altered collagen metabolism might contribute to this enhancement of the anastomotic repair. Further investigations based on these preliminary data and the definition of the exact measures regarding the effects of PEMFs on biologic systems, in general, may lead to an efficient and new adjunctive modality in colorectal surgery.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Khoury GA, Waxman BP. Large bowel anastomoses. 1. The healing process and sutured anastomoses—a review. Br J Surg 1983;70:61–3.
Schrock TR, Deveney CW, Dunphy JE. Factors contributing to leakage of colonic anastomoses. Ann Surg 1973;177:513–8.
Fielding LP, Stewart-Brown S, Blesovsky L, Kearny G. Anastomotic integrity after operations for large-bowel cancer: a multicenter study. BMJ 1980;281:411–4.
Brasken P. Healing of experimental colon anastomosis. Eur J Surg 1991;566(Suppl):2–51.
Graham MF, Blomquist P, Zederfelt B. The alimentary canal. In: Linblad CD, ed. Wound healing. Biochemical and clinical aspects. Philadelphia: WB Saunders, 1992; 433–49.
Miller EJ, Gay S. Collagen structure and function. In: Linblad CD, ed. Wound healing: biochemical and clinical aspects. Philadelphia: WB Saunders, 1992;130–51.
Irvin TT. Collagen metabolism in infected colonic anastomoses. Surg Gynecol Obstet 1976;143:220–4.
Jiborn H, Ahonen J, Zederfelt B. Healing of experimental colonic anastomoses. III. Collagen metabolism in the colon after left colon resection. Am J Surg 1980;139:398–405.
Moss G, Greenstein A, Levy S, Bierenbaum A. Maintenance of GI function after bowel surgery and immediate enteral full nutrition. I. Doubling of canine colorectal anastomotic bursting pressure and intestinal wound mature collagen content. J Parenteral Nutr 1980;4:535–8.
Gottrup F. Healing of incisional wounds in stomach and duodenum: collagen distribution and relation to mechanical strength. Am J Surg 1981;141:222–7.
Kamuro T. The lattice arrangement of collagen fibers in the submucosa of the rat small intestine: scanning electron microscopy. Cell Tissue Res 1988;251:117–21.
Hendriks T, Mastboom WJ. Healing of experimental intestinal anastomoses: parameters for repair. Dis Colon Rectum 1990;33:891–901.
Delaney PV, Lalor D, Fitzgerald P, O'Malley E. The use of ‘collagenase inhibitor’ in prevention of large bowel anastomotic dehiscence. Br J Surg 1974;61:923–4.
Högström H, Haglund U, Zederfeit B. Beneficial effect on intestinal anastomosis of S-2441, a synthetic kallikrein-kinin antagonist: experimental studies in the rat. Am J Surg 1985;150:312–3.
Högström H, Haglund U. Neutropenia prevents decrease in strength of rat intestinal anastomosis: partial effects of oxygen free radical scavengers and allopurinol. Surgery 1986;99:716–20.
Brennan SS, Foster ME, Morgan A, Leaper DJ. Prostaglandins in colonic anastomotic healing. Dis Colon Rectum 1984;27:723–5.
Marino AA, ed. Modern bioelectricity. New York: Marcel Dekker, 1988.
Blank M, ed. Electricity and magnetism in biology and medicine. San Francisco: San Francisco Press, 1993.
Goodman R, Henderson AS. Some biological effects of electromagnetic fields. Bioelectrochemistry and Bioenergetics 1986;15:39–55.
Byus CV, Pieper ST, Adey WR. The effects of low energy 60 Hz environmental electromagnetic fields upon the growth-related enzyme ornithine decarboxylase. Carcinogenesis 1987;8:1385–9.
Goodman R, Chizmadzhevy Y, Shirley-Henderson A. Electromagnetic fields and cells. J Cell Biochem 1993;51:436–41.
Bassett CA. Fundamental and practical aspects of therapeutic uses of pulsed electromagnetic fields. Crit Rev Biomed Eng 1989;17:451–529.
Murray JC, Ferndale RW. Modulation of collagen production in cultured fibroblasts by a low-frequency pulsed magnetic field. Biochim Biophys Acta 1985;838:98–105.
Yen-Patton GP, Patton WF, Beer DM, Jacobson BS. Endothelial cell response to pulsed electromagnetic fields: stimulation of growth rate and angiogenesisin vitro. J Cell Physiol 1988;134:37–46.
Rodemann HP, Bayrenther K, Pfliderer G. The differentiation of normal and transformed human fibroblastsin vitro is influenced by electromagnetic fields. Exp Cell Res 1989;182:610–21.
Lin Y, Nishimura R, Nozaki K,et al. Collagen production and maturation at the experimental ligament defect stimulated by pulsing electromagnetic fields in rabbits. J Vet Med Sci 1993;55:527–31.
Cridland NA, Saunders RD, Cragg TA. Proliferative responses of human fibroblasts exposed to ELF magnetic fields. In: Blank M, ed. Electricity and magnetism in biology and medicine. San Francisco: San Francisco Press, 1993;628–32.
Bassett CA, Mitchell SN, Gaston SR. Pulsing electromagnetic field treatment in ununited fractures and failed arthrodeses. JAMA 1982;24:623–8.
Watkins JP, Auer JA, Morgan SI, Gay S. Healing of surgically created defects in the equine superficial digital flexor tendon: effects of pulsed electromagnetic field therapy on collagen-type transformation and tissue morphologic reorganization. Am J Vet Res 1985;46:2097–103.
Sisken BF, Kanje M, Lundborg G, Herbst E, Kurtz W. Stimulation of rat sciatic nerve regeneration with pulsed electromagnetic fields. Brain Res 1989;485:309–16.
Zienowicz RJ, Thomas BA, Kurtz WH, Orgel MG. A multivariate approach to the treatment of peripheral nerve transection injury: the role of electromagnetic field therapy. Plast Reconstr Surg 1991;87:122–9.
Battini R, Monti MG, Moruzzi MS, Ferrari S, Zaniol P, Barbiroli B. ELF electromagnetic fields affect gene expression of regenerating rat liver following partial hepatectomy. J Bioelectricity 1991;10:131–9.
Stiller MJ, Pak GH, Schupack JL, Thaler S, Kenny C, Jandreau L. A portable pulsed electromagnetic field (PEMF) device to enhance healing of recalcitrant venous ulcers: a double blind, placebo-controlled clinical trial. Br J Dermatol 1992;127:147–54.
Aaron RK, Ciombor D. Therapeutic effects of electromagnetic fields in the stimulation of connective tissue repair. J Cell Biochem 1993;52:42–6.
Jamall IS, Finelli VN, Que Hee SS. A simple method to determine nanogram levels of 4-hydroxyproline in biological tissues. Anal Biochem 1981;112:70–5.
Jiborn H, Ahonen J, Zederfelt B. Healing of experimental colonic anastomoses. I. Bursting strength of the colon after left colon resection and anastomosis. Am J Surg 1978;136:587–94.
Hawley PR, Faulk WP, Hunt TK, Dunphy JE. Collagenase activity in the gastrointestinal tract. Br J Surg 1970;57:896–900.
Hunt TK, Hawley PR, Hale J, Goodson W, Thakral KK. Colon repair: the collagenous equilibrium. In: Hunt TK, ed. Wound healing and wound infection: theory and surgical practice. New York: Appleton-Century-Crofts, 1980:153–9.
Parry DA. The molecular and fibrillar structure of collagen and its relationship to the mechanical properties of connective tissue. Biophys Chem 1988;29:195–209.
Foulds IS, Barker AT. Human skin battery potentials and their possible role in wound healing. Br J Dermatol 1983;109:515–22.
DeLoecker W, Delport PH, Cheng N. Effect of pulsed electromagnetic fields on rat skin metabolism. Biochim Biophys Acta 1989;982:9–14.
Walleczek J. Electromagnetic field effects on the cells of the immune system: the role of calcium signalling. FASEB J 1992;6:3177–85.
Bassett CA. Low-energy pulsing electromagnetic fields modify biochemical processes. Bioessays 1987;6:36–42.
Weaver JC. Electroporation: a general phenomenon for manipulating cells and tissues. J Cell Biochem 1993;51:426–35.
Blank M. Membrane channel energetics: surface charge in protein interactions. In: Blank M, ed. Electricity and magnetism in biology and medicine. San Francisco: San Francisco Press, 1993;228–9.
Paradisi S, Donelli G, Santini MT, Straface E, Malorni W. A 50-Hz magnetic field induces structural and biophysical changes in membranes. Bioelectromagnetics 1993;14:247–55.
Miller DL. Miniature-probe measurements of electric fields and currents induced by a 60-Hz magnetic field in rat and human models. Bioelectromagnetics 1991;12:157–71.
Miller DL. Magnetically induced electric fields measured in rats and compared to a homogeneous rat model. In: Blank M, ed. Electricity and magnetism in biology and medicine. San Francisco: San Francisco Press, 1993;563–5.
Ottani V, Monti MG, Marocutti M,et al. Influence of pulsed electromagnetic fields on regenerating rat liver after partial hepatectomy. J Anat 1984;139:253–63.
Bourguignon GJ, Bourguignon LY. Electric stimulation of protein and DNA synthesis in human fibroblasts. FASEB J 1987;6638:398–402.
McLeod KJ, Lee RC, Ehrlich HP. Frequency dependence of electric field modulation of fibroblast protein synthesis. Science 1987;236:1465–9.
Guzelzsu N, Salkind AJ, Shen X, Patel U, Thaler S, Berg RA. Effect of electromagnetic stimulation with different waveforms on cultured chick tendon fibroblasts. Bioelectromagnetics 1994;15:115–31.
Author information
Authors and Affiliations
Additional information
Suture materials were supplied by Ethicon Limited, Birmingham, United Kingdom.
Read at the Third International Congress on New Technology and Advanced Techniques in Surgery, Luxembourg, June 13 to 17, 1995.
About this article
Cite this article
Menteş, B.B., Taşcilar, Ö., Tatlicioglu, E. et al. Influence of pulsed electromagnetic fields on healing of experimental colonic anastomosis. Dis Colon Rectum 39, 1031–1038 (1996). https://doi.org/10.1007/BF02054695
Issue Date:
DOI: https://doi.org/10.1007/BF02054695