Summary
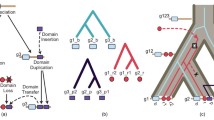
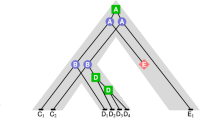
Using computer programs that analyze the evolutionary history and probability of relationship of protein sequences, we have investigated the gene duplication events that led to the present configuration of immunoglobulin C regions, with particular attention to the origins of the homology regions (domains) of the heavy chains. We conclude that all of the sequenced heavy chains share a common ancestor consisting of four domains and that the two shorter heavy chains, alpha and gamma, have independently lost most of the second domain. These conclusions allow us to align corresponding regions of these sequences for the purpose of deriving evolutionary trees. Three independent internal gene duplications are postulated to explain the observed pattern of relationships among the four domains: first a duplication of the ancestral single domain C region, followed by independent duplications of the resulting first and last domains. In these studies there was no evidence of crossing-over and recombination between ancestral chains of different classes; however, certain types of recombinations would not be detectable from the available sequence data.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Barker, W.C., Dayhoff, M.O. (1972). Detecting distant relationships: computer methods and results. In: Atlas of protein sequence and structure, M.O. Dayhoff, ed., Vol.5, pp. 101–110. Washington, D.C.: National Biomedical Research Foundation
Barker, W.C., Dayhoff, M.O. (1976). Immunoglobulins and related proteins. In: Atlas of protein sequence and structure, M.O. Dayhoff, ed., Vol. 5, Suppl. 2, pp. 165–190. Washington, D.C.: National Biomedical Research Foundation
Barker, W.C., Dayhoff, M.O. (1979a). Biophys. J.25, 158a
Barker, W.C., Dayhoff, M.O. (1979b). Unpublished observations
Barker, W.C., Ketcham, L.K., Dayhoff, M.O. (1979). Immunoglobulins. In: Atlas of protein sequence and structure, M.O. Dayhoff ed., Vol. 5, Suppl. 3, pp. 197–227. Washington, D.C.: National Biomedical Research Foundation
Bennich, H.H., Johansson, S.G.O., von Bahr-Lindstrom, H. (1978). The discovery of immunoglobulin E and the determination of its chemical structure. In: Immediate Hypersensitivity: modern concepts and developments, M.K. Bach, ed., pp. 1–36. New York: Marcel Dekker
Cunningham, B.A., Rutishauser, U., Gall, W.E., Gottlieb, P.D., Waxdal, M.J., Edelman, G.M. (1970). Biochemistry9, 3161–3170
Dayhoff, M.O., ed. (1979a). Atlas of protein sequence and structure, Vol.5, Suppl.3, p. 375. Washington, D.C.: National Biomedical Research Foundation
Dayhoff, M.O. (1979b). Survey of new data and computer methods of analysis. In: Atlas of protein sequence and structure, M.O. Dayhoff, ed., Vol.5, Suppl.3, pp. 1–8. Washington, D.C.: National Biomedical Research Foundation
Dayhoff, M.O., McLaughlin, P.J., Barker, W.C., Hunt, L.T. (1975). Naturwissenschaften62, 154–161
Dayhoff, M.O., Schwartz, R.M., Orcutt, B.C. (1979). A model of evolutionary change in proteins. In: Atlas of protein sequence and structure, M.O. Dayhoff, ed., Vol.5, Suppl. 3, pp. 345–352. Washington, D.C.: National Biomedical Research Foundation
Honjo, T., Kataoka, T. (1978). Proc. Natl. Acad. Sci. U.S.A.75, 2140–2144
Huisman, T.H.J., Wrightstone, R.N., Wilson, J.B., Schroeder, W.A., Kendall, A.G. (1972). Arch. Biochem. Biophys.153, 850–853
Kratzin, H., Altevogt, P., Ruban, E., Kortt, A., Staroscik, K., Hilschmann, N. (1975). Hoppe-Seyler's Z. Physiol. Chem.356, 1337–1342
Liu, Y.-S.V., Low, T.L.K., Infante, A., Putnam, F.W. (1976). Science193, 1017–1020
Low, T.L.K., Lui, Y.-S.V., Putnam, F.W. (1976). Science191, 390–392
Margoliash, E., Fitch, W.M. (1967). Science155, 279–284
Natvig, J.B., Kunkel, H.G. (1974). J. Immunol.112, 1277–1284
Needleman, S.B., Wunsch, C.D. (1970). J. Mol. Biol.48, 443–453
Orcutt, B.C., Dayhoff, M.O. (1979). NBR Report No. 08710-790606. Washington, D.C.: National Biomedical Research Foundation
Pink, J.R.L., Buttery, S.H., DeVries, G.M., Milstein, C. (1970). Biochem. J.117, 33–47
Poljak, R.J., Amzel, L.M., Phizackerley, R.P. (1976). Prog. Biophys. Mol. Biol.31, 67–93
Ponstingl, H., Hilschmann, N. (1972). Hoppe-Seyler's Z. Physiol. Chem.353, 1369–1372
Putnam, F.W., Florent, G., Paul, C., Shinoda, T., Shimizu, A. (1973). Science182, 287–291
Rutishauser, U., Cunningham, B.A., Bennett, C., Konigsberg, W.H., Edelman, G.M. (1970). Biochemistry9, 3171–3181
Tsuzukida, Y., Wang, C.-C., Putnam, F.W. (1979). Proc. Natl. Acad. Sci. U.S.A.76, 1104–1108
Watanabe, S., Barnikol, H.U., Horn, J., Bertram, J., Hilschmann, N. (1973). Hoppe-Seyler's Z. Physiol. Chem.354, 1505–1509
Werner, B.G., Steinberg, A.G. (1974). Immunogenetics3, 254–271
Wolfenstein-Todel, C., Frangione, B., Prelli, F., Franklin, E.C. (1976). Biochem. Biophys. Res. Commun.71, 907–914
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Barker, W.C., Ketcham, L.K. & Dayhoff, M.O. Origins of immunoglobulin heavy chain domains. J Mol Evol 15, 113–127 (1980). https://doi.org/10.1007/BF01732665
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF01732665