Abstract
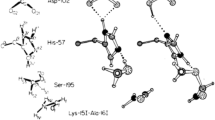
Histidine dihydrochloride crystallizes in the orthorhombic system, space groupP212121, with unit-cell dataa = 6.970(4),b = 16.165(7),c = 8.964(6)Å,V c = 1010.0Å3 andZ = 4. Intensities for 1360 independent reflections were collected on an automated diffractometer. The structure was solved by standard heavy-atom methods and refined by full-matrix least squares, based onF, to anR-value of 0.027. The final weightedR-value and goodness-of-fit are 0.029 and 1.9, respectively. The histidine residue exists in the crystal as the carboxylic acid with protonation of the primary amine nitrogen and the imidazole ring. The overall conformation of the molecule is extended, similar to that observed inL-histidine,DL-histidine hydrochloride dihydrate, and one of the two independent molecules in the structure ofL-N-acetylhistidine. There is an extensive array of hydrogen bonds formed in the crystal, which, presumedly, together with the observed electrostatic interactions, account for the adopted molecular conformation.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Baur, W. H. (1972)Acta Cryst.,B28, 1456.
Bennett, I., Davidson, A. G. H., Harding, M. M. & Morelle, I., (1970)Acta Cryst.,B26, 1722.
Busing, W. R. & Levy, H. A. (1957)J. Chem. Phys.,26, 563.
Busing, W. R., Martin, K. O. & Levy, H. A. (1962) ORFLS. Report ORNL-TM-305, Oak Ridge National Laboratory, Oak Ridge,Tennessee, U.S.A.
Carlstrom, D., Bergin, R. & Falkenberg, G. (1973)Quart. Rev. Biophysics,6, 257.
Churchill, M. R. (1973)Inorg. Chem.,12, 1213.
Cromer, D. T. (1965)Acta. Cryst.,18, 17.
Donohue, J., Lavine, L. R. & Rollett, J. S. (1956)Acta. Cryst.,9, 655.
Donohue, J. & Caron, A. (1964)Acta. Cryst.,17, 1178.
Freeman, H. C. (1967)Advanc. Protein Chem.,22, 235.
Hamilton, W. C. (1968). “Structural Chemistry and Molecular Biology”, edited by A. Rich & N. Davidson, Freeman, San Francisco, California, U.S.A.
Hanson, H. P., Herman, F., Lea, J. D. & Skillman, S. (1964)Acta. Cryst.,17, 1040.
Johnson, C. K. (1965). ORTEP. Report ORNL-3794, Oak Ridge National Laboratory, Oak Ridge, Tennessee, U.S.A.
Kistenmacher, T. J. & Marsh, R. E. (1971)Science,172, 945.
Kistenmacher, T. J., Hunt, D. J. & Marsh, R. E. (1972)Acta Cryst.,B28, 3352.
Kistenmacher, T. J. & Marsh, R. E. (1972)J. Amer. Chem. Soc.,94, 1340.
Kistenmacher, T. J. & Sorrell, T. (1973)Cryst. Struct. Comm.,2, 673.
Madden, J. J., McGandy, E. L. & Seeman, N. C. (1972)Acta Cryst.,B28, 2377.
Madden, J. J., McGandy, E. L., Seeman, N. C., Harding, M. M. & Hoy, A. (1972)Acta Cryst.,B28, 2382.
Marsh, R. E. & Donohue, J. (1967)Advanc. Protein Chem.,22, 235.
Nardelli, M., Fava, G. & Giraldi, G. (1962)Acta Cryst.,15, 737.
Oda, K. & Koyama, H. (1972)Acta Cryst.,B28, 639.
Pauling, L. (1960). “The Nature of the Chemical Bond”, 3rd. ed., Cornell University Press, Ithaca, New York, U.S.A.
Pippy, M. E. & Ahmed, F. R. (1968). NRC Crystallographic Programs, National Research Council, Ottawa, Canada.
Stewart, J. M. (1967). X-RAY 67. Technical Report 67-58, Computer Science Center, University of Maryland, College Park, Maryland, U.S.A.
Stewart, R. F., Davidson, E. R. & Simpson, W. T. (1965)J. Chem. Phys.,42, 3175.
Westhof, E. (1972). Private communication.
Wilson, A. J. C. (1942)Nature (London),150, 152.
Author information
Authors and Affiliations
Additional information
This investigation was supported by the donors of the Petroleum Research Fund, administered by the Americal Chemical Society, and by a National Institutes of Health Biomedical Science Support Grant. The diffraction equipment was purchased through a grant from the National Science Foundation.
Rights and permissions
About this article
Cite this article
Kistenmacher, T.J., Sorrell, T. Further conformational aspects of the amino-acid histidine: crystal and molecular structure of histidine dihydrochloride. Journal of Crystal and Molecular Structure 4, 419–432 (1974). https://doi.org/10.1007/BF01220098
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF01220098