Summary
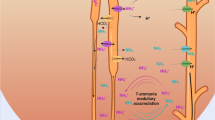
The proximal tubule cell is the major site of renal ammoniagenesis. Glutamine is the major substrate. Deamidation by mitochondrial glutaminase yields glutamate− and NH +4 (not NH3, as traditionally taught). A second NH +4 ion is obtained by deamination of glutamate− to 2-oxo-glutarate2−. NH +4 preferentially enters the tubule lumen primarily, but probably not exclusively, by non-ionic diffusion of NH3. For each NH3 formed in the cell one H+ ion is left behind. H+ and NH3 are secreted on separate routes, but recombine in the lumen to NH +4 and reach the final urine in this form. This processper se does not net-remove H+ from the organism. For this purpose, the anionic products of ammoniagenesis (2-oxo-glutarate2− and others) have to be converted into neutral compounds (CO2, glucose). This metabolism again takes place usually in the tubule cell. For each negative charge one HCO −3 is formed which enters the peritubular blood. Luminal γ-glutamyl transferase-mediated ammoniagenesis contributes to NH +4 accumulation in the proximal tubule to a small extent. The endproximal NH +4 delivery exceeds the filtered load by a factor of 9. Only 1/3 of it reaches the distal convoluted tubule mainly because NH +4 as such is reabsorbed from the thick ascending limb of Henle's loop by secondary active transport or electrodiffusion. Both processes are energized by the active Na+ transport in this segment. Thereby NH3↔NH +4 is accumulated in the medullary interstitium, which establishes the chemical gradient for non-ionic diffusion of NH3 into the lumen of the collecting ducts. This is favoured by the acidic disequilibrium pH in the lumen of this segment. Secretion of NH3↔NH +4 , probably by non-ionic diffusion, also into the descending limb of the loop is hypothesized to maintain (together with the NH +4 reabsorption in the thick ascending limb) the high interstitial NH3↔NH +4 concentration increasing towards to inner medulla. Thus, the principle of counter current multiplication seems to be involved also in NH +4 excretion.
Zusammenfassung
Hauptort der renalen NH3↔NH +4 -Bildung ist die proximale Tubuluszelle. Glutamin ist das wesentliche Substrat dafür. Die Desamidierung durch die mitochondriale Glutaminase ergibt Glutamat− und NH +4 (und nicht NH3, wie oft behauptet). Ein zweites NH +4 -Ion wird aus der Desaminierung von Glutamat− zu 2-Oxo-Glutarat2− gewonnen. NH +4 gelangt in das Tubuluslumen großteils (aber nicht ausschließlich) in der Form von NH3 (nicht-ionische Diffusion). Für jedes sezernierte NH3 bleibt ein H+-Ion in der Zelle zurück, das separat ins Lumen transportiert wird. Im Lumen wird aus beiden wieder NH +4 gebildet, das dann im Urin ausgeschieden wird. Dieser Prozeßper se entfernt keine H+-Ionen aus dem Körper. Zu diesem Zwecke müssen erst die anionischen Metaboliten, die bei der Ammoniagenese entstehen (2-Oxo-Glutarat2− u.a.), in neutrale Stoffe umgewandelt werden (CO2, Glukose). Dies geschieht ebenfalls, wenn auch nicht notwendigerweise, in der Tubuluszelle. Für jede negative Ladung des Substrats entsteht dabei ein HCO −3 -Ion, das die Zelle auf der Blutseite verläßt. Eine luminale NH +4 -Bildung, katalysiert durch γ-Glutamyltransferase, trägt in geringem Ausmaß zur NH +4 -Anhäufung im Lumen bei. Am Ende des proximalen Konvoluts findet sich 9mal mehr NH3↔NH +4 als im Glomerulusfiltrat. Nur ein Drittel davon erreicht das distale Konvolut, vor allem weil NH +4 als solches im dicken, aufsteigenden Teil der Henleschen Schleife durch Co-Transport oder Elektrodiffusion resorbiert wird. Beide Prozesse erhalten ihre Energie durch den aktiven Na+-Transport in diesem Nephronteil. Dadurch wird NH3↔NH +4 u.a. im medullären Interstitium akkumuliert, so daß ein chemischer Gradient für die nicht-ionische Diffusion von NH3 ins Lumen des Sammelrohrs aufgebaut wird. Begünstigt wird dies auch durch den sauren Disäquilibriums-pH-Wert im Lumen dieses Segments. Es wurde vorgeschlagen, daß NH3↔NH +4 , wohl via nicht-ionische Diffusion, auch in den absteigenden Teil der Henleschen Schleife sezerniert wird, um, zusammen mit der NH +4 -Resorption im aufsteigenden Schleifenteil, die hohe interstitielle NH3↔NH +4 -Konzentration, die markwärts zunimmt, aufrechtzuerhalten. Das Prinzip der Gegenstrom-Multiplikation scheint daher auch bei der NH +4 -Ausscheidung eine wesentliche Rolle zu spielen.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Atkinson DE, Bourke E (1984) The role of ureagenesis in pH homeostasis. TIBS 7:297–298
Atkinson DE, Camien MN (1982) The role of urea synthesis in the removal of metabolic bicarbonate and the regulation of blood pH. In: Current topics in cellular regulation, Vol 21. Academic Press, New York, p 261–302
Bachmann C (1984) Treatment of congenital hyperammonemias. Enzyme 32:56–64
Balagura S, Stone WJ (1967) Renal tubular secretion of alpha ketoglutarate in dog. Am J Physiol 212:1319–1326
Buerkert J, Martin D, Trigg D (1982) Ammonium handling by superficial and juxtamedullary nephrons in the rat. J Clin Invest 70:1–12
Burg MB, Green N (1973) Function of the thick ascending limb of Henle's loop. Am J Physiol 224:659–668
Cahill GF Jr (1970) Starvation in man. New Engl J Med 282:668–675
Cohen J, Wittmann E (1963) Renal utilization and excretion of alpha ketoglutarate in dog: effect of alkalosis. Am J Physiol 204:795–811
Curthoys NP, Kuhlenschmidt T (1975) Phosphate-independent glutaminase from rat kidney. Partial purification and idendity with gamma-glutamyltranspeptidase. J Biol Chem 250:2099–2105
Curthoys NP, Shapiro RA, Haser WG (1984) Enzymes of renal glutamine metabolism. In: (Ed.) Häusinger D, and Sies H, Glutamine metabolism in mammalian tissues. Springer, Berlin Heidelberg New York, p 16–31
Glabman S, Klose RM, Giebisch G (1963) Micropuncture study of ammonia excretion in the rat. Am J Physiol 205:127–132
Goldstein L (1966) Relation of glutamate to ammonia production in the rat kidney Am J Physiol 210:661–666
Goldstein L (1976) Ammonia production and excretion in the mammalian kidney. Int Rev Physiol 11:284–315
Good DW, Burg MB (1984) Ammonia production by individual segments of the rat nephron. J Clin Invest 73:602–610
Good DW, Knepper MA (1985) Ammonia transport in the mammalian kidney. Am J Physiol 248:F459-F471
Good DW, Knepper MA, Burg MB (1984) Ammonia and bicarbonate transport by thick ascending limb of rat kidney. Am J Physiol 247:F35-F44
Goodman AD, Fuizy RE, Cahill GF (1966) Renal gluconeogenesis in acidosis, alkalosis and potassium deficiency: Its possible role in regulation of renal ammonia production. J Clin Invest 45:612–619
Greger R (1981) Chloride reabsorption in the rabbit cortical thick ascending limb of the loop of Henle — a sodium dependent process. Pflügers Arch 390:38–43
Halperin ML, Cheema-Dhadli S, Chen CB, West ML, Jungas RL (1986) Is urea formation regulated primarily by acid-base balance? J Clin Chem Clin Biochem 24:267
Halperin ML, Goldstein MB, Stinebaugh BJ, Jungas RL (1985) Biochemistry and Physiology of Ammonium Excretion. In: (Ed.) Seldin DW, Giebisch G, The Kidney: Physiology and Pathophysiology Raven Press, New York, p 1471–1490
Halperin ML, Jungas RL (1983) Metabolic production and renal disposal of hydrogen ions. Kidney Int 24:709–713
Häussinger D, Gerok W, Sies H (1984) Hepatic role in pH regulation: role of the intercellular glutamine cycle. TIBS 7:300–302
Jenkins AD, Dousa TP, Smith LH (1985) Transport of citrate across renal brush border membrane: effects of dietary acid and alkali loading. Am J Physiol 249:F590-F595
Katunuma N, Tomino I, Nishino H (1966) Glutaminase isozymes in rat kidney. Biochem Biophys Res Comm 22:321–328
Knepper MA, Good DW, Burg MB (1984) Mechanism of ammonia secretion by cortical collecting ducts of rabbits. Am J Physiol 247:F729-F738
Lang F, Silbernagl S, Quehenberger P (1980) Hydrogen ion pump bicarbonate leak in the proximal convoluted tubule. In: (Ed.) Schulz I, Sachs G, Forte JG, Ullrich KJ, Hydrogen Ion Transport in Epithelia Elsevier, Amsterdam, p. 303–314
Maren TH, Wadsworth BC, Yale EK, Alonso LG (1954) Carbonic anhydrase inhibition. III. Effects of Diamox on electrolyte metabolism. Bull Johns Hopk Hosp 95: 277–321
McGilvery RW (1979) Biochemistry: A functional approach. Saunders, Philadelphia, p 764–767
Nagami GT, Kurokawa K (1985) Regulation of ammonia production by mouse proximal tubules perfused in vitro. Effect of luminal perfusion. J Clin Invest 75:844–849
Oliver J, Bourke E (1975) Adaptations in urea ammonium excretion in metabolic acidosis in the rat: a reinterpretation. Clin Sci Mol Med 48:515–520
Owen EE, Robinson RR (1963) Amino acid extraction and ammonia metabolism by the human kidney during the prolonged administration of ammonium chloride. J Clin Invest 42:263–276
Pitts RF (1972) Control of renal production of ammonia. Kidney Int 1:297–305
Pitts RF, DeHaas J, Klein J (1963) Relation of renal amino and amide nitrogen extraction to ammonia production. Am J Physiol 204:187–191
Ross B, Lowry M (1981) Recent developments in renal handling of glutamine and ammonia. In: (Ed.) Greger R, Lang F, Silbernagl S, Renal transport of organic substances. Springer, Berlin Heidelberg New York, p 78–92
Ross BD (1976) Effect of inhibition of gluconeogenesis on ammonia production in the perfused rat kidney. Clin Sci Mol Med 50:493–498
Sajo IM, Goldstein MB, Sonnenberg H, Stinebaugh BJ, Wilson DR, Halperin ML (1981) Sites of ammonia addition to tubular fluid in rats with chronic metabolic acidosis. Kidney Int 20:353–358
Scheller D, Silbernagl S (1986) Do HCl or NH4Cl induce metabolic acidosis by different mechanisms? J Clin Chem Clin Biochem 24:267
Shapiro R, Curthoys P (1981) Differential effect of AT-125 on rat renal glutaminase activities. FEBS LETT 133:131–134
Silbernagl S (1980) Tubular reabsorption of L-glutamine studied by free-flow micropuncture and microperfusion of rat kidney. Int J Biochem 12:9–16
Silbernagl S (1986) Ammoniagenesis catalyzed by hippurate-activated gamma-glutamyltranferase in the lumen of the proximal tubule. A microperfusion study in rat kidney in vivo. Pflügers Arch: in press
Silbernagl S, Curthoys NP, Shapiro R (1982) The role of renal tubular brushborder enzymes in metabolism and absorption of glutathione and cysteinyl-glycine. In: Biochemistry of Kidney Functions. (Ed) Morel F, INSERM Symposium No. 21. Elsevier Biomedical Press, Amsterdam New York Oxford, p 303–314
Silbernagl S, Pfaller W, Heinle H, Wendel A (1978) Topology and function of renal gamma-glutamyl transpeptidase. In: (Ed.) Sies H, Wendel A, Functions of glutathione in liver and kidney. Springer, Berlin Heidelberg New York, p 60–69
Silverman M, Vinay P, Shinobu L, Gougoux A, Lemieux G (1981) Luminal and antiluminal transport of glutamine in dog kidney: Effect of metabolic acidosis. Kidney Int 20:359–365
Simon E, Martin D, Buerkert J (1985) Contribution of individual nephron segments to ammonium handling in chronic metabolic acidosis in the rat. Evidence for ammonia disequilibrium in the renal cortex. J Clin Invest 76:855–864
Simpson DP (1983) Citrate excretion: a window on renal metabolism. AM J Physiol 13:F223-F234
Stern L, Backman KA, Hayslett JP (1985) Effect of cortical-medullary gradient for ammonia on urinary excretion of ammonia. Kidney Int 27:652–661
Tannen R (1978) Ammonia metabolism. Am J Physiol 235:F265-F277
Tate SS, Meister A (1974) Interaction of gamma-glutamyl-transpeptidase with amino acids, dipeptides, and derivatives and analogs of glutathione. J Biol Chem 249:7593–7602
Thompson GA, Meister A (1980) Modulation of gammaglutamyl transpeptidase activities by hippurate and related compounds. J Biol Chem 255:2109–2113
Van Slyke DD, Phillips RA, Hamilton PB, Archibald RM, Futcher Ph, Hiller A (1943) Glutamine as source material of urinary ammonia. J Biol Chem 150:481–482
Vieira FL, Malnic G (1968) Hydrogen ion secretion by rat renal cortical tubules as studied by an antimony microelec-trode. Am J Physiol 214:710–718
Walser M (1986) Roles of urea production, ammonium excretion, and amino acid oxidation in acid-base balance. Am J Physiol 19:F181-F188
Welbourne TC (1974) Ammonia production and pathways of glutamine metabolism in the isolated perfused rat kidney. Am J Physiol 226:544–548
Welbourne TC, Dass PD (1981) Role of hippurate in acidosis-induced adaptation in renal gamma-glutamyltransferase. Life Sciences 29:253–258
Welbourne TC, Park PH (1986) Fate of intraluminal glutamine in the perfused renal tubule. Life Sciences 38:941–946
Welbourne TC, Phromphetcharat V (1984) Renal Glutamine Metabolism and Hydrogen Ion Homeostasis. In: (Ed) Häussinger D, Sies H, Glutamine Metabolism in Mammalian Tissues. Springer, Berlin Heidelberg New York Tokyo, pp. 161–177
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Silbernagl, S., Scheller, D. Formation and excretion of NH3↔NH +4 . new aspects of an old problem. Klin Wochenschr 64, 862–870 (1986). https://doi.org/10.1007/BF01725559
Issue Date:
DOI: https://doi.org/10.1007/BF01725559