Summary
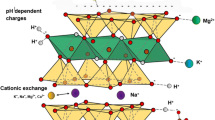
Published data on adsorption and condensation of amino acids, purine and pyrimidine bases, sugars, nucleosides, and nucleotides are analyzed in connection with Bernal's hypothesis that clays and other minerals may have provided the most likely surface for adsorption and condensation of these molecules in prebiotic times. Using surface concentration and reaction rate as the main criteria for the feasibility of condensation reactions, four types of prebiotic environments were analyzed: (1) an ocean-sediment system, (2) a dehydrated lagoon bed produced by evaporation, (3) the surface of a frozen sediment, and (4) a fluctuating system where hydration (rainstorms, tidal variations, flooding) and dehydration (evaporation) take place in a cyclic manner. With the possible exception of nucleotides, low adsorption of organomonomers on sediment surfaces of a prebiotic ocean (pH 8) is expected, and significant condensation is considered unlikely. In dehydrated and frozen systems, high surface concentrations are probable and condensation is more likely. In fluctuating environments, condensation rates will be enhanced and the size distribution of the oligomers formed during dehydration may be influenced by a “redistribution mechanism” in which adsorbed oligomers and monomers are desorbed and redistributed on the solid surface during the next hydration-dehydration cycle.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
Anderson, D.M. (1967). J.Colloid Interface Sci. 25, 174
Anderson, D.M., Banin, A. (1975). Origins of Life 6, 23
Bernal, J.D. (1951). The physical basis of life. London: Routledge and Kegan Paul
Blumstein, A. (1965). J.Polymer Sci. A3, 2653
Bonner, W.A., Flores, J. (1973). Currents Mod.Biol. 5, 103
Bonner, W.A., Kavasmaneck, P.R., Martin, F.S. (1974). Science 186, 143
Brack, A., Orgel, L.E. (1975). Nature 256, 383
Burton, F.G., Neuman, M.W., Neuman, W.F. (1969). Currents Mod.Biol. 3, 20
Burton, F.G., Lohrmann, R., Orgel, L.E. (1974). J.Mol.Evol. 3, 141
Burton, F.G., Neuman, W.F. (1974). Currents in Modern Biol. 4, 47
Cairns-Smith, A.G. (1971). The life puzzle. Toronto and Buffalo: University of Toronto Press
Calvin, M. (1969). Chemical evolution. New York and Oxford: Oxford University Press
Cloos, P., Calicis, B., Fripiat, J.J., Makay, K. (1966). Proc.Int.Clay Conf.Jerusalem 1, 223
Davis, G.A., Worrall, W.E. (1971). Trans.Brit.Ceram.Soc. 70, 71
Degens, E.T., Matheja, J. (1970). In: Prebiotic and biochemical evolution, A.P. Kimball, J. Oro, eds., p. 39. Amsterdam: North Holland
Dose, K. (1975). BioSystems 6, 224
Flores, J.J., Lecki, J.O. (1973). Nature 244, 435
Flores, J.J., Bonner, W.A. (1974). J.Mol.Evol. 3, 49
Fox, S.W. (1969). Encyclopedia Polymer Sci.Technol. 9, 284
Fripiat, J.J. (1972). Int.Clay Conf.Madrid 1972, 2, 537
Fripiat, J.J., Cruz-Cumplido, J. (1974). Ann.Rev.Earth Planet Sci. 2, 239
Fuller, W.D., Sanchez, R.A., Orgel, L.E. (1972). J.Mol.Biol. 67, 25
Gabel, N.W. (1976). In: Progress in molecular and subcellular biology, Vol.5:1. Berlin, Heidelberg, New York: Springer
Good, W. (1973). J.Theoret.Biol. 39, 249
Greenland, D.J. (1956). J.Soil Sci. 7, 329
Greenland, D.J., Laby, R.H., Quirk, J.P. (1965a). Trans.Faraday Soc. 61, 2013
Greenland, D.J., Laby, R.H., Quirk, J.P. (1965b). Trans.Faraday Soc. 61, 2024
Grim, R.E. (1968). Clay mineralogy. New York: MyGraw-Hill
Hanafusa, H., Akabori, S. (1959). Bull.Chem.Soc.Japan 32, 626
Ibanez, J.D., Kimball, A.P., Oro, J. (1970). Science 171, 444
Jackson, T.A. (1971). Experimentia (Basel) 27, 242
Jepson, W.B., Williams, J.F. (1972). Clay Minerals 9, 275
Katchalsky, A. (1973). Naturwissenschaften 60, 215
Kenyon, D.H., Steinman, G. (1969). Biochemical predestination, p. 214. New York: McGraw-Hill
Lahav, N. (1975). J.Mol.Evol. 5, 243
Lahav, N., Anderson, D.M. (1973). Israel J.Chem. 11, 549
Lai, T.M., Mortland, M.M. (1968). Soil Sci.Soc.Am.Proc. 32, 56
Lailach, G.E., Thompson, T.D., Brindley, G.W. (1968). Clays and Clay Minerals 16, 285
Lailach, G.E., Brindley, G.W. (1969). Clays and Clay Minerals 17, 95
McCullough, J.J., Lemmon, R.M. (1974). J.Mol.Evol. 3, 57
Miller, S.L. (1953). Science 117, 528
Miller, S.L. (1974). In: Cosmochemical evolution and the origins of life, Vol. I:139. Dordrecht-Holland: Reidel
Miller, S.L., Orgel, L.E. (1974). The origins of life on earth. Englewood Cliffs, New Jersey: Prentice-Hall
Misfud, A., Fornes, V., Rausell-Colom, J.A. (1970). Reunion Hispano-Belga De Minerales De Argilla, 121
Mitra, S.P., Misra, S.G., Panda, V. (1957). Proc.Natl.Acad.Sci.India 26A, 72
Mortland, M.M. (1970). Advan.Agron. 22, 75
Neuman, M.W., Neuman, W.F., Burton, F.G. (1969). Currents Mod.Biol. 3, 69
Nissenbaum, A., Kenyon, D.H., Oro, J. (1967). J.Mol.Evol. 6, 253
Odom, D.G., Brady, J.T. (1975). J.Mol.Evol. 6, 199
Orgel, L.E. (1972). Israel J.Chem. 10, 287
Orgel, L.E., Lohrmann, R. (1974). Accounts Chem.Res. 7, 368
Oro, J., Stephen-Sherwood, E. (1973). In: Cosmochemical evolution and the origins of life, Vol. I:159. Dordrecht-Holland/Boston, USA: Reidel
Paecht,Horowitz, M., Berges, J., Katchalsky, A. (1970). Nature 228, 636
Park, W.K., Hochstim, A.R., Ponnamperuma, C. (1975). Origins of Life 6, 99
Sagan, C., Khare, B.N. (1971). Science 173, 417
Sawai, H., Lohrmann, R., Orgel, L.E. (1975). J.Mol.Evol. 6, 165
Sawai, H., Orgel, L.E. (1975). J.Mol.Evol. 6, 185
Schwartz, A.W., Van Der Veen, M., Bisseling, T., Chittenden, G.J.F. (1973). BioSystems 5, 119
Sillen, L.G. (1967). Science 156, 1139
Steinman, G. (1966). Science 154, 1344
Steinman, G.D., Kenyon, D.H., Calvin, M. (1966). Biochim.Biophys.Acta 124,339
Stephen-Sherwood, E., Odom, D.G., Oro, J. (1974). J.Mol.Evol. 3, 323
Theng, B.K.G. (1973). The chemistry of clay-organic reactions. New York and Tokyo: Wiley
Thompson, T.D., Brindley, G.W. (1969). Am.Mineral. 54, 858
Warden, J.T., McCullough, J.J., Lemmon, R., Calvin, M. (1974). J.Mol.Evol. 4, 189
Wedepohl, K.H. (1969). In: Handbook of geochemistry, K.H. Wedepohl, ed., Vol. 1, p. 227. Berlin, Heidelberg, New York: Springer
Author information
Authors and Affiliations
Additional information
On leave from the Faculty of Agriculture, Rehovot, The Hebrew University of Jerusalem, Israel.
Rights and permissions
About this article
Cite this article
Lahav, N., Chang, S. The possible role of solid surface area in condensation reactions during chemical evolution: Reevaluation. J Mol Evol 8, 357–380 (1976). https://doi.org/10.1007/BF01739261
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF01739261