Summary
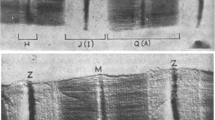
The localization and elasticity of connectin (titin) filaments in skinned fibres of frog skeletal muscle were examined for changes in the localization of connectin and in resting tension during partial depolymerization of thick filaments with a relaxing solution containing increased KCl concentrations. Immunoelectron microscopic studies revealed that deposites of antibodies against connectin at a sarcomere length of 3.0 μm remained at about 0.8 μm from the M-line, until the thick filament was depolymerized to the length of approximately 0.4 μm. On further depolymerization, the bound antibodies were found to move towards the Z-line and, on complete depolymerization, were observed to be within 0.3 μm of the Z-line; a marked decrease in resting tension accompanied this further depolymerization. These results suggest that connectin filament starts from the Z-line, extends to the M-line, and contributes to resting tension. After partial depolymerization of thick filaments, the distances between the anti-connectin deposits and the Z-line and between anti-connectin deposits and the M-line increased with sarcomere length, suggesting that connectin filaments are elastic along their entire length.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Fairbanks, G., Steck, T. L. &Wallach, D. F. H. (1971) Electrophoretic analysis of the major polypeptide of the human erythrocyte membrane.Biochemistry 10, 1607–17.
Funatsu, T., Higuchi, H. &Ishiwata, S. (1990) Elastic filaments in skeletal muscle revealed by selective removal of thin filaments with plasma gelsolin.J. Cell Biol. 110, 53–62.
Fürst, D. O., Osborn, M., Nave, R. &Weber, K. (1988) The organization of titin filaments in the half-sarcomere revealed by monoclonal antibodies in immunoelectron microscopy: a map of ten non-repetitive epitopes starting at the Z-line extends close to the M-line.J. Cell Biol. 106, 1563–72.
Fürst, D. O., Nave, R., Osborn, M. &Weber, K. (1989) Repetitive titin epitopes with known A-band striations also identified by major myosin-associated proteins.J. Cell Sci. 94, 119–25.
Higuchi, H. (1987) Lattice swelling with the selective digestion of elastic components in single skinned fibres of frog muscle.Biophys. J. 52, 29–32.
Higuchi, H. &Umazume, Y. (1985) Localization of the parallel elastic components in frog skinned muscle fibres studied by the depolymerization of the A- and I-bands.Biophys. J. 48, 137–47.
Higuchi, H. &Umazume, Y. (1986) Lattice shrinkage with increasing resting tension in stretched, single skinned fibres of frog muscle.Biophys. J. 50, 385–9.
Higuchi, H., Yoshioka, T. &Maruyama, K. (1988) Positioning of actin filaments and tension generation in skinned muscle fibres released after stretch beyond overlap of the actin and myosin filaments.J. Muscle Res. Cell Motil. 9, 491–8.
Horiuti, K., Higuchi, H., Umazume, Y., Konishi, M., Okazaki, O. &Kurihara, S. (1988) Mechanism of action of 2,3-butane-dione 2-monoxime on contraction of frog skeletal muscle fibres.J. Muscle Res. Cell Motil. 9, 156–64.
Horowits, R., Maruyama, K. &Podolsky, R. J. (1989) Elastic behaviour of connectin filaments during the thick filament movements in activated skeletal muscle.J. Cell Biol. 109, 2169–76.
Itoh, Y., Suzuki, T., Kimura, S., Ohashi, K., Higuchi, H., Sawada, H., Shimizu, T., Sibata, M. &Maruyama, K. (1988) Extensible and less-extensible domains of connectin filaments in stretched vertebrate skeletal muscle sarcomeres as detected by immunofluorescence and immunoelectron microscopy using monoclonal antibodies.J. Biochem. 104, 504–8.
Labeit, S., Barlow, D. P., Gautel, M., Gibson, T., Holt, J., Hsieh, C. L., Francke, U., Leonard, K., Wardale, J., Whiting, A. &Trinick, J. (1990) A regular pattern of two types of 100-residue motif in the sequence of titin.Nature 345, 273–6.
Lombardi, V. &Piazzesi, G. (1990) The contractile response during steady lengthening of stimulated frog muscle fibres.J. Physiol. 431, 141–71.
Maruyama, K. (1986) Connectin, an elastic filamentous protein of striated muscle.Int. Rev. Cytol. 104, 81–114.
Maruyama, K., Kimura, S., Yoshitomi, H., Sawada, H. &Kikuchi, M. (1984) Molecular size and shape ofβ-connectin, an elastic protein of striated muscle.J. Biochem. 95, 123–33.
Maruyama, K., Yoshioka, T., Higuchi, H., Ohashi, K., Kimura, S. &Natori, R. (1985) Connectin filaments link thick filaments and Z-lines in frog skeletal muscle as revealed by immunoelectron microscopy.J. Cell Biol. 101, 2167–72.
Masaki, T. &Takaiti, O. (1974) M-protein.J. Biochem. 75, 367–80.
Natori, R. (1954) The property and contraction process of isolated myofibrils.Jikeikai Med. J. 1, 119–26.
Nave, R., Fürst, D. O. &Weber, K. (1989) Visualization of the polarity of isolated titin molecules: a simple globular head on a long thin rod as the M-band anchoring domain?J. Cell Biol. 109, 2177–87.
Offer, G., Moos, C. &Starr, R. (1973) A new protein of the thick filaments of vertebrate skeletal myofibrils.J. Mol. Biol. 74, 653–76.
Roos, K. P. &Brady, A. J., (1989) Stiffness and shortening changes in myofilament-extracted rat cardiac myocytes.Am. J. Physiol. 256, H539–51.
Wang, K. (1985) Sarcomere-associated cytoskeletal lattices in striated muscle: reviews and hypothesis. InCell and Muscle Motility (edited by Shay, J. W.) Vol. 6, pp. 315–69. New York: Plenum Publishing.
Schönberg, M. (1980) Geometrical factors influencing muscle fibre development. II. Radial forces.Biophys. J. 30, 69–78.
Whiting, A., Wardale, J. &Trinick, J. (1989) Does titin regulate the length of muscle thick filaments?J. Mol Biol. 205, 263–8.
Yoshioka, T., Higuchi, H., Kimura, S., Ohashi, K., Umazume, Y. &Maruyama, K. (1986) Effect of mild trypsin treatment on the passive tension generation and connectin splitting in stretch skinned fibres from frog skeletal muscle.Biomed. Res. 7, 181–6.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Higuchi, H., Suzuki, T., Kimura, S. et al. Localization and elasticity of connectin (titin) filaments in skinned frog muscle fibres subjected to partial depolymerization of thick filaments. J Muscle Res Cell Motil 13, 285–294 (1992). https://doi.org/10.1007/BF01766456
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF01766456