Abstract
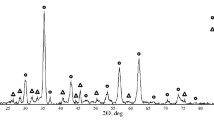
Chemical analysis shows that electric-arc-furnace (EAF) dust obtained from 13 steelmaking factories in Taiwan is composed chiefly of Fe (15–37%), Zn (7–28%) and Mn (1.55–3.99%) and that the mineral composition is mainly (Mn, Zn) Fe2O4, ZnO and ZnCl2·4Zn(OH)2·H2O. It was also found that EAF dust exists as irregular agglomerates from 3 to 20 μm in size and are made up of much smaller round particles from 0.3 to 1 μm in size. ZnCl2 gas in the dust condenses after passing through the EAF gas-cooler system binding small particles of Mn-Zn ferrite together. The agglutinative substance was identified as being ZnCl2·4Zn(OH)2·H2O possibly arising from the interaction between ZnCl2 and ZnO adhering to the Mn-Zn ferrite particles; a conjecture supported by the fact that EAF dust dissolves easily in hydrochloricacid solution leaving only loosely aggregated Mn-Zn ferrite material.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
L. L. Stephen, F. L. Wayne, G. S. Janet and W. K. Gary, Bureau of B Mines, Report of Investigation 8750 (1983).
R. Kaltenhauser, in “Disposal, recycling and recovery of electric furnace exhaust dust” edited by Iron & Steel Society AIME (AIME, 1987) 31.
W. M. Dressel, P. G. Barnard and M. M. Fine, US Bureau of Mines. Report of Investigation 7927 (1974).
E. G. Valdez and K. C. Dean, US Bureau of Mines. Report of Investigation 8000 (1975).
E. R. Krishnan and W. F. Kemner, in “Disposal, recycling and recovery of electric furnace exhaust dust”, edited by Iron and Steel Society, AIME (AIME, 1987) p. 77.
J. G. Eacott, M. C. Robinson, E. Busse, J. E. Burgener and P. E. Burgener, CIM Bull. (1984) 75.
J. Frenay, J. Hissel and S. Feriay, in “Recycling and secondary recovery of metals”, edited by P. R. Taylor, H. Y. Sohn and N. Jarrett (The Metallurgical Society Inc., 1985) p. 195.
E. R. Krishnan, in Environ. Prog. (1983) 184.
B. K. Thomas and D. J. Fray, Metall. Trans. B 12 (1981) 281.
M. C. Jha and P. C. Duyvesteyn, in “Recycling and secondary recovery of metals”, edited by P. R. Taylor, H. Y. Sohn and N. Jarrett (The Metallurgical Society Inc., 1985) p. 143.
D. J. Fray, Trans. Inst. Min. Metall. 95 (1986) 55.
O. K. Srivastava and E. A. Secco, Can. J. Chem. 45 (1967) 579.
J. A. Gadsden, in “Infrared spectra of minerals and related inorganic compounds” (Butterworths, (1975) pp. 14–16, 48, 117.
U. Enz, in “Magnetism and magnetic materials: Historical developments,” Vol. 3, edited by E. P. Wonlfarth (1982) p. 285.
Yutaka Tamaura, Perlas V. Buduan and Takashi Katsura, J. Chem. Soc. Dalton Trans. (1981) 1807.
O. K. Srivastava and E. A. Secco, Can. J. Chem. 45 (1967) 585.
W. Riesenkampf, M. Hamakiewicz, S. Balogopalan, S. Jasienska, J. Korecki and Z. Obuszko, Trans. Inst. Min. Metal. 93 (1984) 59.
H. E. Mcgannon, in “The making, shaping and treating of steel”, 9th Edition (United States Steel, 1970) p. 856.
M. Windholz, S. Budavari, F. B. Rosemary and S. O. Elizabeth in “The Merck Index — An encyclopedia of chemicals, drugs and biologicals”, 10th Edition (1983) pp. 1456, 7.
J. Leidner, in “Plastics waste-recovery of economic value” (Marcel Dekker, New York and Basel, 1981) p. 293.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Li, CL., Tsai, MS. A crystal phase study of zinc hydroxide chloride in electric-arc-furnace dust. Journal of Materials Science 28, 4562–4570 (1993). https://doi.org/10.1007/BF00414243
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF00414243