Abstract
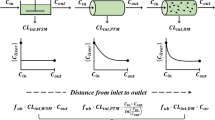
Two commonly used models of hepatic drug clearance are examined. The “well-stirred” model (model I) views the liver as a well-stirred compartment with concentration of drug in the liver in equilibrium with that in the emergent blood. The “parallel tube” model (model II) regards the liver as a series of parallel tubes with enzymes distributed evenly around the tubes and the concentration of drug declines along the length of the tube. Both models are examined under steady-state considerations in the absence of diffusional limitations (cell membranes do not limit the movement of drug molecules). Equations involving the determinants of hepatic drug clearance (hepatic blood flow, fraction of drug in blood unbound, and the hepatocellular enzymatic activity) and various pharmacokinetic parameters are derived. Similarities and differences between the models are explored. Although both models predict similar hepatic drug clearances under a variety of conditions, marked differences between them become apparent in their predictions of the influence of changes in the determinants of drug clearance on various pharmacokinetic parameters.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Abbreviations
- AUC:
-
total area under the blood drug concentration-time profile
- C :
-
drug concentration
- In C Out :
-
concentration of drug entering and leaving the liver, respectively
- Ĉ :
-
logarithmic average concentration of drug in hepatocyte,\(\hat C = \frac{{C_{In} - C_{Out} }}{{In({{C_{In} } \mathord{\left/ {\vphantom {{C_{In} } {C_{Out} }}} \right. \kern-\nulldelimiterspace} {C_{Out} }})}}\)
- CL:
-
steady-state hepatic drug clearance
- CLint :
-
intrinsic hepatic drug clearance
- CLin,t :
-
intrinsic hepatic drug clearance when operating under linear conditions (C L,u ≪K m,i )
- E :
-
steady-state hepatic extraction ratio
- f B :
-
ratio of the unbound drug concentration in plasma water to the whole blood drug concentration
- f P :
-
ratio of the unbound drug concentration in plasma water to the total plasma drug concentration
- f bc :
-
ratio of the unbound drug concentration in plasma water to the total drug concentration in blood cells
- F :
-
systemic availability of a drug given orally
- H :
-
hematocrit
- K m,i :
-
Michaelis-Menten constant of the ith enzyme
- R :
-
rate of drug administration
- t1/2 :
-
elimination half-life of the drug
- v :
-
velocity of a reaction
- V :
-
volume
- Q :
-
hepatic blood flow
- Vmax,i :
-
maximum velocity of the ith enzyme
- τ:
-
interval between doses
- L, B, BC, P, andR :
-
liver, whole blood, blood cells, plasma, and reservoir, respectively
- x and tube:
-
pointx and the tube, respectively
- u :
-
unbound drug
- oral, i.V., inf:
-
oral and intravenous routes and constant intravenous infusion, respectively
- l and ss:
-
linear and steady-state conditions
References
E. Möller, J. R. McIntosh, and D. D. Van Slyke. Studies of urea excretion.J. Clin. Invest. 6:427–465 (1929).
P. A. Shore, B. B. Brodie, and C. A. M. Hogben. The gastric secretion of drugs: A pH partition hypothesis.J. Pharmacol. Exp. Ther. 119:361–369 (1957).
H. O. Heinemann and A. P. Fishman. Nonrespiratory function of the mammalian lung.Physiol. Rev. 49:1–47 (1969).
M. Rowland, S. Riegelman, P. A. Harris, and S. D. Sholkoff. Absorption kinetics of aspirin in man following oral administration of an aqueous solution.J. Pharm. Sci. 61:379–385 (1972).
J. G. Wagner, J. I. Northam, C. D. Alway, and O. S. Carpenter. Blood levels of drug at the equilibrium state after multiple dosing.Nature 201:1301–1302 (1965).
M. Gibaldi, R. N. Boyes, and S. Feldman. Influence of first pass effect on availability of drugs.J. Pharm. Sci. 60:1338–1340 (1971).
M. Rowland. The influence of route of administration on drug availability.J. Pharm. Sci. 61:70–74 (1972).
P. D. Berk, T. F. Blaschke, and J. G. Waggoner. Defective bromosulphthalein clearance in patients with constitutional hepatic dysfunction (Gilbert's syndrome).Gastroenterology 63:472–481 (1972).
G. R. Wilkinson. Pharmacokinetics of drug dispostion: Hemodynamic considerations.Ann Rev. Pharmacol. 15:11–27 (1975).
M. Rowland, L. Z. Benet, and G. G. Graham. Clearance concepts in pharmacokinetics.J. Pharmacokin. Biopharm. 1:123–136 (1973).
D. G. Shand and R. E. Rangno. The disposition of propranolol. I. Elimination during oral absorption in man.Pharmacology 7:159–168 (1972).
R. N. Boyes, H. J. Adams, and B. R. Duce. Oral absorption and disposition kinetics of lidocaine hydrochloride in dogs.J. Pharmacol. Exp. Ther. 174:1–9 (1970).
R. L. Wolen, C. M. Gruber, Jr., G. F. Kiplinger, and N. E. Scholz. Concentration of propoxyphene in human plasma following oral, intramuscular, and intravenous administration.Toxicol. Appl. Pharmacol. 19:480–492 (1971).
R. A. Branch, D. G. Shand, G. R. Wilkinson, and A. S. Nies. Increased clearance of antipyrine and d-propranolol after phenobarbital treatment in the monkey.J. Clin. Invest. 53:1101–1107 (1974).
R. W. Brauer, G. F. Leong, R. F. McElroy, Jr., and R. J. Holloway. Circulatory pathways in the rat liver as revealed by P32 chromic phosphate colloid uptake in the isolated perfused liver preparation.Am. J. Physiol. 184:593–598 (1956).
G. R. Wilkinson and D. G. Shand. Commentary: A physiological approach to hepatic drug clearance.Clin. Pharmacol. Ther. 18:377–390 (1975).
G. Levy and A. Yacobi. Effect of protein binding on the elimination of warfarin.J. Pharm. Sci. 63:805–806 (1974).
K. Winkler, S. Keiding, and N. Tygstrup. Clearance as a quantitative measure of liver function. In P. Paumgartner and R. Presig (eds.),The Liver: Quantitative Aspects of Structure and Functions, Karger, Basel, 1973, pp. 144–155.
K. Winkler, L. Bass, S. Keiding, and N. Tygstrup. The effect of hepatic perfusion on assessment of kinetic constants. In F. Lundquist and N. Tygstrup (eds.),Alfred Benson Symposium VI: Regulation of Hepatic Metabolism, Munksgaard, Copenhagen, 1974, pp. 797–807.
J. R. Gillette. Other aspects of pharmacokinetics. In J. R. Gillette and J. R. Mitchell (eds.),Concepts in Biochemical Pharmacology, Part 3, Springer-Verlag, New York, 1975, pp. 35–85.
C. V. Greenway and R. D. Stark. Hepatic vascular beds.Physiol. Rev. 51:23–65 (1971).
L. Bass, S. Keiding, K. Winkler, and N. Tygstrup. Enzymatic elimination of substrates flowing through the intact liver.J. Theor. Biol. 61:393–409 (1976).
C. A. Goresky and H. L. Goldsmith. Capilliary-tissue exchange kinetics: Diffusional interactions between adjacent capillaries.Adv. Exp. Biol. Med. 37B:773–781 (1973).
S. Keiding, S. Johansen, K. Winkler, K. Tønnesen, and N. Tygstrup. Michaelis-Menten kinetics of galactose elimination by the isolated perfused pig iiver.Am. J. Physiol. 230:1302–1313 (1976).
R. A. Branch, A. S. Nies, and D. G. Shand. The disposition of propranolol. VIII. General implication of the effects of liver blood flow in elimination from the perfused rat liver.Drug Metab. Disp. 1:687–690 (1973).
D. G. Shand, D. M. Kernhauser, and G. R. Wilkinson. Effects of route of administration and blood flow on hepatic drug elimination.J. Pharmacol. Exp. Ther. 195:424–432 (1975).
D. Perrier and M. Gibaldi. Clearance and biologic half-life as indices of intrinsic hepatic metabolism.J. Pharmacol. Exp. Ther. 191:17–24 (1974).
D. G. Shand, R. H. Cotham, and G. R. Wilkinson. Perfusion-limited effects of plasma drug binding on hepatic drug extraction.Life Sci. 19:125–130 (1976).
G. R. Wilkinson. Pharmacokinetics in disease states modifying body perfusion. In L. Z. Benet (ed.),The Effect of Disease States on Drug Pharmacokinetics, American Pharmaceutical Association of Pharmaceutical Sciences, Washington, D.C., 1976, pp. 13–22.
A. Fischer. Dynamics of the circulation of the liver. In C. Rouiller (ed.),The Liver, Vol. I, Academic Press, New York, 1963, pp. 329–378.
B. D. Ross.Perfusion Techniques in Biochemistry: A Laboratory Manual, Clarendon Press, Oxford, 1972.
J. W. Culbertson, R. W. Wilkins, F. J. Ingelfinger, and S. E. Bradley. The effect of upright position upon the hepatic blood flow.J. Clin. Invest. 30:305–311 (1951).
R. E. Stenson, R. T. Constantino, and D. C. Harrison. Interrelationships of hepatic blood flow, cardiac output and blood levels of lidocaine in man.Circulation 43:205–211 (1971).
K. S. Pang and M. Rowland. Hepatic clearance of drugs. II. Experimental evidence for acceptance of the “well-stirred” model over the “parallel tube” model using lidocaine in the perfused rat liverin situ preparation.J. Pharmacokin. Biopharm. 5:655–680 (1977).
T. L. Whitsett, P. G. Dayton, and J. L. McNay. The effect of hepatic blood flow on the hepatic removal of oxyphenbutazone in the dog.J. Pharmacol. Exp. Ther. 177:246–255 (1971).
M. A. Gonzalez, T. N. Tozer, and D. T. T. Chang. Nonlinear tissue disposition: Salicylic acid in rat brain.J. Pharm. Sci. 64:99–103 (1975).
E. R. Garrett and C. A. Hunt. Physicochemical properties, solubility, and protein binding of Δ9-tetrahydrocannabinol.J. Pharm. Sci. 63:1054–1064 (1974).
S. Keiding. Hepatic elimination kinetics: The influence of hepatic blood flow on clearance determinations.Scand. J. Clin. Lab. Invest. 36:113–118 (1976).
Author information
Authors and Affiliations
Additional information
Supported in part by National Institutes of Health Grant GM 16496 and the Patent Fund, School of Pharmacy, University of California, San Francisco.
Abstracted in part from a dissertation submitted by K. Sandy Pang to the Graduate Division, University of California, San Francisco, California, in partial fulfillment of the Doctor of Philosophy degree requirement.
Rights and permissions
About this article
Cite this article
Pang, K.S., Rowland, M. Hepatic clearance of drugs. I. Theoretical considerations of a “well-stirred” model and a “parallel tube” model. Influence of hepatic blood flow, plasma and blood cell binding, and the hepatocellular enzymatic activity on hepatic drug clearance. Journal of Pharmacokinetics and Biopharmaceutics 5, 625–653 (1977). https://doi.org/10.1007/BF01059688
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF01059688