Summary
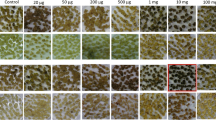
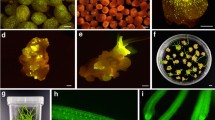
The combination in the nuclear genome of a dominant resistance marker (to select against unfused wild-type cells) and a recessive deficiency marker (to select against unfused mutant cells) in a cell line should provide a system for selecting fusion hybrids between the mutant line and any wild-type line. To test this idea, we fused protoplasts from a non-morphogenic cell line of Nicotiana tabacum which was kanamycin resistant (by transformation) and deficient in nitrate reductase (NR-K+) with protoplasts from N. tabacum cv. Petit Havana clone SR1, which provided resistance against streptomycin as an additional selectable marker (NR+K-SR+). Putative hybrids were selected using a culture medium containing no available reduced nitrogen source and 50 mg/l kanamycin sulphate. After regeneration into plants, the hybrid character was demonstrated from: (i) the morphological variation of the regenerants; (ii) the chromosome number; (iii) the ability to grow on medium without a reduced nitrogen source and containing kanamycin sulphate at 50 mg/l; (iv) the presence of nitrate reductase activity; (v) the presence of the gene coding for neomycin phosphotransferase, which provides resistance to kanamycin sulphate; (vi) callus formation from leaves on medium containing 1 g/l streptomycin or 50 mg/l kanamycin sulphate; (vii) F1 plants containing nitrate reductase and the gene for neomycin phosphotransferase. Fusions between the mutant cell line (NR-K+) and three wild-type tobacco species and subsequent cultivation on medium containing no available nitrogen source but 50 mg/l kanamycin sulphate resulted in callus formation with all combinations, while hybrid plants were only regenerated when N. sylvestris was the fusion partner.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Egger A (1986) Einfluß von NO2 auf Enzyme der assimilatorischen Sulfat- und Nitratreduktion in Nadeln von Fichte (Picea abies L.). Lizentiatsarbeit University of Bern, Switzerland
Fromm ME, Taylor LP, Walbot U (1986) Stable transformation of maize after gene transfer by electroporation. Nature 319:791–793
Gamborg OL (1970) The effect of amino acids and ammonium on the growth of plant cells in suspension culture. Plant Physiol 45:372–375
Hain R, Stabel P, Czernilofsky AP, Steinbiss L, Herrera-Estrella L, Schell J (1985) Uptake, integration, expression and genetic transmission of a selectable chimaeric gene by plant protoplasts. Mol Gen Genet 199:161–168
Hamill JD, Pental D, Cocking EC, Müller AJ (1983) Production of a nitrate reductase deficient streptomycin resistant mutant of Nicotiana tabacum for somatic hybridization studies. Heredity 50:197–200
Harms CT (1983) Somatic hybridization by plant protoplast fusion. In: Potrykus I, Harms CT, Hinnen A, Hütter R, King PJ, Shillito RD (eds) Protoplasts 1983, Lecture Proceedings. Birkhäuser, Basel p 69
Lazar GB (1983) Recent developments in plant protoplast fusion and selection technology. In: Potrykus I, Harms CT, Hinnen A, Hütter R, King PJ, Shillito RD (eds) Protoplasts 1983, Lecture Proceedings. Birkhäuser, Basel, p 61
Linsmaier EM, Skoog F (1965) Organic growth factor requirements of tobacco tissue cultures. Physiol Plant 18:100–127
Lörz H, Baker B, Schell J (1985) Gene transfer to cereal cells mediated by protoplast transformation. Mol Gen Genet 199:178–182
Maliga P, Sz-Breznovits A, Marton L (1973) Streptomycin-resistant plants from callus culture of haploid tobacco. Nature New Biol 244:29–30
Mendel RR, Müller AJ (1979) Nitrate reductase deficient mutant cell lines of Nicotiana tabacum. Further biochemical characterization. Mol Gen Genet 77:145–152
Mendel RR, Alikulov ZA, Lvov NP, Müller AJ (1981) Presence of the molybdenum cofactor in nitrate reductase — deficient mutant cell lines of Nicotiona tabacum. Mol Gen Genet 181:395–399
Müller AJ, Grafe R (1978) Isolation and characterization of cell lines of Nicotiana tabacum lacking nitrate reductase. Mol Gen Genet 161:67–76
Neyra CA, Hageman RH (1975) Nitrate uptake and induction of nitrate reductase in excised roots. Plant Physiol 56:692–695
Paszkowski J, Saul MW (1986) Direct gene transfer to plants. Methods Enzymol 118:668–684
Paszkowski J, Shillito RD, Saul M, Mandak V, Hohn T, Hohn B, Potrykus I (1984) Direct gene transfer to plants. EMBO J 3:2717–2722
Pental D, Hamill JD, Pirrie A, Cocking EC (1986) Somatic hybridization of Nicotiana tabacum and Petunia hybrida. Mol Gen Genet 202:342–347
Potrykus I, Shillito RD (1986) Protoplasts: Isolation, culture, plant regeneration. Methods Enzymol 118:549–578
Potrykus I, Jia J, Lazar GB, Saul M (1984) Hyoscyamus muticus + Nicotiana tabacum fusion hybrids selected via auxotroph complementation. Plant Cell Rep 3:68–71
Potrykus I, Shillito RD, Saul MW, Paszkowski J (1985) Direct gene transfer. State of the art and future potential. Plant Mol Biol Rep 3:117–128
Shillito RD, Paszkowski J, Potrykus I (1983) Agarose plating and a bead-type culture technique enable and stimulate development of protoplast-derived colonies in a number of plant species. Plant Cell Rep 2:244–247
Shillito RD, Saul MW, Paszkowski J, Müller M, Potrykus I (1985) High efficiency direct gene transfer to plants. Biotechnology 3:1099–1103
Author information
Authors and Affiliations
Additional information
Communicated by G. Melchers
Rights and permissions
About this article
Cite this article
Brunold, C., Krüger-Lebus, S., Saul, M.W. et al. Combination of kanamycin resistance and nitrate reductase deficiency as selectable markers in one nuclear genome provides a universal somatic hybridizer in plants. Mol Gen Genet 208, 469–473 (1987). https://doi.org/10.1007/BF00328141
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00328141