Summary
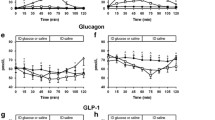
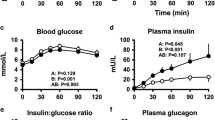
Carbohydrate ingestion results in a fall in glucagon concentration in non-diabetic but not in diabetic individuals. To determine if, and the mechanism by which, lack of postprandial suppression of glucagon contributes to hyperglycaemia, nine subjects with insulin-dependent diabetes mellitus (IDDM) ingested 50 g of glucose containing both [2-3H] glucose and [6-3H] glucose on two occasions. [6-14C] glucose, insulin and low-dose somatostatin were infused intravenously at the same rates on both occasions. A basal glucagon infusion was started either at the same time (“constant glucagon”) or 2 h following (“suppressed glucagon”) glucose ingestion. This resulted in lower (p<0.001) glucagon concentrations during the first 2 h of the suppressed than during the constant glucagon study days (63±1 vs 108±2 pg/ ml). Lack of suppression of glucagon led to higher (p<0.01) postprandial glucose concentrations (10.3±0.9 vs 8.1±0.7 mmol/l) and a greater (p<0.02) integrated glycaemic response. The excessive rise in glucose was due to higher (p<0.02) rates of postprandial hepatic glucose release during the constant than during the suppressed glucagon study days, whether measured using either [6-3H] glucose (2.6±0.2 vs 2.0±0.2 mmol·kg−1 per 6 h) or [2-3H] glucose (3.0±0.3 vs 2.4±0.2 mmol·kg−1 per 6 h) as the meal tracer. Glucose disappearance, initial splanchnic glucose clearance and hepatic glucose cycling did not differ on the two occasions. Thus, the present studies demonstrate that lack of postprandial suppression of glucagon, by increasing hepatic glucose release, contributes to hyperglycaemia in subjects with IDDM.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Abbreviations
- IDDM:
-
Insulin-dependent diabetes mellitus
References
Unger RH (1978) Role of glucagon in the pathogenesis of diabetes: the status of the controversy. Metabolism 27:1691–1709
Lefebvre PJ, Luyckx AS (1979) Glucagon and diabetes: a reappraisal. Diabetologia 16:347–354
Stevenson RW, Steiner KE, Davis MA et al. (1987) Similar dose responsiveness of hepatic glycogenolysis and gluconeogenesis to glucagon in vivo. Diabetes 36:382–389
Lickley HLA, Kemmer FW, El-Tayeb KMA, Vranic M (1987) Importance of glucagon in the control of futile cycling as studied in alloxan-diabetic dogs. Diabetologia 30:175–182
Miyoshi H, Shulman GI, Peters EJ, Wolfe MH, Elahi D, Wolfe RR (1988) Hormonal control of substrate cycling in humans. J Clin Invest 81:1545–1555
Hers HG (1976) The control of glycogen metabolism in the liver. Annu Rev Biochem 45:167–187
Liljenquist JE, Rabin D (1979) Lack of a role for glucagon in the disposal of an oral glucose load in normal man. J Clin Endocrinol Metab 49:937–939
Holst JJ, Madsen OG, Knop J, Schmidt A (1977) The effect of intraportal and peripheral infusions of glucagon on insulin and glucose concentrations and glucose tolerance in normal man. Diabetologia 13:487–490
Sherwin RS, Fisher M, Hendler R, Felig P (1976) Hyperglucagonemia and blood glucose regulation in normal, obese and diabetic subjects. N Engl J Med 294:455–461
Raskin P, Unger RH (1978) Effect of insulin therapy on the profiles of plasma immunoreactive glucagon in juvenile-type and adult-type diabetes. Diabetes 27:411–419
Gerich JE, Tsalikian E, Lorenzi M et al. (1975) Normalization of fasting hyperglucagonemia and excessive glucagon responses to intravenous arginine in human diabetes mellitus by prolonged infusion of insulin. J Clin Endocrinol Metab 41:1178–1180
Tse TF, Clutter WE, Shah SD, Miller JP, Cryer PE (1983) Specificity, temporal relationships, and quantitative aspects. J Clin Invest 72:270–277
Butler RC, Rizza RA (1991) Contribution to postprandial hyperglycemia and effect on initial splanchnic glucose clearance of hepatic glucose cycling in glucose-intolerant or NIDDM patients. Diabetes 40:73–81
Dinneen S, Alzaid A, Miles J, Rizza R (1993) Metabolic effects of the nocturnal rise in cortisol on carbohydrate metabolism in normal humans. J Clin Invest 92:2283–2290
White NH, Skor D, Santiago JV (1982) Practical closed-loop insulin delivery. A system for the maintenance of overnight euglycemia and the calculation of basal insulin requirements in insulin-dependent diabetics. Ann Intern Med 97:210–213
Firth RG, Bell PM, Marsh HM, Hansen I, Rizza RA (1986) Postprandial hyperglycemia in patients with noninsulin-dependent diabetes mellitus. J Clin Invest 77:1525–1532
Katz H, Homan M, Jensen M, Caumo A, Cobelli C, Rizza R (1994) Assessment of insulin action in NIDDM in the presence of dynamic changes in insulin and glucose concentration. Diabetes 43:289–296
Eberts TJ, Sample RH, Glick MR, Ellis GH (1979) A simplified, colorimetric micromethod for xylose in serum or urine with phloroglucinol. Clin Chem 25:1440–1443
Bell PM, Firth RG, Rizza RA (1986) Assessment of insulin action in insulin-dependent diabetes mellitus using [6-14C]glucose, [3-3H]glucose, and [2-3H]glucose. Differences in the apparent pattern of insulin resistance depending on the isotope used. J Clin Invest 78:1479–1486
Steele R, Wall JS, De Bodo RC, Altszuler N, Kiang SP, Bjerknes C (1956) Measurement of size and turnover rate of body glucose pool by the isotope dilution method. Am J Physiol 187:15–24
Rizza R, Verdonk C, Miles J, Service J, Gerich J (1979) Effect of intermittent endogenous hyperglucagonemia on glucose homeostasis in normal and diabetic man. J Clin Invest 63:1119–1123
Bomboy JD Jr, Lewis SB, Lacy WW, Sinclair-Smith BC, Liljenquist JE (1977) Transient stimulatory effect of sustained hyperglucagonemia on splanchnic glucose production in normal and diabetic man. Diabetes 26:177–184
El-Refai M, Bergman RN (1979) Glucagon-stimulated glycogenolysis: time-dependent sensitivity to insulin. Am J Physiol 236:E246-E254
Cherrington AD, Diamond MP, Green DR, Williams PE (1982) Evidence for an intrahepatic contribution to the waning effect of glucagon on glucose production in the conscious dog. Diabetes 31:917–922
Rizza RA, Gerich JE (1979) Persistent effect of sustained hyperglucagonemia on glucose production in man. J Clin Endocrinol Metab 48:352–355
Fradkin J, Shamoon H, Felig P, Sherwin RS (1980) Evidence for an important role of changes in, rather than absolute concentrations of, glucagon in the regulation of glucose production in humans. J Clin Endocrinol Metab 50:698–703
Katz J, Rognstad R (1976) Futile cycles in the metabolism of glucose. In: Horecker BL, Stadtman ER (eds) Current topics in cellular regulation, Academic Press, New York, pp. 237–289
Wajngot A, Chandramouli V, Schumann WC, Kumaran K, Efendic S, Landau BR (1989) Testing of the assumptions made in estimating the extent of futile cycling. Am J Physiol 256:E668-E675
Efendic S, Wajngot A, Vranic M (1985) Increased activity of the glucose cycle in the liver: early characteristic of type 2 diabetes. Proc Natl Acad Sci USA 82:2965–2969
Jaspan JB, Ruddick J, Rayfield E (1984) Transhepatic glucagon gradients in man: evidence for glucagon extraction by human liver. J Clin Endocrinol Metab 58:287–292
Blackard WG, Nelson NC, Andrews SS (1974) Portal and peripheral vein immunoreactive glucagon concentrations after arginine or glucose infusions. Diabetes 23:199–202
Jaspan JB, Polonsky KS, Lewis M et al. (1981) Hepatic metabolism of glucagon in the dog: contribution of the liver to overall metabolic disposal of glucagon. Am J Physiol 240:E233-E244
Paolisso G, Scheen AJ, Albert A, Lefebvre PJ (1989) Effects of pulsatile delivery of insulin and glucagon in humans. Am J Physiol 257:E686-E696
Johansson C, Wisen O, Efendic S, UvnuÄs-Wallensten K (1981) Effects of somatostatin on gastrointestinal propagation and absorption of oral glucose in man. Digestion 22:126–137
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Dinneen, S., Alzaid, A., Turk, D. et al. Failure of glucagon suppression contributes to postprandial hyperglycaemia in IDDM. Diabetologia 38, 337–343 (1995). https://doi.org/10.1007/BF00400639
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF00400639