Summary
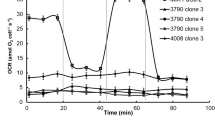
Growth of K-562 cells in culture is inhibited by the antitumor sulfonylureaLY181984 (N-(4-methylphenylsulfonyl)-N′-(4-chlorophenyl)urea) with an ED50 of about 30 μM. LY181984 was shown previously to inhibit NADH oxidation by plasma membranes from HeLa cells and other sources and to influence mitochondrial oxidative phosphorylation. With K-562 cells, NADH oxidation by plasma membranes was transiently stimulated and then inhibited by LY181984. NADH oxidation by whole cells was transiently stimulated and then inhibited by 0.1 to 100 μM LY181984 as well. Both the stimulations and inhibitions of activity were time-dependent. NADH oxidation by lower phase membranes depleted of plasma membranes by aqueous two-phase partition also was inhibited by micromolar and submicromolar concentrations of LY181984. Inhibition did not correlate with mitochondrial presence but rather with membranes that appeared to be fragments of the Golgi apparatus. The oxidation of NADH by whole cells and of plasma membranes that was inhibited by LY181984 was distinguished from mitochondrial NADH oxidation by resistance to inhibition by cyanide and by proceeding under oxygen-depleted conditions or an argon atmosphere. In contrast to the active antitumor agent LY181984, the inactive but chemically-related analog, LY181985 (N-(4-methylphenyl-sulfonyl)-N′-(4-phenylurea), inhibited neither growth nor NADH oxidation with K-562 cells or cell fractions.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Grindey GB (1988) Identification of diarylsulfonylureas as novel anticancer drugs. Proc Am Assoc Cancer Res 29: 535–536
—, Boder GB, Grossman CS, Howbert JJ, Poore GA, Shaw WH, Todd CG, Worzella JF (1987) Further development of diarylsulfonylureas as novel anticancer drugs. Proc Am Assoc Cancer Res 28: 309
Hainsworth JD, Hande KR, Satterlee WG, Kuttesch J, Johnson DH, Grindey GB, Jackson LE, Greco FA (1989) Phase I clinical study of N-[(4-chlorophenyl)amino]carbonyl-2,3-dihydro-1-H-indene-5-sulfonamide (LY186641). Cancer Res 49: 5217–5220
Houghton PJ, Bailey FC, Germain GS, Grindey GB, Witt BC, Houghton JA (1990 a) N-(5-indanylsulfonyl)-N'-(4-chlorophenyl)urea, a novel agent equally cytotoxic to nonproliferating human colon adenocarcinoma cells. Cancer Res 50: 318–322
— —, Houghton JA, Murti KG, Howbert J, Grindey GB (1990 b) Evidence for mitochondrial localization of N-(4-methylphenylsulfonyl)-N′-(4-chlorophenyl)urea in human colon adenocarcinoma cells. Cancer Res 50: 664–668
Howbert JJ, Grossman CS, Crowell TA, Rieder BJ, Harper RW, Kramer KE, Tao EV, Aikins J, Poore GA, Rinzel SM, Grindey GB, Shaw WN, Todd GC (1990) Novel agents effective against solid tumors: the diarylsulfonylureas. Synthesis, activities, and analysis of quantitative structure-activity relationships. J Med Chem 33: 2393–2407
LaRossa RA, Schloss JV (1984) The sulfonylurea herbicide sulfometruon methyl is an extremely potent and selective inhibitor of acetolactate synthase inSalmonella typhimurium. J Biol Chem 259: 8753–8757
Luft JM (1961) Improvements in epoxy resin embedding methods. J Biophys Biochem Cytol 9: 409–414
Morré DJ (1973) Isolation and purification of organelles and endomembrane components from rat liver. In: Chrispeels MJ (ed) Molecular techniques and approaches in developmental biology. Wiley, New York, pp 1–27
—, Morré DM (1989) Mammalian plasma membranes by aqueous two-phase partition. Biotechniques 7: 946–958
—, Reust T, Morré DM (1994 a) Plasma and internal membranes from cultured mammalian cells. Methods Enzymol 228: 448–450
—, Geilen C, Morré DM (1994 b) Golgi apparatus respond to the antitumor sulfonylurea N-(4-methylphenylsulfonyl)-N′-(4-chlorophenyl) urea (LY181984) in sulfonylurea-susceptible but not resistant cell lines. Protoplasma 182: 81–95
—, Merriman R, Tanzer LR, Wu L-Y, Morré DM, MacKellar WC (1995) Inhibition of the NADH oxidase activity of plasma membranes isolated from xenografts and cell lines by the antitumor sulfonylurea, N-(4-methylphenylsulfonyl)-N′-(4-chlorophenyl)urea (LY181984) correlates with drag susceptibility of growth. Protoplasma 184: 203–208
Morré DM, Morré DJ (1995) Mechanism of killing of HeLa cells by the antitumor sulfonylurea, N-(4-methylphenyl)-N′-(4-chlorophenyl)urea (LY181984). Protoplasma 184: 188–195
Navas P, Nowack DD, Morré DJ (1989) Isolation of purified plasma membranes from cultured cells and hepatomas by two-phase partition and preparative free-flow electrophoresis. Cancer Res 49: 2147–2156
Pennington RJ (1961) Biochemistry of dystrophic muscle. Succinate-tetrazolium reductase and adenosine triphosphatase. Biochem J 80: 649–654
Rush GF, Rinzel S, Boder G, Heim RA, Toth JE, Ponsler DG (1992) Effects of diarylsulfonylurea antitumor agents on the function of mitochondria isolated from rat liver and GC3/C1 cells. Biochem Pharmacol 44: 2387–2394
Schloss JV, Lawrence MC, Van Dyk DE (1988) Origin of the herbicide binding site of acetolactate synthase. Nature 331: 360–362
Smith PK, Krohn RI, Hermanson GK, Mallia AK, Gartner FH, Provenzano MD, Fujimoto EK, Goekke NM, Olson BJ, Klenk DC (1985) Measurement of protein using bicinchoninic acid. Anal Biochem 150: 76–85
Sosinski J, Thakar JH, Germain GS, Harwood FC, Houghton PJ (1993) Proliferation-dependent and -independent cytotoxicity by antitumor diarylsulfonylureas. Biochem Pharmacol 45: 2135–2142
Sun IL, Sun EE, Crane FL, Morré DJ, Lindgren A, Löw H (1992) Requirement for coenzyme Q in plasma membrane electron transport. Proc Natl Acad Sci USA 89: 11126–11130
Taylor CW, Alberts DS, Ketcham MA, Satterlee WG, Holdsworth MT, Pleijia PM, Pang Y-M, McCloskey TM, Roe DJ, Hamilton M, Salmon SE (1989) Clinical pharmacology of a novel diarylsulfonylurea anticancer agent. J Clin Oncol 7: 1733–1740
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Moya-Camarena, S.Y., Morré, D.J. & Morré, D.M. Response of NADH oxidation and growth in K-562 cells to the antitumor sulfonylurea, N-(4-methylphenylsulfonyl)-N′-(4-chlorophenyl)urea (LY181984). Protoplasma 188, 151–160 (1995). https://doi.org/10.1007/BF01280366
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF01280366