Summary
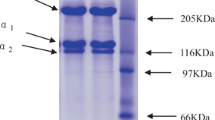
The outer shell of translucent keratin has been dissected from the claws of the lizard,Varanus gouldii. It is free of calcium and hydroxyproline, in contrast to the fibrous support, and contains proteins rich in glycine (28 residues %) and half-cystine (13%). These proteins have been obtained in soluble form by treatment with 2-mercaptoethanol in 8M urea at pH 11 followed by alkylation with iodoacetate to giveS-carboxymethyl kerateines. The three major components resolved by SDS polyacrylamide gel electrophoresis have been isolated by fractional precipitation with ammonium sulfate followed by chromatography on DEAE-cellulose or Sephadex. Two of the components, low in tryptophan content, appear to be homologous and are relatively homogeneous with respect to both size and charge whereas the third, a tryptophan-rich material, appears to contain about 20 different molecular species as judged by gel electrophoresis in urea at pH 8.9. The molecular weights of two of the isolated omponents (the tryptophan-rich and the major of the two tryptophanpoor components) are about 13000 as determined by equilibrium ultracentrifugation studies.
The major lizard claw proteins are therefore similar in size and glycine content to the proteins of avian beak and claw but differ in containing more cystine and less tyrosine. On the other hand, the reptilian proteins resemble the mammalian high-tyrosine proteins (Type II) in cystine content and overall amino acid composition, but differ in size with the lizard proteins being larger. It is suggested however that they are unlikely to be homologous.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Baden HP, Maderson PFA (1970) J Exp Zool 174:225–232
Baden H, Sviokla S, Roth I (1974) J Exp Zool 187:287–294
Bonner WM, Laskey RA (1974) Eur J Biochem 46:83–88
Bradbury JH, Chapman GV, King NLR (1966) Proc Third Int Wool Text Res Conf Paris I:359–364
Davis BJ (1964) Ann NY Acad Sci 121:404–427
Eastoe JE (1967) Composition of collagen and allied proteins. In: Ramachandran GN (ed) Treatise on collagen. Vol I. Academic Press Inc, London: pp 1–72
Fraser RDB, MacRea TP, Rogers GE (1972) Keratins, their composition structure and biosynthesis. Charles C. Thomas, Springfield, Ill
Frenkel MJ, Gillespie JM (1976) Aust J Biol Sci 29:467–479
Gillespie JM (1960) Aust J Biol Sci 13:81–103
Gillespie JM (1972) Comp Biochem Physiol 41B:723–734
Harrap BS, Woods EF (1964) Biochem J 92:8–18
Inglis AS, McMahon DTW, Roxburgh CM, Takayanagi H (1976) Anal Biochem 72:86–94
Laemmli UK (1970) Nature 227:680–685
Marshall RC, Gillespie JM (1976) Aust J Biol Sci 29:1–10
Marshall RC, Gillespie JM (1982) Comp Biochem Physiol (in press)
Marshall RC, Gillespie JM, Inglis AS, Frenkel MJ (1980) Proc 6th Int Wool Text Res Conf Pretoria II:147–158
Roark DE, Yphantis DA (1969) Ann NY Acad Sci 164:245–278
Rudall KM (1947) Biochim Biophys Acta 1:549–562
Sastry LVS, Ramachandran LK (1965) Biochim Biophys Acta 97:281–287
Sengel D, Dhouailly D, Mauger A (1980) Region-specific determination of epidermal differentiation in amniotes. In: Spearman RIC, Riley RA (eds) The skin of vertebrates. Linnean Society Symposium Series No. 9. Academic Press Inc, London, pp 185–197
Walker ID, Bridgen J (1976) Eur J Biochem 67:283–293
Weber K, Osborn M (1969) J Biol Chem 244:4406–4412
Wyld JA (1979) The characteristics of reptilian keratins: an analysis of the molecular events associated with the evolution of the vertebrate epidermis. Thesis, Univ Conn, Storrs
Wyld JA, Brush AH (1979) J Mol Evol 12:331–347
Yphantis DA (1964) Biochemistry 3:297–317
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Gillespie, J.M., Marshall, R.C. & Woods, E.F. A comparison of lizard claw keratin proteins with those of avian beak and claw. J Mol Evol 18, 121–129 (1982). https://doi.org/10.1007/BF01810831
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF01810831