Abstract
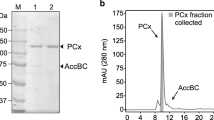
Pyruvate decarboxylase (EC 4.1.1.1) from the ethanol producing bacterium Zymomonas mobilis was purified to homogeneity. This enzyme is an acidic protein with an isoelectric point of 4.87 and has an apparent molecular weight of 200,000±10,000. The enzyme showed a single band in sodium dodecylsulfate gel electrophoresis with a molecular weight of 56,500±4,000 which indicated that the enzyme consists of four probably identical subunits. The dissociation of the cofactors Mg2+ and thiamine pyrophosphate at pH 8.9 resulted in a total loss of enzyme activity which could be restored to 99.5% at pH 6.0 in the presence of both cofactors. For the apoenzyme the apparent K m values for Mg2+ and thiamine pyrophosphate were determined to be 24 μM and 1.28 μM. The apparent K m value for the substrate pyruvate was 0.4 mM. Antiserum prepared against this purified pyruvate decarboxylase failed to crossreact with cell extracts of the reportedly pyruvate decarboxylase positive bacteria Sarcina ventriculi, Erwinia amylovora, or Gluconobacter oxydans, or with cell extracts of Saccharomyces cerevisiae.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Abbreviations
- Tris-buffer:
-
0,01 M tris-HCl buffer, containing 1 mM MgCl2 0.1 mM EDTA, 1.0 mM thiamine pyrophosphate, 2 mM mercaptopropanediol, pH 7.0
References
Balla H, Ullrich J (1980) Wheat germ pyruvate decarboxylase: improved purification and properties in comparison with the yeast enzyme. Hoppe-Seyler's Z Physiol Chem 361:1265
Bauchop T, Dawes EA (1959) Metabolism of pyruvic and formic acids of Zymosarcina ventriculi. Biochim Biophys Acta 36: 294–296
Boiteux A, Hess B (1970) Allosteric properties of yeast pyruvate decarboxylase. FEBS Lett 9:293–296
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Bräu B, Sahm H (1986) Cloming and expression of the structural gene for pyruvate decarboxylase of Zymononas mobilis in Escherichia coli. Arch Microbiol 144:296–301
Bringer S, Sprey B, Sahm H (1979) Purification and properties of alcohol oxidase from Poria contigua. Eur J Biochem 101: 563–570
Bringer S, Finn RK, Sahm H (1984a) Effect of oxygen on the metabolism of Zymomonas mobilis. Arch Microbiol 139:376–381
Bringer S, Sahm H, Swyzen W (1984b) Ethanol production by Zymomonas mobilis and its application on an industrial scale. Biotechnol Bioeng Symp 14:311–319
Dawes EA, Ribbons DW, Large PJ (1966) The route of ethanol formation in Zymomonas mobilis. Biochem J 98:795–803
DeLey J, Schell J (1962) Lactate and pyruvate catabolism in acetic acid bacteria. J Gen Microbiol 29:589–601
Finn RK, Bringer S, Sahm H (1984) Fermentation of arabinose to ethanol by Sarcina ventriculi. Appl Microbiol Biotechnol 19:161–166
Gounaris AD, Turkenkopf J, Civerchia LL, Greenlie J (1975) Pyruvate decarboxylase. III. Specificity restrictions for thiamine pyrophosphate in the protein association step, sub-unit structure. Biochim Biophys Acta 405:492–499
Green AA, Hughes WL (1955) Protein fractionation on the basis of solubility in aqueous solutions of salts and organic solvents. In: Colowick SP, Kaplan NO (eds) Methods in enzymology, vol 1. Academic Press, New York London, pp 67–90
Haq A (1984) Occurrence of pyruvate decarboxylase in Erwinia amylovora. Pakistan J Sci Ind Res 27:8–13
Haq A, Dawes EA (1971) Pyruvic acid metabolism and ethanol formation in Erwinia amylovora. J Gen Microbiol 68:295–306
Holzer H, Beaucamp K (1961) Nachweis und Charakterisierung von α-Lactyl-Thiaminpyrophosphat (“aktives Pyruvat”) und α-Hydroxyäthyl-Thiaminpyrophosphat (“aktiver Acetaldehyd”) als Zwischenprodukte der Decarboxylierung von Pyruvat mit Pyruvatdecarboxylase aus Bierhefe. Biochim Biophys Acta 46:225–243
Hopmann RFW (1980) Hydroxyl-ion-induced subunit dissociation of yeast cytoplasmic pyruvate decarboxylase. Eur J Biochem 110:311–318
Hoppner TC, Doelle HW (1983) Purification and kinetic characteristics of pyruvate decarboxylase and ethanol dehydrogenase from Zymomonas mobilis in relation to ethanol production. Eur J Appl Microbiol Biotechnol 17:152–157
King TE, Cheldelin VH (1954) Pyruvic carboxylase of Acetobacter suboxydans. J Biol Chem 208:821–831
Laemmli UK (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227:680–685
Lee KJ, Skotnicki ML, Tribe DE, Rogers PL (1981) The effect of temperature on the kinetics of ethanol production by strains of Zymomonas mobilis. Biotechnol Lett 3:291–296
Maurer HR (1971) Gel systems and their composition. In: Maurer HR (ed) Disc electrophoresis. De Gruyter, Berlin New York, pp 41–52
McGill DJ, Dawes EA (1971) Glucose and fructose metabolism in Zymomonas anaerobia. Biochem J 125:1050–1068
Morey AV, Juni E (1968) Studies on the nature of the binding of thiamine pyrophosphate to enzymes. J Biol Chem 243:3009–3019
Neuberg C, Karczag L (1911) Über zuckerfreie Hefegärungen. IV. Carboxylase, ein neues Enzym der Hefe. Biochem Z 36:68–81
Rhigetti P, Drysdale JW (1971) Isoelectric focusing in polyacrylamide gels. Biochim Biophys Acta 236:17–28
Rogers PL, Lee KJ, Skotnicki ML, Tribe DE (1982) Ethanol production by Zymomonas mobilis. Adv Biochem Eng 23: 37–84
Schellenberger A, Winter K, Hübner G, Schwaiberger R, Helbig D, Schumacher S, Thieme R, Bouillon G, Rädler K-P (1966) Rekombinations- und Funktionskintik der Hefe-Pyruvat-Decarboxylase. Z Physiol Chem 346:123–147
Scrutton MC (1971) Assay of enzymes of CO2 metabolism. In: Morris JR, Ribbons DW (eds) Methods in enzymology, vol 6A. Academic press, New York San Francisco London, pp 479–541
Singer TP, Pensky J (1952) Isolation and properties of the α-carboxylase of wheat germ. J Biol Chem 196:375–388
Stephens PE, Darlison MG, Lewis HM, Guest JR (1983) The pyruvate dehydrogenase EC 1.2.4.1 complex of Escherichia coli K-12 nucleotide sequence encoding the pyruvate dehydrogenase component. Eur J Biochem 133:155–162
Stephenson MP, Dawes EA (1971) Pyruvic acid and formic acid metabolism in Sarcina ventriculi and the role of ferredoxin. J Gen Microbiol 69:331–343
Stollar D, Levine L (1963) Two-dimensional immunodiffusion. In: Colowick SP, Kaplan NO (eds) Methods in enzymology, vol 6. Academic Press, New York London, pp 848–854
Swings J, DeLey J (1977) The biology of Zymomonas. Bacteriol Rev 41:1–46
Ullrich J (1970) Yeast pyruvate decarboxylase (2-oxoacid carboxylyase, EC 4.1.1.1). Assay of thiamine pyrophosphate. In: McCormick DB, Wright LD, (eds) Methods in enzymology, vol 18. Academic Press, New York London, pp 109–115
Ullrich J (1974) Thiaminpyrophosphat. In: Bergmeyer HU (ed) Methoden der enzymatischen Analyse, Band 2. Verlag Chemie, Weinheim, pp 2236–243
Ullrich J (1982) Structure-function relationships in pyruvate decarboxylase of yeast and wheat germ. Ann NY Acad Sci 378: 287–305
Vennesland B (1951) Keto acid decarboxylases In: Boyer PD, Lardy H, Myrbäck K (eds) The enzymes, vol 2. Academic Press, New York London, pp 183–215
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Bringer-Meyer, S., Schimz, K.L. & Sahm, H. Pyruvate decarboxylase from Zymomonas mobilis. Isolation and partial characterization. Arch. Microbiol. 146, 105–110 (1986). https://doi.org/10.1007/BF00402334
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00402334