Summary
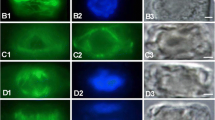
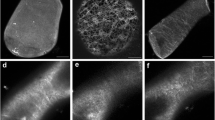
Tip cells of dark-grown protonemata of the mossCeratodon purpureus are negatively gravitropic (grow upward). They possess a unique longitudinal zonation: (1) a tip group of amylochloroplasts in the apical dome, (2) a plastid-free zone, (3) a zone of significant plastid sedimentation, and (4) a zone of mostly non-sedimenting plastids. Immunofluorescence of vertical cells showed microtubules distributed throughout the cytoplasm in a mostly axial orientation extending through all zones. Optical sectioning revealed a close spatial association between microtubules and plastids. A majority (two thirds) of protonemata gravistimulated for >20 min had a higher density of microtubules near the lower flank compared to the upper flank in the plastid-free zone. This apparent enrichment of microtubules occurred just proximal to sedimented plastids and near the part of the tip that presumably elongates more to produce curvature. Fewer than 5% of gravistimulated protonemata had an enrichment in microtubules near the upper flank, whereas 14% of vertical protonemata were enriched near one of the side walls. Oryzalin and amiprophos-methyl (APM) disrupted microtubules, gravitropism, and normal tip growth and zonation, but did not prevent plastid sedimentation. We hypothesize that a microtubule redistribution plays a role in gravitropism in this protonema. This appears to be the first report of an effect of gravity on microtubule distribution in plants.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Abbreviations
- APM:
-
amiprophos-methyl
- DIC:
-
differential interference contrast
- DMSO:
-
dimethyl sulfoxide
- EGTA:
-
ethylene glycolbis-(β-amino-ethylether) N,N,N',N'-tetraacetic acid
- FITC:
-
fluorescein isothiocyanate
- GS:
-
gravitropic stimulus
- MT:
-
microtubule
- PIPES:
-
piperazine-N,N'-bis-2-ethanesulfonic acid
References
Banbury GH (1962) Geotropism of lower plants. In: Bünning E (ed) Physiologie der Bewegungen: Bewegungen durch Einflüsse der Temperatur, Schwerkraft, chemischer Faktoren und aus inneren Ursachen. Springer, Berlin Göttingen Heidelberg, pp 344–377 [Ruhland W etal (eds) Handbuch der Pflanzenphysiologie-Encyclopedia of plant physiology, vol 17, part 2]
Bartnik E, Sievers A (1988) In-vivo observations of a spherical aggregate of endoplasmic reticulum and of Golgi vesicles in the tip of fast-growingChara rhizoids. Planta 176: 1–9
Doonan JH, Cove DJ, Lloyd CW (1985) Immunofluorescence microscopy of microtubules in intact cell lineages of the moss,Physcomitrella patens. I. Normal and CIPC-treated cells. J Cell Sci 75: 131–147
— —, Corke FMK, Lloyd CW (1987) Pre-prophase band of microtubules, absent from tip-growing moss filaments, arises in leafy shoots during transition to intercalary growth. Cell Motil Cytoskeleton 7: 138–153
— —, Lloyd CW (1988) Microtubules and microfilaments in tip growth: evidence that microtubules impose polarity on protonemal growth inPhyscomitrella patens. J Cell Sci 89: 533–540
Hartmann E (1984) Influence of light on phototropic bending of moss protonemata ofCeratodon purpureus (Hedw.) Brid. J Hattori Bot Lab 55: 87–98
—, Klingenberg B, Bauer L (1983) Phytochrome-mediated phototropism in protonemata of the mossCeratodon purpureus Brid. Photochem Photobiol 38: 599–603
Jenkins GI, Courtice GRM, Cove DJ (1986) Gravitropic responses of wild-type and mutant strains of the mossPhyscomitrella patens. Plant Cell Environ 9: 637–644
Kiss JZ, Sack FD (1989) Reduced gravitropic sensitivity in roots of a starch-deficient mutant ofNicotiana sylvestris. Planta 180: 123–130
Morejohn LC, Bureau TE, Mole-Bajer J, Bajer AS, Fosket DE (1987) Oryzalin, a dinitroaniline herbicide, binds to plant tubulin and inhibits microtubule polymerization in vitro. Planta 172: 252–264
Sack FD, Kiss JZ (1989) Plastids and gravity perception. In: Boyer CD, Shannon JC, Hardison RC (eds) Physiology, biochemistry, and genetics of nongreen plastids. American Society of Plant Physiologists, Rockville, Maryland, pp 171–181
Schmiedel G, Schnepf E (1979) Side branch formation and orientation in the caulonema of the moss,Funaria hygrometrica: normal development and fine structure. Protoplasma 100: 367–383
— — (1980) Polarity and growth of caulonema tip cells of the mossFunaria hygrometrica. Planta 147: 405–413
Schnepf E (1986) Cellular polarity, Annu Rev Plant Physiol 37: 23–47
Tewinkel M, Volkmann D (1987) Observations on dividing plastids in the protonema of the mossFunaria hygrometrica Sibth. Planta 172: 309–320
Volkmann D, Sievers A (1979) Graviperception in multicellular organs. In: Haupt W, Feinleib ME (eds) Physiology of movements. Springer, Berlin Heidelberg New York, pp 573–600 [Pirson A, Zimmermann MH (eds) Encyclopedia of plant physiology, ns, vol 7]
Wacker I, Quader H, Schnepf E (1988) Influence of the herbicide Oryzalin on cytoskeleton and growth ofFunaria hygrometrica protonemata. Protoplasma 142: 55–67
Walker LM, Sack FD (1990) Amyloplasts as possible statoliths in gravitropic protonemata of the mossCeratodon purpureus. Planta 181: 71–77
White RG, Sack FD (1990) Actin microfilaments in presumptive statocytes of rootcaps and coleoptiles. Amer J Bot 77: 17–26
Young JC, Sack FD (1989) Videomicroscopy-based observation of gravitropism in individual protonema of the mossCeratodon. Am Soc Gravitat Space Biol Bull 2: 61
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Schwuchow, J., Sack, F.D. & Hartmann, E. Microtubule distribution in gravitropic protonemata of the mossCeratodon . Protoplasma 159, 60–69 (1990). https://doi.org/10.1007/BF01326635
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF01326635