Abstract
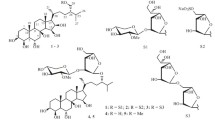
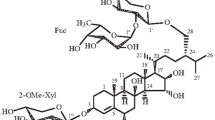
The structures of two triterpene oligosides from the holothurianStichopus cloronotus (Brandt) have been established; they are: 23ξ-acetoxy-3β-[O-β-D-quinovopyranosyl-(1→2)-β-D-xylopyranosyloxy]holost-7(8)-ene and 23ξ-acetoxy-3β-[O-β-D-glucopyranosyl-(1→2)-β-D-xylopyranosyloxy]holost-7(8)-ene.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Literature cited
I. Kitagawa, H. Yamanaka, M. Kobayashi, I. Yosioka, and T. Sugawara, Chem. Pharm. Bull.,26, 3722 (1978).
I. Kitagawa, T. Nishino, T. Matsuno, H. Akutsu, and Y. Kyogoku, Tetrahedron Lett., No. 11, 985 (1978).
I. Kitagawa, T. Nishino, and Y. Kyogoku, Tetrahedron Lett., No. 16, 1419 (1979).
I. Rothberg, B. M. Tursch, and C. Djerassi, J. Org. Chem.,38, 209 (1973).
V. A. Stonik, V. F. Sharypov, A. I. Kalinovskii, and G. B. Elyakov, Dokl. Akad. Nauk SSSR,245, 1133 (1979).
V. M. Easterwood and B. S. L. Heeff, Svensk. Pappers.,23, 768 (1969).
S. Hakomori, J. Biochem. (Tokyo),55, 205 (1964).
P. A. Gorin and M. Mazurek, Can. J. Chem.,53, 1212 (1975).
A. I. Kalinovskii, V. F. Sharypov, V. A. Stonik, A. K. Dzizenko, and G. B. Elyakov, Bioorg. Khim.,6, 86 (1980).
P. Karrer and A. Boettcher, Helv. Chim. Acta,36, 571 (1953).
Additional information
Pacific Ocean Institute of Bioorganic Chemistry, Far Eastern Scientific Center, Academy of Sciences of the USSR, Vladivostok. Translated from Khimiya Prirodnykh Soedinenii, No. 2, pp. 181–184, March–April, 1981.
Rights and permissions
About this article
Cite this article
Sharypov, V.F., Chumak, A.D., Stonik, V.A. et al. Glycosides of marine invertebrates. X. The structure of stichoposides A and B from the holothurianStichopus cloronotus . Chem Nat Compd 17, 139–142 (1981). https://doi.org/10.1007/BF00634731
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00634731