Abstract
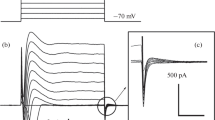
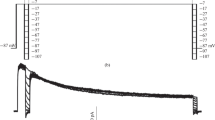
The existence and properties of volume-activated Cl− currents were studied in 15 different cell types (endothelium: human umbilical vein, human aorta, bovine pulmonary artery; fibroblasts: Swiss 3T3, L, C3H 10T1/2 and COS-1; epithelium: KB3, HeLa and A6; blood cells: RBL-2H3 and Jurkat; endothelioma cells derived from both subcutaneous and thymic hemangiomas; skin: IGR1 melanoma). Volume-activated Cl− currents with common characteristics, i.e. small conductance, outward rectification, higher permeability for iodide than for chloride and sensitivity to block by 5-nitro-2-(3-phenylpropylamino)benzoic acid (NPPB) could be elicited in all cells. The block of this current by tamoxifen and dideoxyforskolin is different for the various cell types, as well as the time course and the amplitude of the responses induced by repetitive applications of hypotonicity. Volume-activated Cl− channels with similar biophysical properties are therefore widespread among mammalian cells. This may reflect either a single Cl− channel that is ubiquitously expressed or a family of functionally related Cl− channels with cell specific expression patterns.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Baer CE (1994) Drugs transported by P-glycoprotein inhibit a 40 pS outwardly rectifying chloride channel. Biochem Biophys Res Commun 200: 513–521
Diaz M, Valverde MA, Higgins CF, Rucareanu C, Sepulveda FV (1993) Volume-activated chloride channels in HeLa cells are blocked by verapmil and dideoxyforskolin. Pflügers Arch 422: 347–353
Doroshenko P (1991) Second messengers mediating activation of chloride current by intracellular GTPγS in bovine chromaffin cells. J Physiol (Lond) 436: 725–738
Doroshenko P, Neher E (1992) Volume-sensitive conductance in bovine chromaffin cell membrane. J Physiol (Lond) 449: 197–218
Doroshenko P, Penner R, Neher E (1991) A novel chloride conductance in the membrane of bovine chromaffin cells activated by intracellular GTPγS. J Physiol (Lond) 436: 711–724
Dubois J-M (1991) Role of potassium channels in mitogenesis. Prog Biophys Mol Biol 59: 1–21
Gill DR, Hyde SC, Higgins CF, Valverde MA, Minteng GM, Sepulveda FV (1992) Separation of drug transport and chloride channel functions of human multidrug resistance P-glycoprotein. Cell 71: 23–32
Hofer AM, Machen TE (1993) Technique for in situ measurement of calcium in intracellular inositol 1,4,5 triphosphatesensitive stores using the fluorescent indicator mag-fura-2. Proc Nat Acad Sci USA 90: 2598–2602
Horn R, Marty A (1988) Muscarinic activation of ionic currents measured by a new whole-cell recording method. J Gen Physiol 92: 145–159
Kelly MEM, Dixon SJ, Sims SM (1994) Outwardly rectifying chloride current in rabbit osteoclasts is activated by hyposmotic stimulation. J Physiol (Lond) 475: 377–389
Kunzelmann K, Kubitz R, Grolik M, Wart R, Greger R (1992) Small-conductance Cl− channels in HT29 cells: activation by Ca2+, hypotonic cell swelling and 8-Br-cGMP. Pflügers Arch 421: 238–246
Lewis RS, Ross PE, Cahalan MD (1993) Chloride channels activated by osmotic stress in T lymphocytes. J Gen Physiol 101: 801–826
Maranaka Y, Eaton DC (1990) Chloride channels in the apical membrane of a distal nephron A6 cell line. Am J Physiol 258: C352-C368
Mintenig GM, Valverde MA, Sepulveda FV, Gill DR, Hyde SC, Higgins CF (1994) Specific inhibitors distinguish the chloride channel and drug transporter functions associated with the human multidrug resistance P-glycoprotein. Receptors Channels 1: 305–313
Nilius B, Oike M, Zahradnik I, Droogmans G (1994) Activation of Cl− channels by hypotonic stress in human endothelial cells. J Gen Physiol 103:787–805
Nilius B, Sehrer J, Droogmans G (1994) Permeation properties and modulation of volume-activated Cl− currents in human endothelial cells. Br J Pharmacol (in press)
Nilius B, Sehrer J, Viana F, Raeymaekers L, De Greef C, Droogmans G (1994) Properties of volume-activated chloride channels in various cell types. Pflügers Arch [Suppl] 427: R59
Oike M, Droogmans G, Nilius B (1994) Mechanosensitivity of endothelial cells is mediated by arachidonic acid. Proc Nat Acad Sci USA 91:2940–2944
Oike M, Droogmans G, Nilius B (1994) The volume-activated chloride current in human endothelial cells depends on intracellular ATP. Pflügers Arch 427:184–186
Parker JC (1993) In defense of cell volume? Am J Physiol 265:C1191-C1200
Paulmichl M, Gschwentner M, Wöll E, Schmarda A, Ritter M, Kanin G, Ellemuter H, Waitz W, Deetjen P (1993) Insight into structure-function relation of chloride channels. Cell Physiol Biochem 3: 374–387
Schwiebert EM, Mills JW, Stanton BA (1994) Actin-based cytoskelton regulates a chloride channel and volume in a renal cortical collecting duct cell line. J Biol Chem 269: 7081–7089
Valverde MA, Diaz M, Sepulveda M, Gill DG, Hyde SC, Higgins CF (1992) Volume regulated chloride channels are associated with the human multidrug-resistance P-glycoprotein. Nature 355: 830–833
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Nilius, B., Sehrer, J., Viana, F. et al. Volume-activated Cl− currents in different mammalian non-excitable cell types. Pflügers Arch. 428, 364–371 (1994). https://doi.org/10.1007/BF00724520
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00724520