Abstract
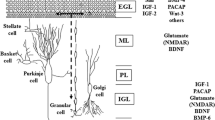
Chronic exposure of dissociated cerebellar cultures to 50μM kainate results in a complete loss of [3H]-GABA release which is a marker of GABAergic interneurons. No loss of granule cells was found and the glutamatergic nature of the granule cells appeared unaltered by the kainate treatment, since evoked release of [3H]-d-aspartate was maintained after kainate exposure. Glial cells in such cultures are virtually eliminated by treatment with an antimitotic such as cytarabin. In consequence a pure culture of cerebellar granule cells virtually free of stellate, basket and glial cells may be obtained by a combined chronic treatment of the cultures with kainate and cytarabin.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Lasher, R. S., and Zagon, I. S. 1972. The effect of potassium on neuronal differentiation in cultures of dissociated newborn rat cerebellum. Brain Res. 41:482–438.
Messer, A. 1977. The maintenance and identification of mouse cerebellar granule cells in monolayer culture. Brain Res. 103:1–12.
Currie, D. N. 1980. Identification of cell type by immunofluorescence in defined cell cultures of cerebellum. Pages 75–87,in Giacobini, E., Vernadakis, A., and Shahar, A. (eds.), Tissue Culture in Neurobiology, Raven Press, New York.
Drejer, J., Larsson, O. M., Kvamme, E., Svenneby, G., Hertz, L., and Schousboe, A. 1985. Ontogenetic development of glutamate metabolizing enzymes in cultured cerebellar granule cells and in cerebellum in vivo. Neurochem. Res. 10:49–62.
Drejer, J., Larsson, O. M., and Schousboe, A. 1982. Characterization ofl-glutamate uptake into and release from astrocytes and neurons cultured from different brain regions. Exp. Brain Res. 47:259–269.
Gallo, V., Ciotti, M. T., Coletti, A., Aloisi, F., and Levi, G. 1982. Selective release of glutamate from cerebellar granule cells differentiating in culture. Proc. Natl. Acad. Sci. USA 79:7919–7923.
Nicoletti, F., Wroblewski, J. T., Novelli, A., Alho, H., Guidotti, A., and Costa, E. 1986. The activation of inositol phospholipid metabolism as a signal-transducing system for excitatory amino acids in primary cultures of cerebellar granule cells. J. Neurosci. 6:1905–1911.
Palaiologos, G., Hertz, L., and Schousboe, A. 1988. Evidence that aspartate aminotransferase activity and ketodicarboxylate carrier function are essential for biosynthesis of transmitter glutamate. J. Neurochem. 51:317–320.
Lasher, R. S. 1974. The uptake of [3H]-GABA and differentiation of stellate neurons in cultures of dissociated postnatal rat cerebellum. Brain Res. 69:235–254.
Currie, A. N., and Dutton, G. R. 1980. [3H]-GABA uptake as a marker for cell type in primary cultures of cerebellum and olfactory bulb. Brain Res. 100:473–481.
Pearce, B. R., Currie, D. N., Beale, R., Dutton, G. R. 1981. Potassium-stimulated, calcium-dependent release of [3H] GABA from neuron-and gliaenriched cultures of cells dissociated from rat cerebellum. Brain Res. 206:485–489.
Gallo, V., Ciotti, M. T., Aloisi, F., Levi, G. 1986. Developmental features of rat cerebellar neural cells cultured in a chemically defined medium. J. Neurosci. Res. 15:289–301.
Aloisi, F., Ciotti, M. T., and Levi, G. 1985. Characterization of GABAergic neurons in cerebellar primary cultures and selective neurotoxic effects of a serum fraction. J. Neurosci. 5:2001–2008.
Herndon, R. M., and Coyle, J. T. 1977. Selective destruction of neurons by a transmitter agonist. Science, N.Y. 198:71–72.
Tran, V. T., and Snyder, S. H. 1979. Amino acid neurotransmitter candidates in rat cerebellum: selective effects of kainic acid lesions. Brain Res. 167:345–353.
Seil, F. J., Blank, N. K., and Leiman A. L. 1979. Toxic effects of kainic acid on mouse cerebellum in tissue culture. Brain Res. 161:253–265.
Messer, A., and Maskin, P. 1980. Short-term effects of kainic acid on rat cerebellar cells in monolayer cultures. Neurosci. Lett. 19:173–177.
Garthwaite G., and Garthwaite, J. 1984. Differential sensitivity of rat cerebellar cells in vitro to the neurotoxic effects of excitatory amino acid analogues. Neurosci. Lett. 48:361–367.
Drejer, J., Honoré, T., and Schousboe, A. 1987. Excitatory amino acid-induced release of3H-GABA from cultured mouse cerebral cortex interneurons. J. Neurosci. 7:2910–2916.
Drejer, J., Honoré, T., Meier, E., and Schousboe, A. 1986. Pharmacologically distinct glutamate receptors on cerebellar granule cells. Life Sci. 38:2077–2085.
Mosmann, T. 1983. Rapid colorimetric assay for cellular growth and survival: application to proliferation and cytotoxicity assays. J. Immunol. Methods 65:55–63.
Schousboe, A., Meier, E., Drejer, J., and Hertz, L. 1989. Preparation of cultures of mouse (rat) cerebellar granule cells.in Shahar, A., DeVellis, J., Vernadakis, A., and Haber, B. (eds), A dissection and tissue culture manual for the nervous system, Alan R. Liss, New York, in press.
Schousboe, A., and Pasantes-Morales, H. 1989 Potassium-stimulated release of3H-taurine from cultured GABAergic and glutamatergic neurons. J. Neurochem. 53: in press.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Drejer, J., Schousboe, A. Selection of a pure cerebellar granule cell culture by kainate treatment. Neurochem Res 14, 751–754 (1989). https://doi.org/10.1007/BF00964953
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00964953