Summary
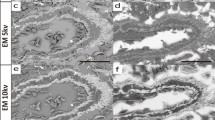
We investigated the localization of amylase in normal human lungs and the female genital tract using immunohistochemical and histochemical methods. Immunohistochemical procedures were applied to formaldehydefixed, paraffin-embedded specimens as well as to cryostat sections of periodate/lysine/paraformaldehyde (PLP)-fixed tissues. The starch-substrate-film method was used for the histochemical investigation of unfixed frozen sections. Amylase immunoreactivity was observed in ciliated epithelial cells of the bronchus and in serous cells of the bronchial glands but not in the alveolar epithelium. Immunoreactive amylase was also found in the cytoplasm of the ciliated epithelium of the fallopian tubes, especially in the apical part of the cytoplasm and in ciliary vesicles. Immunoreactive amylase was also found to be present in the surface epithelial cells and glands of the uterine cervix, as well as in the superficial part of the endometrial glands. The distribution of amylase activity revealed using histochemistry was similar to that observed in cryostat sections of PLP-fixed tissues after immunohistochemical staining. Amylase amtigenicity was better preserved in cryostat sections of PEP-fixed materials than in formaldehyde-fixed, paraffinembedded specimens. The results are discussed in relation to pulmonary and female-genital-tract diseases.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Ammann RW, Berk, JE, Fridhandler L, Ueda M, Wegmann W (1973) Hyperamylasemia with carcinoma of the lung. Ann Intern Med 78:521–525
Blaustein A (1981) Surface (germinal) epithelium and related ovarian neoplasms. Pathol Annu 16 (Part 1): 247–294
Bruns DE, Mills SE, Savory J (1982) Amylase in Fallopian tube and serous ovarian neoplasms. Immunohistochemical localization. Arch Pathol Lab Med 106:17–20
Clarke PD, Bain BC, Davies A, Levin GE, Lambert HP (1981) Aspiration in seriously ill patients: a study of amylase in bronchial secretions. J Clin Pathol 34:803–805
Corlette MB, Dratch M, Sorger K (1978) Amylase elevation attributable to an ovarian neoplasm. Gastroenterology 74:907–909
Flege JB (1966) Ruptured tubal pregnancy with elevated serum amylase levels. Arch Surg 92:397–398
Fukayama M, Hayashi Y, Koike M, Ogawa M, Kosaki G (1986) Immunohistochemical localization of pancreatic secretory trypsin inhibitor in fetal and adult pancreatic and extrapancreatic tissues. J Histochem Cytochem 34:227–235
Gomi K, Kameya T, Tsumuraya M, Shimosato Y, Zeze F, Abe K, Yoneyama T (1976) Ultrastructural, histochemical, and biochemical studies of two cases with amylase, ACTH, and β-MSH producing tumor. Cancer 38:1645–1654
Green CL (1957) Identification of alpha-amylase as a secretion of the human Fallopian tube and “tubelike” eithelium of Müllerian and mesonephric duct origin. Am J Obstet Gynecol 73:402–408
Harada K, Kitamura M (1971) Amylase isoenzyme in a patient with primary bronchial carcinoma. Med Biol 82:155–158 (in Japanese)
Hayakawa T, Kameya A, Mizuno R, Noda A, Kondo T, Hirabayashi N (1984) Hyperamylasemia with papillary serous cystadenocarcinoma of the ovary. Cancer 54:1662–1665
Hayashi Y, Nakayama T, Kitamura M (1975) Biochemical studies on tumor-producing amylase. Jpn J Clin Pathol 23:234 (in Japanese)
Hodes ME, Sisk CJ, Karn RC, Ehrlich CE, Lehrner LM, Roth LM, Morley DJ, Merritt AD (1985) An amylase-producing serous cystadenocarcinoma of the ovary. Oncology 42:242–247
Jirsova Z, Kraus R, Martinek J (1977) Ciliary vesicles as a form of ciliogenesis in the tubal epithelium. Fol Morphol 25:383–386
Maeda M, Otuki M, Yuu H, Saeki S, Yamasaki T, Baba S (1982) Salivary-type hyperamylasemia in primary lung cancer: observation of a possible precursor of the salivary-type isoamylase. Eur J Cancer Clin Oncol 18:123–128
McLean IW, Nakane PK (1974) Periodate-lysine-paraformaldehyde fixative. A new fixative for immunoelectron microscopy. J Histochem Cytochem 22:1077–1083
Mellersh A, Clark A, Hafiz S (1979) Inhibition of Neisseria gonorrhoeae by normal human saliva. Br J Vener Dis 55:20–23
Morohoshi T, Nakamura N, Hayashi K, Kanda M (1980) Amylase producing lung cancer. Electronmicroscopical and biochemical studies. Virchows Arch (Pathol Anat) 387:125–132
Nakayama T, Hayashi Y, Kitamura M (1976) Amylase isoenzyme found in lung and ovarian cancers. In: Fishman WH, Sell S (eds) Onco-developmental gene expression. Academic Press, New York, pp 455–462
Otsuki M, Yuu H, Maeda M, Saeki S, Yamasaki T, Baba S (1977) Amylase in the lung. Cancer 39:1656–1663
Roberts I, Chopra S, Warshaw AL (1982) Carcinoma of the lung with marked hyperamylasemia and elevated serum calcitonin. Am J Gastroenterol 77:43–44
Robles F, López de la Osa E, Lerner U, Johannisson E, Brenner P, Hagenfeldt K, Diczfalusy E (1972) α-Amylase, glycogen synthetase and phosphorylase in the human endometrium: influence of the cycle and of the Cu-T device. Contraception 6:373–384
Shear M, Pearse AGE (1963) A starch substrate film method for the histochemical localization of amylase. Exp Cell Res 32:174–177
Skude G, Mårdh PA, Weström L (1976) Amylases of the genital tract. I. Isoamylases of genital tract tissue homogenate and peritoneal fluid. Am J Obstet Gynecol 126:652–656
Sternberger LA (1979) Immunocytochemistry, 2nd edn. John Wiley and Sons, New York
Yamashita K, Tachibana Y, Nakayama T, Kitamura M, Endo Y, Kobata A (1980) Structural studies of the sugar chains of human parotidα-amylase. J Biol Chem 255:5635–5642
Author information
Authors and Affiliations
Additional information
This work was supported by a grant from the Tokyo Metropolitan Government
Rights and permissions
About this article
Cite this article
Hayashi, Y., Fukayama, M., Koike, M. et al. Amylase in human lungs and the female genital tract. Histochemistry 85, 491–496 (1986). https://doi.org/10.1007/BF00508431
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00508431