Abstract
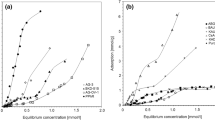
Aqueous 1,1,2-trichloroethene (TCE) adsorption isotherms were obtained on Ambersorb1® 563 and 572 adsorbents and Filtrasorb2® 400 granular activated carbon (GAC). The data for Ambersorb 563 adsorbent covers TCE concentrations from 0.0009 to 600 mg/L. The data for each adsorbent was fit to 15 isotherm equations to determine an optimum equation.
The best equation for the TCE adsorption isotherms is the Dubinin-Astakov (DA) isotherm. The DA isotherm coefficients were used to estimate the TCE micropore volume and the adsorption potential distribution. For each adsorbent, the TCE micropore volume is equivalent to the N2 porosimetry micropore volume. The mean adsorption potential is 18.8, 13.0, and 8.9 kJ/mol, with coefficients of variation of 0.37, 0.53, and 0.67, for Ambersorb 563 and 572 adsorbents and Filtrasorb 400 GAC, respectively. Thus, Ambersorb 563 adsorbent has the most energetic and most homogeneous adsorption volume, while Filtrasorb 400 GAC has the least energetic and most heterogeneous adsorption volume. For these reasons, Ambersorb 563 adsorbent has the highest TCE capacity at low concentrations, whereas Filtrasorb 400 GAC has the highest TCE capacity at high concentrations. The performance of Ambersorb 572 adsorbent is generally intermediate to the other two adsorbents.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Abbreviations
- A :
-
Adsorption potential (J mol−1)=R×T×ln (c s /c).
- BET:
-
Brunauer-Emmett-Teller isotherm function, Eq. (I.3).
- c :
-
Concentration (mg L−1)
- c s :
-
Solubility in water (1370 mg L−1 for TCE at 25°C (Riddick et al., 1986))
- DA:
-
Dubinin and Astakov isotherm function, Eq. (I.9).
- E :
-
Characteristic energy (J mol−1).
- EI-n:
-
n-th order exponential isotherm function, Eq. (I.12).
- HK:
-
Höll and Kirch isotherm function, Eq. (I.10).
- K :
-
Strength of adsorption parameter (dimensions depend on equation).
- m :
-
Adsorbent mass (g).
- MJ:
-
Marczewski and Jaroniec isotherm function, Eq. (I.11).
- n :
-
Order of EI function.
- q :
-
Adsorption capacity (mg g−1).
- q ∞ :
-
Maximum capacity at either infinite concentration or at the solubility limit (mg g−1).
- q m :
-
Capacity at monolayer coverage (mg g−1).
- R :
-
Gas constant, 8.314510 J mol−1 K−1
- RP:
-
Radke and Prausnitz isotherm function, Eq. (I.7).
- Slope:
-
Slope of isotherm function (L g−1).
- T :
-
Temperature (K)
- V :
-
Volume (L)
- X(A):
-
Adsorption potential distribution (mol J−1).
- α e :
-
Coefficient of thermal expansion (0.00115 for TCE at 25°C (Riddick et al., 1986))
- α, β :
-
Heterogeneity or shape of isotherm parameters (dimensions depend on equation).
- η, υ :
-
Exponent.
- ΔH :
-
Differential molar enthalpy of adsorption (J mol−1).
- ΔS :
-
Differential molar entropy of adsorption (J mol−1 K−1).
- γ :
-
Relative adsorption or fraction of micropores filled (dimensionless).
- σ :
-
Standard deviation of subscripted variable.
References
Bansal, R.C., J.B. Donnet, and F. Stoeckli,Active Carbon, pp. 137–147 (Marcel Dekker, NY, 1988).
Benz, M., B. Haist, and G. Zimmer, “Micropollutant Behavior in Activated Carbon Filters,”Third Int. Conf. Fund. Adsorp. 1989, 121–130 (1991).
Bering, B.P., M.M. Dubinin, and V.V. Serpinsky, “On Thermodynamics of Adsorption in Micropores,”J. Coll. Interface Sci.,38, 185–194 (1972).
Bevington, P.R.,Data Reduction and Error Analysis for the Physical Sciences, a) Chapter 10, b) pp. 98–99, McGraw Hill, NY, (1969).
Browne, T.E. and Y. Cohen, “Aqueous-Phase Adsorption of Trichloroethene and Chloroform onto Polymeric Resins and Activated Carbon,”Ind. Eng. Chem. Res.,29, 1338–1345 (1990).
Derylo-Marczewska, A. and M. Jaroniec, “Adsorption of Organic Solutes from Dilute Solutions on Solids,”Surf. and Colloid Sci.,14, 301–379 (1987).
Dobbs, R.A. and J.M. Cohen,Carbon Adsorption Isotherms for Toxic Organics, EPA/600/8-80/023, USEPA, Cincinnati, OH (1980).
Jaroniec, M., and R. Madey,Physical Adsorption on Heterogeneous Solids, Elsevier, NY, 1988.
Jaroniec, M., R. Madey, and J. Choma, “Thermodynamic Functions Associated with the Exponential Isotherm Equation for Gas Adsorption on Heterogeneous Microporous Solids,”J. Coll Interface Sci.,126, 69–73 (1988).
Jossens, L., J.M. Prausnitz, W. Fritz, E.U. Schlünder, and A.L. Myers, “Thermodynamics of Multi-Solute Adsorption from Dilute Aqueous Solutions,”Chem. Eng. Sci.,33, 1097–1106 (1978).
Nirmalakhandan, N.N., and R.E. Speece, “Prediction of Activated Carbon Adsorption Capacities for Organic Vapors Using Quantitative Structure-Activity Relationship Methods,”Environ. Sci. Technol.,27, 1512–1516 (1993).
Pasha Publications, Inc.,1989 Guide to Superfund Sites, Arlington, VA, 1989.
Randtke, S.J. and V.L. Snoeyink, “Evaluating GAC Adsorptive Capacity,”J. Amer. Water Works Assoc.,75, 406–413 (1983).
Riddick, J.A., W.B. Bunger, and T.K. Sakano,Organic Solvents: Physical Properties and Methods of Purification, 4th Ed., p. 521, John Wiley & Sons, NY, 1986.
Sircar, S. “Estimation of Micropore Volume of Activated Carbons from Vapor Adsorption Isotherms,”Proceedings of the XVIIIth Biennial Conference on Carbon, pp. 92–93 (July 19–24, 1987).
Sontheimer, H., J.C. Crittenden, and R.S. Summers, a) Chapter 3, b) Appendix A.1,Activated Carbon for Water Treatment, Distributed by AWWA Research Foundation, Denver, CO, 1985.
Speth, T.F., “Predicting Equilibria for Single Solute and Multicomponent Aqueous Adsorption onto Activated Carbon,” Thesis submitted in partial fulfillment of a Master of Science degree in Civil Engineering, Michigan Technological University, Houghton, MI (1986).
Speth, T.F. and R.J. Miltner, “Technical Note: Adsorption Capacity of GAC for Synthetic Organics,”J. Amer. Water Works Assoc.,82, 72–75 (1990).
Urano, K., E. Yamamoto, M. Tonegawa, and K. Fujie, “Adsorption of Chlorinated Organic Compounds on Activated Carbon from Water,”Wat. Res.,25, 1459–1464 (1991).
Weber, Jr., W.J., M.C. Carter, K.P. Olmstead, and L.E. Katz,Evaluation of the MIDAS System for Development of Design Data for GAC Adsorbers, EPA/600/2-91/048 (1991).
Westrick, J.J., J.M. Mello, and R.F. Thomas, “The Groundwater Supply Survey,”J. Am. Water Works Assoc. 76, 52–59 (1984).
Wood, G.O., “Activated Carbon Adsorption Capacities for Vapors,”Carbon,30, 593–599 (1992).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Parker, G.R. Optimum isotherm equation and thermodynamic interpretation for aqueous 1,1,2-trichloroethene adsorption isotherms on three adsorbents. Adsorption 1, 113–132 (1995). https://doi.org/10.1007/BF00705000
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00705000