Summary
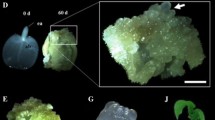
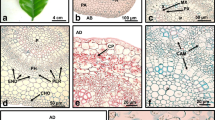
Seeds of theArabidopsis thaliana mutant primordia timing (pt) were germinated in 2,4-dichlorophenoxyacetic acidcontaining liquid medium. The seedlings formed somatic embryos and nonembryogenic and embryogenic callus in vitro in a time period of approximately two to three weeks. Embryogenesis and callus formation were monitored with respect to origin, structure, and development. Ten days after germination globular structures appeared in close vicinity of and on the shoot apical meristem (SAM). Somatic embryos formed either directly on the SAM region of the seedling or indirectly on embryogenic callus that developed at the SAM zone. Globular structures developed along the vascular tissue of the cotyledons as well, but only incidentally they formed embryos. Upon deterioration, the cotyledons formed callus. Regular subculture of the embryogenic callus gave rise to high numbers of somatic embryos. Such primary somatic embryos, grown on callus, originated from meristematic cell clusters located under the surface of the callus. Embryos at the globular and heart-shape stage were mostly hidden within the callus. Embryos at torpedo stage appeared at the surface of the callus because their axis elongated. Secondary somatic embryos frequently formed directly on primary ones. They preferentially emerged from the SAM region of the primary somatic embryos, from the edge of the cotyledons, and from the hypocotyl. We conclude that the strong regeneration capacity of thept mutant is based on both recurrent and indirect embryogenesis.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Abbreviations
- 2,4-D:
-
2,4-dichlorophenoxyacetic acid
- DIC:
-
days in culture
- SAM:
-
shoot apical meristem
References
Chaudhury AM, Letham S, Craig S, Dennis ES (1993)amp1: a mutant with high cytokinin levels and altered embryonic pattern, faster vegetative growth, constitutive photomorphogenesis and precocious flowering. Plant J 4: 907–916
Choi JH, Sung ZR (1989) Induction, commitment, and progression of plant embryogenesis. In: Kung S, Arntzen CJ (eds) Plant biotechnology. Butterworths, Boston, pp 141–159
Clark S (1997) Organ formation at the vegetative shoot meristem. Plant Cell 9: 1067–1076
Conway LJ, Poethig RS (1997) Mutations ofArabidopsis thaliana that transform leaves into cotyledons. Proc Natl Acad Sci USA 94: 10209–10214
Cutter EG (1965) Recent experimental studies of the shoot apex and shoot morphogenesis. Bot Rev 31: 7–113
das Neves LO, Duque SRL, de Almeida JS, Fevereiro PS (1999) Repetitive somatic embryogenesis inMedicago truncatula ssp. Narbonensis andM. truncatula Gaertn cv. Jemalong. Plant Cell Rep 18: 398–405
de Vries SC, Booij H, Meyerink P, Huisman G, Wilde HD, Thomas TL, van Kammen A (1988) Acquisition of embryogenic potential in carrot cell suspension cultures. Planta 176: 196–204
Dubrovsky JG, Tykarska T (1995) Visualization of the radicle within the axis of developing and germinatingBrassica napus L. embryos. Environ Exp Bot 35: 93–104
Hou Y, von Arnim F, Deng X-W (1993) A new class ofArabidopsis constitutive photomorphogenic genes involved in regulating cotyledon development. Plant Cell 5: 329–339
Huang B, Yeoman MM (1995) Somatic embryogenesis inArabidopsis thaliana L. In: Bajaj YPS (ed) Somatic embryogenesis and synthetic seed II. Springer, Berlin Heidelberg New York Tokyo, pp 371–384 (Biotechnology in agriculture and forestry, vol 31)
Jürgens G, Mayer U, Torres Ruiz RA, Berleth T, Miséra S (1991) Genetic analysis of pattern formation in theArabidopsis embryo. Development 1 Suppl: 27–38
Kerstetter RA, Hake S (1997) Shoot meristem formation in vegetative development. Plant Cell 9: 1001–1010
Laparra H, Bronner R, Hahne G (1997) Amyloplasts as a possible indicator of morphogenetic potential in sunflower protoplasts. Plant Sci 122: 183–192
Luo Y, Koop HU (1997) Somatic embryogenesis in cultured immature zygotic embryos and leaf protoplasts ofArabidopsis thaliana ecotypes. Planta 202: 387–396
Medford JI (1992) Vegetative apical meristems. Plant Cell 4: 1029–1039
Meyerowitz EM (1989)Arabidopsis, a useful weed. Cell 56: 263–269
— (1998) Genetic control of cell division patterns in developing plants. Cell 88: 299–308
Mordhorst AP, Toonen MAJ, de Vries SC (1997) Plant embryogenesis. Crit Rev Plant Sci 16: 535–576
—, Voerman KJ, Hartog MV, Meijer EA, van Went J, Koornneef M, de Vries SC (1998) Somatic embryogenesis inArabidopsis thaliana is facilitated by mutations in genes repressing meristematic cell divisions. Genetics 149: 549–563
Murashige T, Skoog F (1962) A revised medium for rapid growth and bioassay with tobacco tissue cultures. Physiol Plant 15: 473–497
O'Neill CM, Mathias RJ (1993) Regeneration of plants from protoplasts ofArabidopsis thaliana L. cv. Columbia (C24), via direct embryogenesis. J Exp Bot 44: 1579–1585
Parra R, Amo-Marco JB (1998) Secondary somatic embryogenesis and plant regeneration in myrtle (Myrtus communis L.). Plant Cell Rep 18: 325–330
Pillon E, Terzi M, Baldan B, Mariani P, Lo Schiavo F (1996) A protocol for obtaining embryogenic cell lines fromArabidopsis. Plant J 9: 573–577
Puigderrajols R, Fernandez-Guijarro B, Toribio M, Molinas M (1996) Origin and early development of secondary embryos inQuercus suber L. Int J Plant Sci 157: 674–684
Rodkiewicz B, Fyk B, Szczuka E (1994) Chlorophyll and cutin in early embryogenesis inCapsella, Arabidopsis, andStellaria investigated by fluorescence microscopy. Sex Plant Reprod 7: 287–289
Sangwan RS, Bourgeois Y, Dubois F, Sangwan-Norreel BS (1992) In vitro regeneration ofArabidopsis thaliana from cultured zygotic embryos and analysis of regenerants. J Plant Physiol 140: 588–595
Steeves TA, Sussex IM (1989) Patterns in plant development. Cambridge University Press, New York
Sussex IM (1989) Developmental programming of the shoot meristem. Cell 56: 225–229
Tetteroo FAA, Hoekstra FA, Karssen CM (1995) Induction of complete desiccation tolerance in carrot (Daucus carota L.) embryoids. J Plant Physiol 145: 349–3566
Wu Y, Haberland G, Zhou C, Koop HU (1992) Somatic embryogenesis, formation of morphogenetic callus and normal development in zygotic embryos ofArabidopsis thaliana in vitro. Protoplasma 169: 89–96
Zimmermann JL (1993) Somatic embryogenesis: a model for early development in higher plants. Plant Cell 5: 1411–1423
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
von Recklinghausen, I.R., Iwanowska, A., Kieft, H. et al. Structure and development of somatic embryos formed inArabidopsis thaliana pt mutant callus cultures derived from seedlings. Protoplasma 211, 217–224 (2000). https://doi.org/10.1007/BF01304489
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF01304489