Abstract
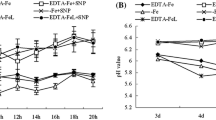
To understand the whole-plant processes which influence the Fe nutrition of developing seeds, we have characterized root Fe(III)-reductase activity and quantified whole-plant Fe balance throughout the complete 10-week (10-wk) life cycle of pea (Pisum sativum L., cv. Sparkle). Plants were grown hydroponically in complete nutrient solution with a continuous supply of chelated Fe; all side shoots were removed at first appearance to yield plants with one main shoot. Root Fe(III)-reductase activity was assayed with Fe(III)-EDTA. Flowering of the experimental plants began on wk 4 and continued until wk 6; seed growth and active seed import occurred during wks 5–10. Vegetative growth terminated at wk 6. Iron(III) reduction in whole-root systems was found to be dynamically modulated throughout the plant's life cycle, even though the plants were maintained on an Fe source. Iron(III)-reductase activity ranged from 1–3 μmol Fe reduced · g −1 DW · h−1 at early and late stages of the life cycle to 9.5 μmol Fe reduced · g−1 DW · h−1 at wk 6. Visual assays demonstrated that Fe(III)-reductase activity was localized to extensive regions of secondary and tertiary lateral roots during this peak activity. At midstages of growth (wks 6–7), root Fe(III)-reductase activity could be altered by changes in internal shoot Fe demand or external root Fe supply: removal of all pods or interruption of phloem transport from the reproductive portion of the shoot (to the roots) resulted in lowered root Fe(III)-reductase activity, while removal of Fe from the nutrient solution resulted in a stimulation of this activity. Total shoot Fe content increased throughout the 10-wk growth period, with Fe content in the non-seed tissues of the shoot declining by 50% of their maximal level and accounting for 35% of final seed Fe content. At maturity, total seed Fe represented 74% of total shoot Fe; total Fe in the roots (apoplasmic and symplasmic Fe combined) was minimal. These studies demonstrate that the root Fe(III)-reductase system responds to Fe status and/or Fe requirements of the shoot, apparently through shoot-to-root communication involving a phloem-mobile signal. During active seed-fill, enhanced root Fe(III)-reductase activity is necessary to generate sufficient Fe2+ for continued root Fe acquisition. This continuing Fe supply to the shoot is essential for the developing seeds to attain their Fe-content potential. Increased rates of root Fe(III) reduction would be necessary for seed Fe content to be enhanced in Pisum sativum.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Abbreviations
- BPDS:
-
bathophenanthrolinedisulfonic acid
- DAF:
-
days after flowering
- DW:
-
dry weight
- EDDHA:
-
N,N′-ethylenebis[2-(2-hydroxyphenyl)-glycine]
- wk:
-
week
References
Bienfait, H.F. (1988) Proteins under the control of the gene for Fe-efficiency in tomato. Plant Physiol. 88, 785–787
Bienfait, H.F. (1989) Prevention of stress in iron metabolism of plants. Acta Bot. Neerl. 38, 105–129
Bienfait, H.F., Bino, R.J., van der Bliek, A.M., Duivenvoorden, J.F., Fontaine, J.M. (1983) Characterization of ferric reducing activity in roots of Fe-deficient Phaseolus vulgaris. Physiol. Plant. 59, 196–202
Bienfait, H.F., de Weger, L.A., Kramer, D. (1987) Control of the development of iron-efficiency reactions in potato as a response to iron deficiency is located in the roots. Plant Physiol. 83, 244–247
Bienfait, H.F., van den Briel, W., Mesland-Mul, N.T. (1985) Free space iron pools in roots. Generation and mobilization. Plant Physiol. 78, 596–600
Brüggemann, W., Moog, P.R., Nakagawa, H., Janiesch, P., Kuiper, P.J.C. (1990) Plasma membrane-bound NADH:Fe3+ -EDTA reductase and iron deficiency in tomato (Lycopersicon esculentum). Is there a Turbo reductase? Physiol. Plant. 79, 339–346
Chaney, R.L., Brown, J.C., Tiffin, L.O. (1972) Obligatory reduction of ferric chelates in iron uptake by soybeans. Plant Physiol. 50, 208–213
Grusak, M.A. (1994) Iron transport to developing ovules of Pisum sativum. I. Seed import characteristics and phloem iron-loading capacity of source regions. Plant Physiol. 104, 649–655
Grusak, M.A., Pezeshgi, S. (1994) Uniformly 15N-labeled soybean seeds produced for use in human and animal nutrition studies: Description of a recirculating hydroponic growth system and whole plant nutrient and environmental requirements. J. Sci. Food Agric. 64, 223–230
Grusak, M.A., Welch, R.M., Kochian, L.V. (1989) A transport mutant for the study of plant root iron absorption. In: Plant membrane transport: The current position, pp. 61–66, Dainty, J., De Michelis, M.I., Marrè, E., Rasi-Caldogno, F., eds. Elsevier Science Publishers, Amsterdam Oxford New York
Grusak, M.A., Welch, R.M., Kochian, L.V. (1990a) Physiological characterization of a single-gene mutant of Pisum sativum exhibiting excess iron accumulation. I. Root iron reduction and iron uptake. Plant Physiol. 93, 976–981
Grusak, M.A., Welch, R.M., Kochian, L.V. (1990b) Does iron deficiency in Pisum sativum enhance the activity of the root plasmalemma iron transport protein? Plant Physiol. 94, 1353–1357
Grusak, M.A., Kochian, L.V., Welch, R.M. (1993) Spatial and temporal development of iron(III) reductase activity in root systems of Pisum sativum (Fabaceae) challenged with iron-deficiency stress. Am. J. Bot. 80, 300–308
Guinel, F.C., LaRue, T.A. (1990) Formation of wall appositions in leaves and lateral roots of an iron-accumulating pea mutant. Can. J. Bot. 68, 1340–1348
Hocking, P.J., Pate, J.S. (1977) Mobilization of minerals to developing seeds of legumes. Ann. Bot. 41, 1259–1278
Holden, M.J., Luster, D.G., Chaney, R.L., Buckhout, T.J., Robinson, C. (1991) Fe3+-Chelate reductase activity of plasma membranes isolated from tomato (Lycopersicon esculentum Mill.) roots. Comparison of enzymes from Fe-deficient and Fe-sufficient roots. Plant Physiol. 97, 537–544
Isaac, R.A. (1990) Iron in plants. Colorimetric method. In: Official methods of analysis ofthe Association of Official Analytical Chemists, Fifteenth edn., p. 40, Helrich, K., ed. Association of Official Analytical Chemists, Inc., Arlington, Vir.
Jolley, V.D., Brown, J.C. (1991) Factors in iron-stress response mechanism enhanced by Zn-deficiency stress in Sanilac, but not Saginaw navy bean. J. Plant Nutr. 14, 257–265
Kneen, B.E., LaRue, T.A., Welch, R.M., Weeden, N.F. (1990) Pleiotropic effects of brz. A mutation in Pisum sativum (L.) cv ‘Sparkle’ conditioning decreased nodulation and increased iron uptake and leaf necrosis. Plant Physiol. 93, 717–722
Kochian, L.V. (1991) Mechanisms of micronutrient uptake and translocation in plants. In: Micronutrients in agriculture, 2nd edn., pp. 229–296, Mordvedt, J.J., Cox, F.R., Shuman, L.M., Welch, R.M., eds. Soil Science Society of America, Madison, Wis.
Landsberg, E.C. (1982) Shoot-root interactions in the regulation of Fe-deficiency stress-induced H+-efflux from sunflower roots. In: Plant nutrition 1982, Proceedings of the Ninth International Plant Nutrition Colloquium, pp. 310–315, Scaife, A., ed. Commonwealth Agricultural Bureaux, Slough
Landsberg, E.C. (1984) Regulation of iron-stress-response by whole-plant activity. J. Plant Nutr. 7, 609–621
Landsberg, E.C. (1986) Function of rhizodermal transfer cells in the Fe stress response mechanism of Capsicum annuum L. Plant Physiol. 82, 511–517
Lesuisse, E., Raguzzi, F., Crichton, P.R. (1987) Iron uptake by the yeast Saccharomyces cerevisiae: involvement of a reduction step. J. Gen. Microbiol. 133, 3229–3236
Maas, F.M., van de Wetering, D.A.M., van Beusichem, M.L., Bienfait, H.F. (1988) Characterization of phloem iron and its possible role in the regulation of Fe-efficiency reactions. Plant Physiol. 87, 167–171
Romera, F.J., Alcántara, E., de la Guardia, M.D. (1992) Role of roots and shoots in the regulation of the Fe efficiency responses in sunflower and cucumber. Physiol. Plant, 85, 141–146
Römheld, V., Marschner, H. (1983) Mechanism of iron uptake by peanut plants. I. FeIII reduction, chelate splitting, and release of phenolics. Plant Physiol. 71, 949–954
Römheld, V., Marschner, H. (1986) Mobilization of iron in the rhizosphere of different plant species. Adv. Plant Nutr. 2, 155–204
Schmidt, W., Janiesch, P., Brüggemann, W. (1990) FeEDTA reduction in roots of Plantago lanceolata by a NADH-dependent plasma membrane-bound redox system. J. Plant Physiol. 136, 51–55
Sijmons, P.C., Lanfermeijer, F.C., de Boer, A.H., Prins, H.B.A., Bienfait, H.F. (1984) Depolarization of cell membrane potential during trans-plasma membrane electron transfer to extracellular electron acceptors in iron-deficient roots of Phaseolus vulgaris L. Plant Physiol. 76, 943–946
Stephan, U.W., Scholz, G. (1993) Nicotianamine: mediator of transport of iron and heavy metals in the phloem? Physiol. Plant. 88, 522–529
Welch, R.M., Norvell, W.A., Schaefer, S.C., Shaff, J.E., Kochian, L.V. (1993) Induction of iron(III) and copper(II) reduction in pea (Pisum sativum L.) roots by Fe and Cu status: Does the root-cell plasmalemma Fe(III)-chelate reductase perform a general role in regulating cation uptake? Planta 190, 555–561
Yip, R. (1994) Iron deficiency: Contemporary scientific issues and international programmatic approaches. J. Nutr. 124, 1479S-90S
Author information
Authors and Affiliations
Corresponding author
Additional information
This project has been funded in part with federal funds from the U.S. Department of Agriculture, Agricultural Research Service under Cooperative Agreement number 58-6250-1-003. The contents of this publication do not necessarily reflect the views or policies of the U.S. Department of Agriculture, nor does mention of trade names, commercial products, or organizations imply endorsement by the U.S. Government. The author wishes to acknowledge S. Pezeshgi and K. Koch for their excellent technical assistance, L. Loddeke for editorial comments, and A. Gillum for assistance with the figures.
Rights and permissions
About this article
Cite this article
Grusak, M.A. Whole -root iron(III)-reductase activity throughout the life cycle of iron-grown Pisum sativum L. (Fabaceae): relevance to the iron nutrition of developing seeds. Planta 197, 111–117 (1995). https://doi.org/10.1007/BF00239946
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00239946