Summary
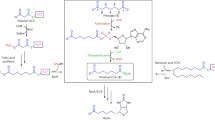
Imidazole catalysis of phenylalanyl transfer from phenylalanine adenylate anhydride to the hydroxyl groups of homopolyribonucleotides was investigated as a chemical model of the biochemical aminoacylation of tRNA. Imidazole catalyzed transfer of phenylalanine to poly (U) increases from pH 6.5 to 7.7 and decreases above pH 7.7. At pH 7.7 approximately 10% of the phenylalanyl residues are transferred to poly (U). At pH 7.1, transfer to poly (U) was five times as great as to poly (A) and transfer to a poly (A) poly (U) double helix was negligible. At pH 7.1 approximately 45 mole percent linkages to poly (U) were monomeric phenylalanine; the remainder of the linkages were peptides of phenylalanine. The number of linkages and their lability to base and neutral hydroxylamine indicates that phenylalanine and its peptides are attached as esters to the 2′ hydroxyl groups throughout poly (U) and the 2′ (3) hydroxyl groups at the terminus of poly (U). These results do model the contemporary process of aminoacyl transfer to tRNA and continue to suggest that a histidine residue is in the active site of aminoacyl-tRNA-synthetases.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Beljanski, M., Bourgarel, P. (1968). C.R.Acad.Sci., Ser.D. 266, 845
Beljanski, M., Fischer-Ferraro, D., Bourgarel, P. (1968). Eur.J.Biochem. 4, 184
Berg, P. (1958). J.Biol.Chem. 233, 608
Chambers, R.W. (1971). On the recognition of tRNA by its aminocacyl-tRNA ligase. In: Progress in nucleic acid chemistry and molecular biology, Vol. 11, J.N. Davidson, W.E. Cohn, eds., p. 489. New York: Academic Press
Gilbert, W. (1963). J.Mol.Biol. 6, 389
Gottikh, B.P., Kraevskii, A.A., Tsilevich, T.L., Rudzite, L.N. (1970). Izv.Akad.Nauk. S.S.S.R., Ser.Khim. p. 113
Gottikh, B.P., Kraevskii, A.A., Tarussova, N.B., Purygin, P.P., Tsilevich, T.L. (1970). Tetrahedron 26, 4419
Jencks, W.P. (1957). Biochim.Biophys.Acta 24, 227
Jencks, W.P., Carriuolo, J. (1959). J.Biol.Chem. 234, 1272
Krampitz, G., Fox, S.W. (1969). Proc.Nat.Acad.Sci. USA 62, 399
Lacey, J.C., Jr., White, W.E., Jr. (1972). Biochem.Biophys.Res.Comm. 47, 565
Lipmann, F., Tuttle, L.C. (1945). J.Biol.Chem. 159, 21
Lohrmann, R., Orgel, L.E. (1973). Nature 244, 418
Michelson, A.M., Massoulie, J., Guschlbauer, W. (1967). Synthetic polynucletides. In: Progress in nucleic acid research and molecular biology, Vol. 6, J.N. Davidson, W.E. Cohn, eds., p. 83. New York: Academic Press
Nakashima, T., Lacey, J.C., Jr., Jungck, J., Fox, S.W. (1970). Naturwiss. 57, 67
Nakashima, T., Fox, S.W. (1972). Proc.Nat.Acad.Sci. USA 69, 106
Rohlfing, D.L., Fox, S.W. (1969). Catalytic activities of thermal polyanhydro-α-amino acids. In: Advances in catalysis, Vol. 20, D.D. Eley, H.Pines, P.B. Weisz, eds., p. 373. New York: Academic Press
Tsilevich, T.L., Kraevskii, A.A., Gottikh, B.P. (1971). Izv.Acad.Nauk. S.S.S.R., Ser.Khim. p. 811
Weber, A.L., Fox, S.W. (1973). Biochim.Biophys.Acta 319, 174
Weber, A.L., Lacey, J.C., Jr. (1974). Biochim.Biophys.Acta 349, 226
White, W.E., Jr., Lacey, J.C., Jr., Weber, A.L. (1973). Biochim. Biophys.Res.Comm. 51, 283
Yang, J.T., Samejima, T. (1969). Optical rotary dispersion and circular dichroism of nucleic acids. In: Progess in nucleic acid research and molecular biology, Vol. 9, J.N. Davidson, W.E. Cohn, eds., p. 223. New York: Academic Press
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Weber, A.L., Lacey, J.C. Aminoacyl transfer from an adenylate anhydride to polyribonucleotides. J Mol Evol 6, 309–320 (1975). https://doi.org/10.1007/BF01794637
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF01794637