Abstract
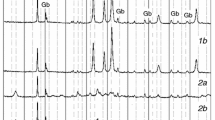
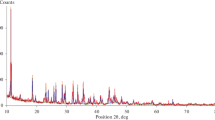
The reaction pathway of the thermal decomposition of synthetic brochantite, Cu4(OH)6SO4, to copper(II) oxide was investigated through the detailed kinetic characterization of the thermal dehydration and desulferation processes. The dehydration process was characterized by dividing into two overlapped kinetic processes with a possible formation of an intermediate compound, Cu4O(OH)4SO4. The dehydrated sample, Cu4O3SO4, was found first to be amorphous by means of XRD, followed by the crystallization to a mixture of CuO and CuO-CuSO4 at around 776 K. The specific surface area and the crystallization behaviour of the amorphous dehydrated compound depend largely on the dehydration conditions. The thermal desulferation process is influenced by the gross diffusion of the gaseous product SO3, which is governed by the advancement of the overall reaction interface from the top surface of the sample particle assemblage to the bottom.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
H. Tanaka and N. Koga, Thermochim. Acta, 133 (1988) 221.
H. Tanaka and N. Koga, J. Chem. Educ., 67 (1990) 612.
H. Tanaka, M. Kawano and N. Koga, Thermochim. Acta, 182 (1991) 281.
G. Pannetier, J. M. Bregeault, G. Djega-Marladassou and M. Grandon, Bull. Soc. Chim. France, (1963) 2616.
E. V. Margulis, Russ. J. Inorg. Chem., 7 (1962) 935.
P. Ramamurthy and E. A. Secco, Can. J. Chem., 48 (1970) 3510.
S. V. S. Prasad and V. S. Rao, J. Thermal Anal., 30 (1985) 603.
E. A. Secco, Can. J. Chem., 66 (1988) 329.
H. L. Friedman, J. Polym. Sci. C, 6 (1964) 183.
T. Ozawa, J. Thermal Anal., 31 (1986) 547.
N. Koga, Thermochim. Acta, 258 (1995) 145.
T. Ozawa, Bull. Chem. Soc. Jpn., 38 (1965) 1881; J. Thermal Anal., 2 (1970) 301; Thermochim. Acta, 100 (1986) 109.
N. Koga and J. Malek, Thermochim. Acta, 283 (1996) 69.
I. Uzunov, D. Klissurski and L. Teocharov, J. Thermal Anal., 44 (1995) 685.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Koga, N., Criado, J.M. & Tanaka, H. Reaction pathway and kinetics of the thermal decomposition of synthetic brochantite. Journal of Thermal Analysis 49, 1467–1475 (1997). https://doi.org/10.1007/BF01983705
Issue Date:
DOI: https://doi.org/10.1007/BF01983705