Summary
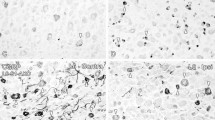
In a mouse model of genital herpes simplex virus type 2 (HSV-2) infection, roots of the lower spinal cord were examined 5 days to 6 months after inoculation. Using immunoperoxidase methods on paraffin sections, viral antigen was found in sensory ganglia, their proximal roots and distal nerves on days 5 and 6 after infection. In Epon sections, most mice had focal sensory root abnormalities in lower thoracic, lumbar or sacral levels. At days 7 and 10, lesions showed chiefly nerve fiber degeneration, particularly of large myelinated fibers. At 2 weeks, lesions contained relatively large bundles of small unmyelinated fibers with immature axon-Schwann cell relationships. From 3 to 6 weeks, lesions again contained many more small unmyelinated fibers than normal but, in increasing proportions, axons in bundles were isolated from their neighbors by Schwann cell cytoplasm, and Schwann cells having 1∶1 relationships with axons showed mesaxon or thin myelin sheath formation. At later times, the proportion of small unmyelinated axons decreased in parallel with increased numbers of small myelinated axons. By 6 months, affected roots showed a relative reduction in large myelinated fibers, increased proportions of small myelinated fibers and Schwann cell nuclei. Numbers of unmyelinated fibers were reduced relative to 3- to 6-week lesions. Axonal degeneration and regeneration appears to be the chief pathological change in sensory roots in this model. If regenerated fibers arise from latently infected neurons, then establishment of latency is not a relatively silent event, but is associated with major long-lasting, morphologically detectable effects.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Aguayo AJ, Peyronnard JM, Bray GM (1973) A quantitative ultrastructural study of regeneration from isolated proximal stumps of transected unmyelinated nerves. J Neuropathol Exp Neurol 32:256–270
Baringer JR, Griffith JF (1970) Experimental herpes simplex encephalitis: early neuropathological changes. J Neuropathol Exp Neurol 29:89–104
Cook ML, Stevens JG (1973) Pathogenesis of herpetic neuritis and ganglionitis in mice: evidence for intra-axonal transport of infection. Infect Immunol 7:272–288
Deatly AM, Spivack JG, O'Boyle DR, Fraser NW (1988) Latent herpes simplex virus type 1 transcripts in peripheral and central nervous tissues of mice map to similar regions of the viral genome. J Virol 62:749–756
Georgsson G, Martin JR, Stoner GL, Webster HD (1987) Virus spread and initial pathological changes in the nervous system in genital herpes simplex virus type 2 infection in mice. Acta Neuropathol (Berl) 72:377–388
Itoyama Y, Sekizawa T, Openshaw H, Kogure K, Kuroiwa Y (1984) Immunocytochemical localization of herpes simplex virus antigen in the trigeminal ganglia of experimentally infected mice. J Neurol Sci 66:67–75
Kristensson K, Vahlne A, Persson LA, Lycke E (1978) Neural spread of herpes simplex virus types 1 and 2 in mice after corneal or subcutaneous (footpad) inoculation. J Neurol Sci 35:331–340
Martin JR, Stoner GL (1984) The nervous system in genital herpes simplex virus type 2 infection. Lethal panmyelitis or nonlethal demyelinative myelitis or meningitis. Lab Invest 51:556–566
Martin JR, Suzuki S (1987) Inflammatory sensory polyradiculopathy and reactivated peripheral nervous system infection in a genital herpes model. J Neurol Sci 79:155–171
Peters A, Palay SL, Webster HD (1976) The fine structure of the nervous system. Saunders, Philadelphia, pp 112–116
Roizman B, Batterson W (1985) Herpesviruses and their replication. In: Fields BN (ed) Virology. Raven Press, New York, pp 497–526
Soffer D, Martin JR (1988) Remyelination of central nervous system lesions in experimental genital herpes simplex virus infection. J Neurol Sci 86:83–95
Stevens JG, Wagner EK, Devi-Rao GB, Cook ML, Feldman LT (1987) RNA complementary to a herpes virus alpha gene mRNA is prominent in latently infected neurons. Science 235:1056–1059
Tenser RB, Dawson M, Ressel SJ, Dustan ME (1982) Detection of herpes simplex virus mRNA in latently infected trigeminal ganglion neurons by in situ hybridization. Ann Neurol 11:285–291
Townsend JJ (1981) The demyelinating effect of corneal HSV infections in normal and nude (athymic) mice. J Neurol Sci 50:435–441
Townsend JJ, Baringer JR (1978) Central nervous system susceptibility to herpes simplex infection. J Neuropathol Exp Neurol 37:255–262
Townsend JJ, Collins PK (1986) Peripheral nervous system demyelination with herpes simplex virus. J Neuropathol Exp Neurol 45:419–425
Trapp BD, Itoyama Y, Sternberger NH, Quarles RH, Webster HD (1981) Immunocytochemical localization of Po proteins in golgi complex membranes and myelin of developing rat Schwann cells. J Cell Biol 90:1–6
Webster HD, Martin JR, O'Connell MF (1973) The relationships between interphase Schwann cells and axons before myelination: a quantitative electron microscopic study. Dev Biol 32:401–416
Yamada KM, Spooner BS, Wessells NK (1971) Ultrastructure and function of growth cones and axons of cultured nerve cells. J Cell Biol 49:614–635
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Soffer, D., Martin, J.R. Axonal degeneration and regeneration in sensory roots in a genital herpes model. Acta Neuropathol 77, 605–611 (1989). https://doi.org/10.1007/BF00687888
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00687888