Abstract
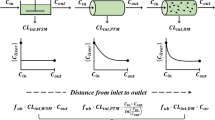
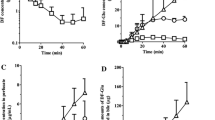
Theoretical analysis of two models of hepatic drug clearance revealed that one powerful discriminator between them is the effect of changes of hepatic blood flow on either the emergent drug concentration or the availability of a highly extracted compound when operating under linear conditions. Lidocaine (extraction ratio 0.997) was employed in the discriminatory studies. The behavior of this drug under linear conditions (input lidocaine concentrations < 5 mg/ liter) to changes in hepatic blood flow rate (10–16 ml/min per liver) was examined in the perfused rat liver in situpreparation. The steady-state output lidocaine concentration in the blood leaving the liver was predicted better by a “well-stirred” model than by a “parallel tube” model. As anticipated, the clearance of a poorly extracted compound, antipyrine (extraction ratio 0.08),was unaltered by changes in hepatic blood flow. These experimental findings, and the data from the literature, point to the acceptance of the “well-stirred” model, which describes the liver as a well-stirred compartment with the drug in the hepatic venous blood being in equilibrium with that in the liver.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
K. S. Pang and M. Rowland. Hepatic clearance of drugs. I. Theoretical considerations of a “well-stirred” model and a “parallel tube” model. Influence of hepatic blood flow, plasma and blood cell binding, and the hepatocellular enzymatic activity on hepatic drug clearance.J. Pharmokin. Biopharm. 5:625–653 (1977).
M. Rowland, L. Z. Benet, and G. E. Graham. Clearance concepts in pharmacokinetics.J. Pharmacokin. Biopharm. 1:123–136 (1973).
K. Winkler, S. Keiding, and N. Tygstrup. Clearance as a quantitative measure of liver function. In P. Paumgartner and R. Presig (eds.),The Liver: Quantitative Aspects of Structure and Function, Karger, Basel, 1973, pp. 144–155.
K. Winkler, L. Bass, S. Keiding, and N. Tygstrup. The effect of hepatic perfusion on the assessment of kinetic constants. In F. Lundquist and N. Tygstrup (eds.),Alfred Benzon Symposium VI: The Regulation of Hepatic Metabolism, Munksgaard, Copenhagen, 1974. pp. 797–807.
G. E. Mortimore. Effect of insulin on potassium transfer in isolated rat liver.Am. J. Physiol. 200:1315–1319 (1961).
B. D. Ross.Perfusion Techniques in Biochemistry: A Laboratory Manual, Clarendon Press, Oxford, 1972.
N. Benowitz and M. Rowland. Determination of lidocaine in blood and tissues.Anesthesiology 39:639–641 (1973).
H. Halkin, P. Meffin, K. L. Melmon, and M. Rowland. Influence of congestive heart failure on blood levels of lidocaine and its active monodeethylated metabolite.Clin. Pharmacol. Ther. 17:669–676 (1975).
P. J. Meffin, R. L. Williams, T. F. Blaschke, and M. Rowland. Application of salivary concentration data to pharmacokinetic studies with antipyrine.J. Pharm. Sci. 66:135–137 (1977).
H. G. Weder, J. Schildknecht, and A. Kesselring. A new equilibrium dialysing system.Am. Lab. 10:15–21 (1971).
O. H. Lowry, N. J. Rosenbrough, A. L. Falr, and R. J. Randall. Protein measurements with the Folin phenol reagent.J. Biol. Chem. 193:265–275 (1951).
G. T. Tucker, R. N. Boyes, P. O. Bridenbaugh, and D. C. Moore. Binding of anilide-type local anesthetics in human plasma. II. Implicationsin vivo, with special reference to transplacental distribution.Anesthesiology 34:538–549 (1971).
B. B. Brodie and J. Axelrod. The fate of antipyrine in man.J. Pharmacol. Exp. Ther,98:97–104 (1950).
R. W. Brauer, G. F. Leong, R. F. McElroy, Jr., and R. J. Holloway. Hemodynamics of the vascular tree of the isolated rat liver preparation.Am. J. Physiol. 186:537–542 (1956).
J. J. Ashley and G. Levy. Inhibition of diphenylhydantoin elimination by its major metabolite.Res. Commun. Pathol. Pharmacol. 4:297–306 (1972).
E. Jähnchen and G. Levy. Inhibition of phenylbutazone elimination by its metabolite oxyphenbutazone.Proc. Soc. Biol. Exp. Med. 141:963–965 (1972).
J. B. Keenaghan and R. N. Boyes. The tissue distribution, metabolism and excretion of lidocaine in rats, guinea pigs, dogs, and man.J. Pharmacol. Exp. Ther. 180:454–463 (1972).
R. W. Brauer, G. F. Leong, R. F. McElroy, Jr., and R. J. Holloway. Circulatory pathways in the rat liver as revealed by P32 chromic phosphate colloid uptake in the isolated perfused liver preparation.Am. J. Physiol. 184:593–598 (1956).
T. L. Whitsett, P. G. Dayton, and J. L. McNay. The effect of hepatic blood flow on the hepatic removal rate of oxyphenbutazone in the dog.J. Pharmacol. Exp. Ther. 177:246–255 (1971).
C. M. Metzler.A User's Manual for NONLIN, Technical Report 7292/69/7292/025, Upjohn Co., Kalamazoo, Mich., Nov. 25, 1969.
H. G. Boxenbaum, S. Riegelman, and R. M. Elashoff. Statistical estimations in pharmacokinetics.J. Pharmacokin. Biopharm. 2:123–148 (1974).
R. A. Branch, A. S. Nies, and D. G. Shand. The disposition of propranolol. VIII. General implication of the effects of liver blood flow in elimination from the perfused rat liver.Drug Metab. Disp. 1:687–690 (1973).
D. G. Shand, D. M. Kernhauser, and G. R. Wilkinson. Effects of route of administration and blood flow on hepatic drug elimination.J. Pharmacol. Exp. Ther. 195:424–432 (1975).
S. Keiding, S. Johansen, K. Winkler, K. Tønnesen, and N. Tygstrup. Michaelis-Menten kinetics of galactose elimination by the isolated perfused pig liver.Am. J. Physiol. 230:1302–1313 (1976).
O. Levenspiel.Chemical Reaction Engineering, 2nd ed., Wiley, New York, 1972.
R. W. Brauer. Liver circulation and function.Physiol. Rev. 43:115–213 (1963).
S. S. Kety. Theory and applications of the exchange of inert gas at the lungs and tissues.Pharmacol. Rev. 3:1–41 (1951).
K. B. Bischoff, R. L. Dedrick, and D. S. Zaharko. Preliminary model for methotrexate pharmacokinetics.J. Pharm. Sci. 59:149–154 (1970).
Author information
Authors and Affiliations
Additional information
Supported in part by National Institutes of Health Grant GM 16496 and the Patent Fund, Graduate Division, University of California, San Francisco.
Abstracted in part from a dissertation submitted by K. Sandy Pang to the Graduate Division, University of California, San Francisco, California, in partial fulfillment of the Doctor of Philosophy degree requirements.
Rights and permissions
About this article
Cite this article
Pang, K.S., Rowland, M. Hepatic clearance of drugs. II. Experimental evidence for acceptance of the “well-stirred” model over the “parallel tube” model using lidocaine in the perfused rat liverin situ preparation. Journal of Pharmacokinetics and Biopharmaceutics 5, 655–680 (1977). https://doi.org/10.1007/BF01059689
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF01059689