Abstract
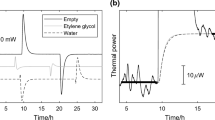
A calorimetric method for the study of solid-vapor interactions is described. In one calorimetric chamber a vapor evaporates; in a second calorimetric chamber the vapor is absorbed by a sample. The two chambers are connected by a tube and form part of a double twin microcalorimeter. As vapor is sorbed by the sample the vapor pressure in the sorption chamber will increase from a low value to near saturation. The flow rate of the vapor is governed by diffusion through the tube between the vessels. From the thermal power measured in the vaporization calorimeter it is possible to evaluate the sorption isotherm, and using information from both calorimeters the heat of sorption may be calculated as a function of equilibrium vapor pressure. By conducting experiments with different sized samples it is also possible to study the kinetics of the sorption process. The paper describes some recent improvements of the technique and gives examples of its use.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
I. Wadsö and L. Wadsö, Thermochim. Acta, 273 (1996) 277.
E. Calvet, C. R. Acad. Sci. Paris, 236 (1953) 377, 486.
R. W. Wood, L. D. Hansen and J. W. Crawford, United States Patent 5266492, Nov. 30, 1993.
L. Wadsö, J. Mater. Sci., 29 (1994) 2367.
Author information
Authors and Affiliations
Additional information
This work has been supported by the Swedish Natural Science Research Council (to IW), the Swedish Building Research Council (to LW) and the Lisshed Foundation, Stockholm (to LW).
Rights and permissions
About this article
Cite this article
Wadsö, I., Wadsö, L. A second generation twin double microcalorimeter. Journal of Thermal Analysis 49, 1045–1052 (1997). https://doi.org/10.1007/BF01996792
Issue Date:
DOI: https://doi.org/10.1007/BF01996792