Abstract
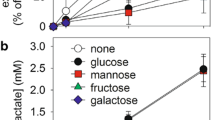
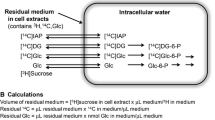
Uptake and metabolism of mannose were studied in astroglia-rich primary cultures derived from neonatal rat brains. A saturable component of mannose uptake was found with half-maximal uptake at 6.7±1.0 mM mannose. In addition, a non-saturable component dominated the uptake at high concentrations of mannose. Glucose, cytochalasin B, or phloretin in the incubation buffer inhibited the carrier-mediated uptake of mannose. Within the astroglial cells mannose is phosphorylated to mannose-6-phosphate. In cell homogenates, the KM value of mannose-phosphorylating activity was determined to be 24±7 μM. The Vmax value of this activity is only 40% that of glucose-phosphorylating activity. Mannose-6-phosphate was converted to fructose-6-phosphate by mannose-6-phosphate isomerase. The specific activity of this enzyme in homogenates of astroglial cultures was higher than that of hexokinase. Two products of mannose utilization in astroglial cells are glycogen and lactate. The amounts of each of these products increased with increasing concentrations of mannose. In contrast to the generation of lactate, that of glycogen from mannose was enhanced in the presence of insulin. In conclusion, we suggest that mannose is taken up into the cells of astroglia-rich primary cultures by the glial glucose transporter and is metabolized to fructose-6-phosphate within the astroglial cells.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
McIllwain, H., and Bachelard, H. S. 1985. Biochemistry and the Central Nervous System. 5th edn., Churchill Livingstone, New York.
Oldendorf, W. H.: 1971. Brain uptake of radiolabeled amino acids, amines, and hexoses after arterial injection. Am. J. Physiol. 221:1629–1639.
Pardridge, W. M., and Oldendorf, W. H. 1975. Kinetics of blood-brain barrier transport of hexoses. Biochim. Biophys. Acta 382:377–392.
Fuglsang, A., Lomholt, M., and Gjedde, A. 1986. Blood-brain transfer of glucose and glucose analogs in newborn rats. J. Neurochem. 46:1417–1428.
Chain, E. B., Rose, S. P. R., Masi, I., and Pocchiari, F. 1969. Metabolism of hexoses in rat cerebral cortex slices. J. Neurochem. 16:93–100.
Sloviter, H. A., and Kamimoto, T. 1970. The isolated, perfused rat brain preparation metabolizes mannose but not maltose. J. Neurochem. 17:1109–1111.
Betz, A. L., Csejtey, J., and Goldstein, G. W. 1979. Hexose transport and phosphorylation by capillaries isolated from rat brain. Am. J. Physiol. 236:C96-C102.
Vinters, H. V., Beck, D. W., Bready, J. V., Maxwell, K., Berliner, J. A., Hari, M. N., and Cancilla, P. A. 1985. Uptake of glucose analogues into cultured cerebral microvessel endothelium. J. Neuropathol. Exp. Neurol. 44:445–458.
Takakura, Y., Kuentzel, S. L., Raub, T. J., Davies, A., Baldwin, S. A., and Borchardt, R. T. 1991. Hexose uptake in primary cultures of bovine brain microvessel endothelial cells. I. Basic characteristics and effects of D-glucose and insulin. Biochim. Biophys. Acta 1070:1–10.
Hara, M., Matsuda, Y., Hirai, K., Okumura, N., and Nakagawa, H. 1989. Characteristics of glucose transport in neuronal cells and astrocytes from rat brain in primary culture. J. Neurochem. 52:902–908.
Heidenreich, K. A., Gilmore, P. R., and Garvey, W. T. 1989. Glucose transport in primary cultured neurons. J. Neurosci. Res. 22:397–407.
Sols, A., and Crane, R. K. 1954. Substrate specificity of brain hexokinase. J. Biol. Chem. 210:581–595.
Bruns, F. H., Noltmann, E., and Willemsen, A. 1958. Phosphomannose-isomerase. I. Über die Aktivitätsmessung und die Sulfhydryl- sowie die Metallabhängigkeit der Enzymwirkung in einigen tierischen Geweben. Biochem. Z. 330:411–420.
Kizer, D. E., and McCoy, T. A. 1960. Phosphomannose isomerase activity in a spectrum of normal and malignant rat tissues. Proc. Soc. Exp. Biol. Med. 103:772–774.
Hernandez, D., and de la Fuente, M. 1989. Mannose toxicity in Ehrlich ascites tumor cells. Biochem. Cell Biol. 67:311–314.
Dringen, R., and Hamprecht, B. 1992. Investigation on the function of glycogen in astrocytes. Biol. Chem. Hoppe-Seyler 372:951.
Cataldo, A. M., and Broadwell, R. D. 1986. Cytochemical identification of cerebral glycogen and glucose-6-phosphatase activity under normal and experimental conditions: I. Neurons and glia. J. Electron. Micros. Tech. 3:413–437.
Ignacio, P. C., Baldwin, B. A., Vijayan, V. K., Tait, R. C., and Gorin, F. A. 1990. Brain isozyme of glycogen phosphorylase: immunohistological localization within the central nervous system. Brain Res. 529:42–49.
Pfeiffer, B., Elmer, K., Roggendorf, W., Reinhart, P. H., and Hamprecht, B. 1990. Immunohistochemical demonstration of glycogen phosphorylase in rat brain slices. Histochemistry 94:73–80.
Pfeiffer, B., Meyermann, R., and Hamprecht, B. 1992. Immunohistochemical co-localization of glycogen phosphorylase with the astroglial markers glial fibrillary acidic protein and S-100 protein in rat brain sections. Histochemistry 97:405–412.
Reinhart, P. H., Pfeiffer, B., Spengler, S., and Hamprecht, B. 1990. Purification of glycogen phosphorylase from bovine brain and immunocytochemical examination of rat primary cultures using monoclonal antibodies raised against this enzyme. J. Neurochem. 54:1474–1483.
Dringen, R., and Hamprecht, B. 1991. Modulation of astrocytic glycogen by hexoses. J. Neurochem. 57, Suppl.:S127D.
Hamprecht, B., and Löffler, F. 1985. Primary glial cultures as a model for studying hormone action. Meth. Enzymol. 109:341–345.
Stahl, B., Wiesinger, H., and Hamprecht, B. 1989. Characteristics of sorbitol uptake in rat glial primary cultures. J. Neurochem. 53:665–671.
Dringen, R., and Hamprecht, B. 1992. Glucose, insulin and insulin-like growth factor I regulate the glycogen content of astroglia-rich primary cultures. J. Neurochem. 58:511–517.
Bradford, M. M. 1976. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 72:248–254.
Lowry, O. H., Rosebrough, N. J., Farr, A. L., and Randall, R. J. 1951. Protein measurement with the Folin phenol reagent. J. Biol. Chem. 193:265–275.
Roeder, L. M., Williams, I. B., and Tildon, J. T. 1985. Glucose transport in astrocytes: Regulation by thyroid hormone. J. Neurochem. 45:1653–1657.
Tildon, J. T., and Roeder, L. M. 1988. Transport of 3-hydroxy [3-14C]butyrate by dissociated cells from rat brain. Am. J. Physiol. 255:C133-C139.
Hanes, C. S. 1932. CLXVII. Studies on plant amylases. I. The effect of starch concentration upon the velocity of hydrolysis by the amylase of germinated barley. Biochem. J. 26:1406–1421.
Bloch, R. 1973. Inhibition of glucose transport in the human erythrocyte by cytochalasin B. Biochemistry 12:4799–4801.
LeFevre, P. G., and Marshall, J. K. 1959. The attachment of phloretin and analogues to human erythrocytes in connection with inhibition of sugar transport. J. Biol. Chem. 234:3022–3026.
Raff, M. C., Fields, K. L., Hakomori, S. I., Mirsky, R., Pruss, R. M., and Winter, J. 1979. Cell-type-specific markers for distinguishing and studying neurons and the major classes of glial cells in culture. Brain Res. 174:283–308.
Roeder, L. M., Williams, I. B., and Tildon, J. T. 1985. Glucose transport in astrocytes: regulation by thyroid hormone. J. Neurochem. 45:1653–1657.
Gould, G. W., Thomas, H. M., Jess, T. J., and Bell, G. I. 1991. Expression of human glucose transporters in Xenopus oocytes: Kinetic characterization and substrate specificities of the erythrocyte, liver, and brain isoforms. Biochemistry 30:5139–5145.
Kahlenberg, A., and Dolansky, D. 1972. Structural requirements of D-glucose for its binding to isolated human erythrocyte membranes. Can. J. Biochem. 50:638–643.
Renner, E. D., Plagemann, P. G. W., and Bernlohr, R. W. 1972. Permeation of glucose by simple and facilitated diffusion by Novikoff rat hepatoma cells in suspension culture and its relationship to glucose metabolism. J. Biol. Chem. 247:5765–5776.
Germinario, R. J., Chang, Z., Manuel, S., and Oliveira, M. 1985. Control of sugar transport in human fibroblasts independent of glucose metabolism or carrier-substrate interaction. Biochem. Biophys. Res. Commun. 128:1418–1424.
Neefjes, J. J., Lindhout, J., Broxterman, H. J. G., van der Marel, G. A., van Boom, J. H., and Ploegh, H. L. 1989. Non-carrier-mediated uptake of the mannosidase I inhibitor 1-deoxymannojirimycin by K562 erythroleukemic cells. J. Biol. Chem. 264:10271–10275.
Barnett, J. E. G., Holman, G. D., and Munday, K. A. 1973. Structural requirements for binding to the sugar-transport system of the human erythrocyte. Biochem. J. 131:211–221.
Lusk, J. A., Manthorpe, C. M., Kao-Jen, J., and Wilson, J. E. 1980. Predominance of the cytoplasmic form of brain hexokinase in cultured astrocytes. J. Neurochem. 34:1412–1420.
Nigam, V. N. 1969. Pathways of glycogen synthesis in Novikoff ascites-hepatoma cells. Biochem. J. 115:315–322.
Garfinkel, L., Garfinkel, D., Matsiras, P., and Matschinsky, B. 1987. Kinetic properties of hexokinase as assembled with a microcomputer data base. Biochem. J. 244:351–357.
Snyder, C. D., and Wilson, J. E. 1983. Relative levels of hexkinase in isolated neuronal, astrocytic, and oligodendroglial fractions from rat brain. J. Neurochem. 40:1178–1181.
Swanson, R. A., Yu, A. C. H., Chan, P. H., and Sharp, F. R. 1990. Glutamate increases glycogen content and reduces glucose utilization in primary astrocyte cultures. J. Neurochem. 54:490–496.
Freinkel, N., Lewis, N. J., Akazawa, S., Roth, S. I., and Gorman, L. 1984. The honeybee syndrome-implications of the teratogenicity of mannose in rat-embryo culture. N. Engl. J. Med. 310:223–230.
Buchanan, T., Freinkel, N., Lewis, N. J., Metzger, B. E., and Akazawa, S. 1985. Fuel-mediated teratogenesis. Use of D-mannose to modify organogenesis in the rat embryo in vivo. J. Clin. Invest. 75:1927–1934.
De la Fuente, M., Penas, P. F., and Sols, A. 1986. Mechanism of mannose toxicity. Biochem. Biophys. Res. Commun. 140:51–55.
Deuel, Jr. H. J., Hallman, L. F., Murray, S., and Hilliard, J. 1938. Studies on ketosis: XV. The comparative metabolism of d-mannose and d-glucose. J. Biol. Chem. 125:79–84.
Dringen, R., and Hamprecht, B. 1990. Modulation of glycogen content in astrocytes. Biol. Chem. Hoppe-Seyler 371:780–781.
Stang, R. H. C., and Bachelard, H. S. 1971. Effect of insulin on levels and turnover of intermediates of brain carbohydrate metabolism in vivo. J. Neurochem. 18:1799–1807.
Sloviter, H. A., and Yamada, H. 1971. Absence of direct action of insulin on metabolism of the isolated perfused rat brain. J. Neurochem. 18:1269–1274.
Probst, I., and Unthan-Fechner, K. 1985. Activation of glycolysis by insulin with a sequential increase of the 6-phosphofructo-2-kinase activity, fructose-2,6-bisphosphate level and pyruvate kinase activity in cultured rat hepatocytes. Eur. J. Biochem. 153:347–353.
Schudt, C. 1979. Regulation of glycogen synthesis in rat-hepatocyte cultures by glucose, insulin and glucocorticoids. Eur. J. Biochem. 97:155–160.
Agius, L., Peak, M., and Alberti, K. G. M. M. 1990. Regulation of glycogen synthesis from glucose and gluconeogenic precursors by insulin in periportal and perivenous rat hepatocytes. Biochem. J. 266:91–102.
Lopes-Cardozo, M., Larsson, O. M., and Schousboe, A. 1986. Acetoacetate and glucose as lipid precursors and energy substrates in primary cultures of astrocytes and neurons from mouse cerebral cortex. J. Neurochem. 46:773–778.
Edmond, J., Robbins, R. A., Bergstrom, J. D., Cole, R. A., and de Vellis, J. 1987. Capacity for substrate utilization in oxidative metabolism by neurons, astrocytes, and oligodendrocytes from developing brain in primary culture. J. Neurosci. Res. 18:551–561.
Wiesinger, H., Thiess, U., and Hamprecht, B. 1990. Sorbitol pathway activity and utilization of polyols in astroglia-rich primary cultures. Glia 3:277–282.
Auestadt, N., Korsak, R. A., Morrow, J. W., and Edmond, J. 1991. Fatty acid oxidation and ketogenesis by astrocytes in primary culture. J. Neurochem. 56:1376–1386.
Vicario, C., Arizmendi, C., Malloch, G., Clark, J. B., and Medina, J. M. 1991. Lactate utilization by isolated cells from early neonatal rat brain. J. Neurochem. 57:1700–1707.
Hawkins, R. A., and Mans, A. M. 1983. Intermediary metabolism of carbohydrates and other fuels. Pages 259–294,in A. Lajtha (ed.), Handbook of Neurochemistry, 2nd edn., Plenum Press, New York.
Akazawa, S., Metzger, B. E., and Freinkel, N. 1986. Relationship between glucose and mannose during late gestation in normal pregnancy and pregnancy complicated by diabetes mellitus: concurrent concentrations in maternal plasma and amniotic fluid. J. Clin. Endocrinol. Metab. 62:984–989.
Herman, R. H. 1971. Mannose metabolism. I. Am. J. Clin. Nutr. 24:488–498.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Dringen, R., Bergbauer, K., Wiesinger, H. et al. Utilization of mannose by astroglial cells. Neurochem Res 19, 23–30 (1994). https://doi.org/10.1007/BF00966724
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00966724