Abstract
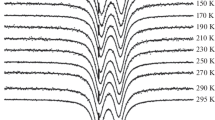
Electronic and Mössbauer absorption spectra and electron microprobe data are correlated for iron-bearing orthopyroxenes. The correlation provides a means of quantitatively determining the distribution of Fe2+ between the M(1) and M(2) sites of orthopyroxene crystals from electronic spectra and electron microprobe analysis. The electronic spectra are used to analyze the changes in the Fe2+ distribution produced during heating experiments and confirm earlier results from Mössbauer spectra. Two components of the spin-allowed transition of Fe2+ in the M(1) site are identified at about 13,000 cm−1 and 8,500 cm−1 in γ. Molar absorptivity (ɛ) values for all spin-allowed Fe2+ absorption bands in the near-infrared region are determined. The M(2) Fe2+ band at ∼5,000 cm−1 in β is the analytically most useful for site occupancy determinations. It remains linear with concentration (ɛ=9.65) over the entire compositional range. The band at ∼10,500 cm−1 in α is the most sensitive to M(2) Fe2+ concentration (ɛ=40.8), but deviates from linearity at high iron concentrations. The origins of spin-forbidden transitions in the visible region are examined.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Bloss, F.D.: An Introduction to the Methods of Optical Crystalography. New York: Holt Rinehart and Winston 1961
Burnham, C.W., Ohashi, Y., Hafner, S.S., Virgo, D.: Cation distribution and atomic thermal vibrations in an iron-rich orthopyroxene. Am. Mineral. 56, 850–875 (1971)
Burns, R.G.: Mineralogical Applications of Crystal Field Theory. Cambridge: Cambridge University Press 1970
Deer, W.A., Howie, R.A., Zussman, J.: An Introduction to the Rock-Forming Minerals. New York: John Wiley and Sons 1966
Evans, B.J., Ghose, S., Hafner, S.S.: Hyperfine splitting of Fe57 and Mg-Fe order-disorder in orthopyroxene (MgSiO3-FeSiO3) solid solution. J. Geol. 75, 306–322 (1967)
Ghose, S.: Mg2+-Fe2+ order in an orthopyroxene, Mg0.93Fe1.07Si2O6. Z. Krist. 122, 81–99 (1965)
Ghose, S., Hafner, S.: Mg2+-Fe2+ distribution in metamorphic and volcanic orthopyroxenes. Z. Krist. 125, 157–162 (1967)
Gibbons, R.V.: Experimental effects of high shock pressure on materials of geologic and geophysical interest. Ph. D. Thesis. California Institute of Technology (1974)
Goldman, D.S., Rossman, G.R.: Identification of a mid-infrared electronic absorption band of Fe2+ in the distorted M(2) site of orthopyroxene, (Mg, Fe)SiO3. Chem. Phys. Lett. 41 (3), 474–475 (1976)
Goldman, D.S., Rossman, G.R.: The spectra of iron in orthopyroxene revisited: The splitting of the ground state. Am. Mineral. 62, 151–157 (1977)
Howie, R.A.: Cell parameters of orthopyroxenes. Mineral. Soc. Am. Spec. Paper 1, 213–222 (1963)
Kleck, W.C.: Cavity minerals at Summit Rock, Oregon. Am. Mineral. 55, 1396–1404 (1970)
Langer, K., Abu-Eid, R.M.: Measurements of the polarized absorption spectra of synthetic transition metal-bearing silicate microcrystals in the spectral range 44,000–4,000 cm−1. Phys. Chem. Minerals 1, 273–299 (1977)
Runciman, W.A., Sengupta, D., Marshall, M.: The polarized spectra of iron in silicates. I Enstatite. Am. Mineral. 58, 444–450 (1973)
Saxena, S.K., Ghose, S.: Mg2+-Fe2+ order-disorder and the thermodynamics of the orthopyroxene crystalline solution. Am. Mineral. 56, 532–559 (1971)
Virgo, D., Hafner, S.S.: Re-evaluation of the cation distribution in orthopyroxenes by the Mössbauer effect. Earth Planet Sci. Lett. 4, 265–269 (1968)
Virgo, D., Hafner, S.S.: Fe2+, Mg order-disorder in heated orthopyroxenes. Mineral. Soc. Am. Spec. Paper 2, 67–81 (1969)
Virgo, D., Hafner, S.S.: Fe2+, Mg order-disorder in natural orthopyroxenes. Am. Mineral. 55, 201–223 (1970)
Author information
Authors and Affiliations
Additional information
Contribution No. 3058
Rights and permissions
About this article
Cite this article
Goldman, D.S., Rossman, G.R. Determination of quantitative cation distribution in orthopyroxenes from electronic absorption spectra. Phys Chem Minerals 4, 43–53 (1979). https://doi.org/10.1007/BF00308358
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00308358