Abstract
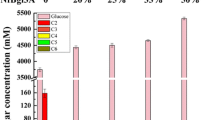
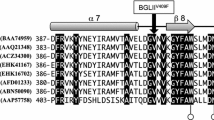
Hydrolytic enzymes from the filamentous fungus Trichoderma harzianum have been described as critical elements of the mycoparasitic action of Trichoderma against fungal plant pathogens. In this report we describe the first genomic and cE)NA clones encoding a β-1,6-endoglucanase gene. The deduced protein sequence has limited homology with other β-glucanases. Northern experiments show a marked repression of mRNA accumulation by glucose. The protein has been successfully produced in Saccharomyces cerevisiae upon construction of a transcriptional fusion of the cDNA with a yeast promoter. This S. cerevisiae recombinant strain shows a strong lytic action on agar plates containing β-1,6-glucan.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Ausubel FM, Brent R, Kington RE, Moore DD, Seidman JG, Smith JA, Struhl K (eds) (1989) Current Protocols in Molecular Biology. Greene and Wiley-Interscience, New York
Blaiseau PL, Lafay JF (1992) Primary structure of a chitinase-encoding gene (chit) from the filamentous fungus Aphanocladium album. Gene 120: 243–248
Cesarini G, Murray JAH (1987) Genetic engineering, principles and methods, vol 9. Plenum Press, New York
Chambers RS, Broughton MJ, Cannon RD, Carne A, Emerson GW, Sullivan PA (1993) An exo-β-(1,3)-glucanase of Candida albicans: purification of the enzyme and molecular cloning of the gene. J Gen Microbiol 139: 325–334
Chen L, Fincher GB, Hoj PB (1993) Evolution of polysaccharide hydrolase substrate specificity. Catalytic amino acids are conserved in barley 1,3-1,4- and 1,3-β-glucanases. J Biol Chem 268: 13318–13326
Chérif M, Benhamou N (1990) Cytochemical aspects of chitin breakdown during the parasitic action of a Trichoderma sp. on Fusarium oxysporum f. sp. radicis-licopersici. Phytopathology 80: 1406–1414
Dawson C, Belloch C, García-López MD, Uruburu F (1990) Catalogue of strains, Spanish Type Culture Collection. University of Valencia, Valencia, Spain
De la Cruz J (1994) PhD thesis. University of Sevilla, Spain
De la Cruz J, Hidalgo-Gallego A, Lora JM, Benitez T, Pintor-Toro JA, Llobell A (1992) Isolation and characterization of three chitinases from Trichoderma harzianum. Eur J Biochem 206: 859–867
De la Cruz J, Rey M, Lora JM, Hidalgo-Gallego A, Dominguez F, Pintor-Toro JA, Llobell A, Benitez T (1993) Carbon source control on β-glucanases, chitobiase and chitinase from Trichoderma harzianum. Arch Microbiol 159: 316–322
Devereux J, Haeberli P, Smithies O (1984) A comprehensive set of sequence analysis for the VAX. Nucleic Acids Res 12: 387–395
Fuller RS, Brake A, Thorner J (1989) Yeast prohormone processing enzyme (KEX2 gene product) is a Ca2+-dependent serine protease. Proc Natl Acad Sci USA 86: 1434–1438
García I, Lora JM, De la Cruz J, Benítez T, Llobell A, Pintor-Toro JA (1994) Cloning and characterization of a chitinase (CHIT42) cDNA from the mycoparasitic fungus Trichoderma harzianum. Curr Genet, in press
Geremía RA, Glodman GH, Jacobs D, Ardiles W, Vila SB, van Montagu M, Herrera-Estrella A (1993) Molecular characterization of a proteinase-encoding gene, pbr1, related to mycoparasitism by Trichoderma harzianum. Mol Microbiol 8: 603–613
von Heijne G (1983) Patterns of amino acids near signal-sequence cleavage sites. Eur J Biochem 133: 17–21
Ito H, Fukuda Y, Murata K, Kimura A (1983) Transformation of intact yeast cells treated with alkali cations. J Bacteriol 153: 163–168
Julius D, Brake A, Blair L, Kunisawa R, Thorner J (1984) Isolation of the putative structural gene for the lysine-arginine-cleaving endopeptidase required for processing of yeast prepro-α-factor. Cell 37: 1075–1089
Keitel T, Simon O, Borriss R, Heinemann U (1993) Molecular and active-site structure of a Bacillus 1,3-1,4-β-glucanase. Proc Natl Acad Sci USA 90: 5287–5291
Laemmli UK (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227: 680–685
Lora JM, De la Cruz J, Benitez T, Llobell A, Pintor-Toro JA (1994) A putative catabolite-repressed cell wall protein from the mycoparasitic fungus Trichoderma harzianum. Mol Gen Genet 242: 461–466
Macarron R, van Beeumen J, Henrissat B, De la Mata I, Claeyssens M (1993) Identification of an essential glutamate residue in the active site of endoglucanase III from Trichoderma reesei. FEBS Lett 316: 137–140
Matsudaira PT (ed) (1989) A practical guide to protein and peptide purification for microsequencing. Academic Press, New York
Mauch F, Mauch-Mani B, Boller T (1988) Antifungal hydrolases in pea tissue II. Inhibition of fungal growth by combinations of chitinase and βl-1,3-glucanase. Plant Physiol 88: 936–942
Muthukumar G, Suhng S-H, Magee PT, Jewell RD, Primerano DA (1993) The Saccharomyces cerevisiae SPR1 gene encodes a sporulation-specific exo-1,3-β-glucanase which contributes to ascospore thermoresistance. J Bacteriol 175: 386–394
Papavizas GC (1985) Trichoderma and Gliocladium: biology, ecology and potential for biocontrol. Annu Rev Phytopathol 23: 23–54
Park C-M, Bruenn JA, Chandrashekar G, Flurkey WF, Bozarth RF, Koltin Y (1994) Structure and heterologous expression of the Ustilago maydis viral toxin KP4. Mol Microbiol 11: 155–164
Rombouts FM, Phaff HJ (1976) Lysis of yeast cell walls. Lytic β-1,6-glucanase from Bacillus circulans. Eur J Biochem 63: 109–120
Sambrook J, Fritsch EF, Maniatis T (1989) Molecular cloning: a laboratory manual, 2nd edn. Cold Spring Harbor Laboratory Press, Cold Spring Harbor, New York
Seta-Buurlage MB, Ponstein AS, Vloemans SA, Melchers LS, Van den Elzen PJM, Cornelissen BJC (1993) Only specific chitinases and β-1,3-glucanases exhibit antifungal activity. Plant Physiol 101: 857–863
Shibata Y, Fujimbara T (1973) Enzymic hydrolysis of glucans containing β-1,3 and β-1,6-linkages: β-1,6-glucan hydrolase of fungi. J Ferment Technol 51: 216–226
Stark MJR, Boyd A (1986) The killer toxin of Kluyveromyces lactis: characterization of the toxin subunits and identification of the genes which encode them. EMBO J 5: 1995–2002
Tao J, Ginsberg I, Bauerjee N, Koltin Y, Held W, Bruenn JA (1990) The Ustilago maydis KP6 killer toxin: structure, expression in Saccharomyces cerevisiae and relationship to other cellular toxins. Mol Cell Biol 10: 1373–1381
Vázquez de Aldana CR, Correa J, San Segundo P, Bueno A, Nebrada AR, Méndez E, Del Rey F (1991) Nucleotide sequence of the exo-β-1,3-glucanase-encoding gene, EXG1, of the yeast Saccharomyces cerevisiae. Gene 97: 173–182
Watanabe T, Kobori K, Miyashita K, Fujii T, Sakai H, Uchida M, Tanaka H (1993) Identification of glutamic acid 204 and aspartic acid 200 in chitinase A1 of Bacillus circulans WL-12 as essential residues for chitinase activity. J Biol Chem 268: 18567–18572
Yamamoto S, Kobayashi R, Nagasaki S (1974) Purification and Properties of an endo β-1,6-glucanase from Rhizopus chinensis R-69. Agric Biol Chem 38: 1493–1500
Author information
Authors and Affiliations
Additional information
Communicated by C. A. M. J. J. van den Hondel
Rights and permissions
About this article
Cite this article
Lora, J.M., De la Cruz, J., Llobell, A. et al. Molecular characterization and heterologous expression of an endo-β-1,6-glucanase gene from the mycoparasitic fungus Trichoderma harzianum . Molec. Gen. Genet. 247, 639–645 (1995). https://doi.org/10.1007/BF00290356
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00290356