Summary
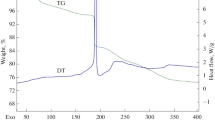
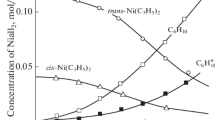
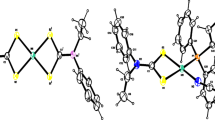
Complexes [NiL2]X2·nH2O (L=diethylenetriamine; n=O when X=CF3CO2 or CCl3CO2; n=1 when X=Cl or Br, and n=3 when X=0.5SO4 or 0.5SeO4) and NiLX2·nH2O (n=1 when X=Cl or Br; n=3 when X=0.5SO4 or 0.5SeO4) have been synthesised and investigated thermally in the solid state. NiLSO4 was synthesised pyrolytically in the solid state from [NiL2]SO4·[NiL2]X2 (X=Cl or Br) undergo exothermic irreversible phase transitions (242–282° C and 207–228° C; ΔH=−11.3 kJ mol−1 and −1.9 kJ mol−1 for [NiL2]Cl2 and [NiL2]Br2, respectively). [NiL2]-phenomenon (158–185° C; ΔH=2.0 kJ mol−1). NiLX2· nH2O (n=1 or 3) undergo simultaneous deaquation-isomerisation upon heating. All the complexes possess octahedral geometry.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
G. H. Searle and D. A. House,Aust. J. Chem.,40, 361 (1987). and refs. therein.
H. H. Schmidtke and Garthoff,Inorg. Chim. Acta,2, 357 (1968).
N. F. Curtis and H. K. J. Powell,J. Chem. Soc. A., 3069 (1968).
F. R. Keene and G. H. Searle,J. Chem. Soc., Chem. Comm., 893 (1968).
M. Dwyer and G. H. Searle,J. Chem. Soc., Chem. Comm., 726 (1972).
K. Okiyama, S. Sato and Y. Saito,Acta Cryst. B 35, 2389 (1979).
I. Lifschitz, J. G. Bos and K. M. Dijkema,Z. Anorg. Allg. Chem.,242, 97 (1930): P. Pfeiffer and H. Glaser,J. Prakt. Chem.,151, 134 (1938); R. Tsuchiya, A. Uehara and T. Yoshikuni,Inorg. Chem.,21, 590 (1982).
G. De, P. K. Biswas and N. Ray Chaudhuri,J. Chem. Soc., Dalton Trans., 2591 (1984).
A. Ghosh, G. De and N. Ray Chaudhuri,J. Thermal Anal.,30, 447 (1985).
G. De and N. Ray Chaudhuri,Transition Met. Chem.,10, 476 (1985).
A. Ghosh, G. De and N. Ray Chaudhuri,Transition Met. Chem.,11, 81 (1986).
A. Ghosh, G. De and N. Ray Chaudhuri,J. Chem. Res. (s), 104 (1987).
A. K. Mukherjee, M. Mukherjee, A. J. Welch, A. Ghosh, G. De and N. Ray Chaudhuri,J. Chem. Soc., Dalton Trans., 997 (1987).
A. Ghosh, S. Koner and N. Ray Chaudhuri,Thermochim. Acta,124, 297 (1988).
S. Roy, A. Ghosh and N. Ray Chaudhuri,Transition Met. Chem., TMC 1724, in press.
P. Paoletti, S. Biazini and M. Cannas,J. Chem. Soc., Chem. Comm., 513 (1969).
Tentative Proposals for Nomenclature of Absolute Configurations Concerned with Six-Coordinated Complexes Based on the Octahedron,Inorg. Chem.,9, 1 (1970).
D. R. Bloomquist and R. D. Willett,Coord. Chem. Rev.,47, 125 (1982).
R. J. Pylkii, R. D. Willett and H. W. Dodgen,Inorg. Chem.,23, 594 (1984).
L. Menabue, G. C. Pellacani, L. P. Battagila, A. B. Corradi, F. Sandrolini, A. Motori, R. J. Pylkki and R. D. Willett,J. Chem. Soc., Dalton Trans., 2187 (1984).
Y. Yoshikawa, N. Kato, Y. Kimura, S. Utsuko and G. N. Natu,Bull. Chem. Soc. Jpn.,59, 2123 (1986).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Koner, S., Ghosh, A. & Chaudhuri, N.R. Thermally induced isomerisation and decomposition of diethylenetriamine complexes of nickel(II) in the solid state. Transition Met Chem 13, 291–296 (1988). https://doi.org/10.1007/BF01025677
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF01025677