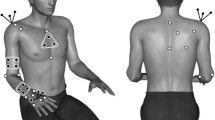
Abstract
A protocol for the evaluation of functional activities in subjects with Duchenne muscular dystrophy (DMD) was designed. The aim of our study was to define objective clinical criteria for the evaluation both of the clinical status of the patient and of the natural history of the illness itself. A protocol with such criteria is particularly necessary when testing the efficacy of treatment. 43 still-ambulant children with DMD between the ages of 3.10 yr and 10.4 yr were examined. Of this number 19 children were evaluated every 4 months over a period of 12 months; of these 14 formed part of a randomized double blind trial with L-carnitine (1.2–1.8 g/day) versus placebo.
Sommario
È stato elaborato un protocollo di valutazione delle attività funzionali in soggetti affetti da distrofia muscolare Duchenne (DMD).
Scopo del nostro studio è stato quello di definire dei criteri clinici oggettivi di valutazione sia dello stato di malattia del paziente, sia della evoluzione naturale della malattia stessa. Esigenza questa particolarmente sentita ogni qualvolta si debba verificare l'efficacia o meno di un qualsiasi intervento terapeutico.
Sono stati esaminati 43 bambini con DMD, ancora deambulanti, di età compresa tra i 3.10 ed i 10.4 anni. Di questi, 19 bambini, di cui 14 facevano parte di un trial randomizzato in doppio cieco con L-carnitina verso placebo (1.5–1.8 g/die), sono stati valutati ogni 4 mesi per un periodo di 12 mesi.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Adibi S.A.:Roles of branched-chain aminoacids in metabolic regulation J Lab Clin Med. 95: 475–484, 1980.
Archibald K.C., Vignos P.J.:A study of contractures in muscular dystrophy. Arch. Phys. Med Rehabil 40: 150–157, 1959.
Borum P.R., Broquist H.P., Roelofs R.I.:Muscle carnitine levels in neuromuscular diseases. J. Neurol Sci 34: 279–286, 1977.
Brooke M.H.:Functional evaluation and grading system for neuromuscular patients. “A clinician's view of neuromuscular diseases”, pp. 30–33, The Williams & Wilkins Company, 1977.
Brooke M.H., Griggs R.C., MENDELL J.R et al.:Clinical trial in Duchenne dystrophy. I. The Design of the protocol. Muscle Nerve 4: 186–197, 1981.
Carrier H.N., Berthillier G.Carnitine levels in normal children and adults and in patients with diseased muscle. Muscle Nerve 3: 326–334, 1980.
Cotellessa L., Marcucci F., et al.:Glass capillary determination of NT -Methylhistidine in urine. J. Chromatogr 221: 149–154, 1980.
Drachman D.B., Toyka K.V., Myer E.:Prednisone in Duchenne muscular dystrophy. Lancet 14: 1409–1416, 1974.
Hinsbergh V.W., Veerkamp J.H. et al.:Effect of L-carnitine on the oxidation of leucine and valine by rat skeletal muscle. Biochem Med 20; 115–124, 1978.
Hosking G.P., Cavanaugh N.P.C. et al.:Oral treatment of carnitine myopathy. Lancet 1: 853, 1977.
Karpati G., Carpenter S. et al.:The syndrome of systemic carnitine deficiency: clinical, morphologic, biochemical and pathophysiologic features. Neurology 25: 16–24, 1975.
Medical Research Council.Aids to the investigation of peripheral nerve injuries. Her Majesty's Stationery Office, London, England, 1976.
Mendell J.R., Wiechers D.O.:Lack of benefit of allopurinol in Duchenne dystrophy. Muscle Nerve, 2: 53–56, 1979.
Ringel S.P.,Stern L.Z. et al.:Randomized, double blind, placebo-controlled trial of ontosein (Superoxide dismutase) in Duchenne. Muscular Dystrophy Research: Advances and New Trends: 198–205, Excerpta Medica, 1980.
Roelofs R.I., Saavedra De Aranjo G. et al:Treatment of Duchenne's muscular dystrophy with penicillamine. Results of a double blind trial. Archs Neurol. 36: 266–268, 1979.
Roses M.S., Nicholson M.T. et al.:Evaluation and detection of Duchenne's and Becker's muscular dystrophy carriers by manual muscle testing. Neurology, 27: 20–25, 1977.
Swinyard C.A., Deaver G.G., Greenspan L.:Gradients of functional ability of importance in rehabilitation of patients with progressive muscular and neuromuscular diseases. Archs phys Med., 38: 574, 1957.
Thomson W.H.S., Smith I.:X-linked recessive (Duchenne) muscular dystrophy (DMD) and purine metabolism: effects of oral allopurinol and adenylate. Metabolism; 27: 151–163, 1978.
Zellweger H., Hanson J.W.:Psychometric studies in muscular dystrophy type IIIa (Duchenne). Dev Med Child Neurol. 9: 576–581, 1968.
Author information
Authors and Affiliations
Additional information
This work has been in part supported by a grant from the “Dino and Enzo Ferrari Foundation for Muscular Dystrophy Research”.
Rights and permissions
About this article
Cite this article
Cornelio, F., Dworzak, F., Morandi, L. et al. Functional evaluation of Duchenne muscular dystrophy: Proposal for a protocol. Ital J Neuro Sci 3, 323–330 (1982). https://doi.org/10.1007/BF02043581
Issue Date:
DOI: https://doi.org/10.1007/BF02043581